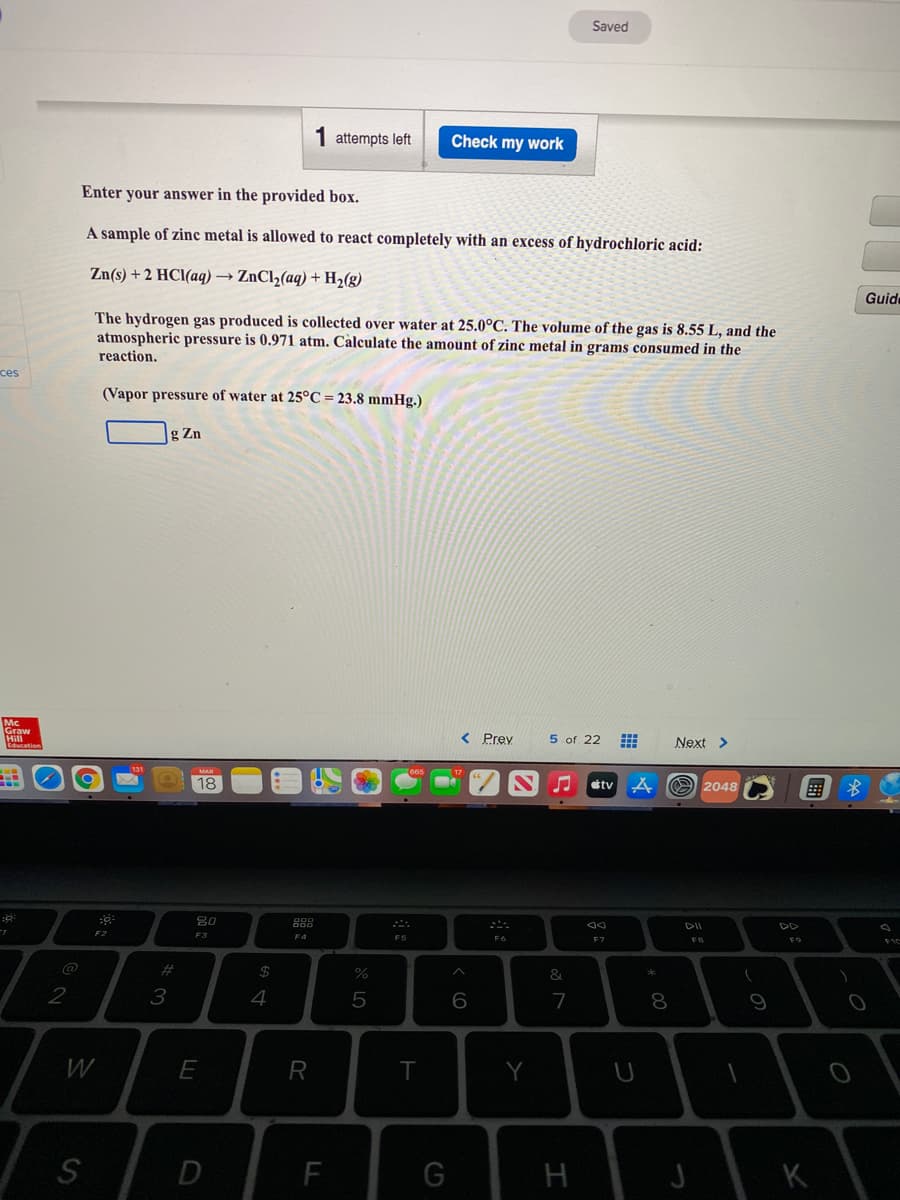
1 attempts left Check my work Enter your answer in the provided box. A sample of zine metal is allowed to react completely with an excess of hydrochloric acid: Zn(s) + 2 HCl(aq) → ZnCl2(aq) + H2(g) Guid The hydrogen gas produced is collected over water at 25.0°C. The volume of the gas is 8.55 L, and the atmospheric pressure is 0.971 atm. Càlculate the amount of zinc metal in grams consumed in the reaction. ces (Vapor pressure of water at 25°C = 23.8 mmHg.) g Zn
1 attempts left Check my work Enter your answer in the provided box. A sample of zine metal is allowed to react completely with an excess of hydrochloric acid: Zn(s) + 2 HCl(aq) → ZnCl2(aq) + H2(g) Guid The hydrogen gas produced is collected over water at 25.0°C. The volume of the gas is 8.55 L, and the atmospheric pressure is 0.971 atm. Càlculate the amount of zinc metal in grams consumed in the reaction. ces (Vapor pressure of water at 25°C = 23.8 mmHg.) g Zn
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter10: Gases And Their Properties
Section: Chapter Questions
Problem 96IL
Related questions
Question
100%
Transcribed Image Text:Saved
attempts left
Check my work
Enter your answer in the provided box.
A sample of zinc metal is allowed to react completely with an excess of hydrochloric acid:
Zn(s) + 2 HCI(aq) → ZnCl½(aq) + H2(g)
Guide
The hydrogen gas produced is collected over water at 25.0°C. The volume of the gas is 8.55 L, and the
atmospheric pressure is 0.971 atm. Càlculate the amount of zinc metal in grams consumed in the
reaction.
ces
(Vapor pressure of water at 25°C = 23.8 mmHg.)
g Zn
Graw
< Prey
5 of 22
Next >
18
tv
O 2048
888
DII
F2
F3
F4
F6
F7
FB
F9
F10
2#
$
%
*
3
4
7
8
W
E
R
T
Y
D
F
G
H
K
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co