Q: What coefficients would you need to balance the following equation, just put the coefficients in the…
A: • The given unbalanced chemical equation is, Mg + HCl → MgCl2 + H2
Q: Iron ore is impure, Fe2O3. When Fe2O3 is heated with an excess of carbon (coke), metallic iron and…
A:
Q: How many grams of \(\rm NH_3\) can be produced from 2.42 \({\rm mol}\) of \(\rm N_2\) and excess…
A: The reaction taking place will be N2 + H2 ------> NH3 Balancing : Since we have 2 N in LHS.…
Q: 1. Ag₂0, NaOH 2. H₂O H
A:
Q: Find the coefficients necessary to balance the following chemical equation: x,Cr,O¯ + »Fe*+ xH* xCr*…
A: The unbalanced redox reaction given is, x1 Cr2O72- + x2 Fe2+ + x3 H+ → x4 Cr3+ + x5 Fe3+ + x6 H2O…
Q: 1.003 g of soduim chloride was recovered in the reaction of Hydrochloric acid + Sodium carbonate →…
A: The reaction of hydrochloric acid with sodium carbonate forms sodium chloride, carbon dioxide and…
Q: Suppose a student starts with 2.4185 g of a sand mixture and separates the components into 1.3012 g…
A: The mass percentage is the simplest way of representing the mass of the components present in the…
Q: 2. You start with 2.46 g of KCIO345) and heat it. Use stoichiometry to calculate the mass (g) of KCI…
A:
Q: 7. Nitrogen + Oxygen ----> Dinitrogen Pentoxide + 3. Plumbic Chromate + Aluminum Sulfate ---->…
A: Nitrogen + Oxygen = dinitrogen pentaoxide While balancing the chemical equation, charge and mass…
Q: Q1: Calculate the mass needed to prepare 2.0L of 0.05M AGNO, (M. Mass 169.87g/mol) from its primary…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: 1-KNO,(ag) + H,0(1)
A: The treatment of acid with base leads to the formation of salt and water. The special name of this…
Q: How many milliliters of 2.212 M H2SO4 are needed to contain 0.82 moles of H,SO,?
A:
Q: 4. What mass of AgI can be produced from a 0.512-g sample that assays 20.1% AlI,?
A: Balanced equation could be written as All3 + 3Ag+ --> 3 Agl + Al3+ As shown in the equation, one…
Q: 4. (aq) (aq) BeSO, (aq) + H,O(1)
A: Given_____ (aq) + ____ (aq) → BeSO4(aq) + H2O (l)
Q: 1.960 g of the sulfate of a metal M2(SO4) 3 are transformed into the corresponding chloride, MCl3,…
A: Given information, Mass of M2(SO4)3 = 1.960 g Mass of MCl3 = 1.585 g Additional information; Mass of…
Q: A sample weighing 3.056 g is a mixture of Fe, O, (molar mass = 159.69 g/mol) and Al, 0, (molar mass…
A:
Q: The single largest use of phosphoric acid is in the production of fertilizer. How many grams of…
A: We have given that The single largest use of phosphoric acid is in the production of fertilizer.…
Q: Give a detailed mass-to-mass stoichiometry calculation that can be understood easily.
A: Given that - Mass of Seignette salt = 1.20 kgs = 1200 g Mass of cream of tartar = 1.5 kgs = 1500 g…
Q: 6.00 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have…
A:
Q: balance this chemical reaction ____ C6H12O6 → _____CO2 + _____ C2H6O
A: Given : ___ C6H12O6 → ____CO2 + ____C2H6O To balance :- The given equation by writing…
Q: Formaldehyde (CH2O, MM = 30.03) has a multitude of uses including tissue preservation. Formalin is a…
A: Molarity (M) :- Number of moles of solute dissolved in one litre of solution is known as molarity M…
Q: 13) NaOH, Br excess NH,
A: The following Reaction is Hoffman bromide Reaction in which we have to convert the Amide group into…
Q: (c) O 1) LIAIH4, Et₂O 2) H₂O+ 3) NaOH 4) CH, Br
A: 4. (a) Ans :The epoxide ring in presence of Grignard Reagent will undergo SN2 reaction with attack…
Q: D- Ba(CIo), (aq) + H1_0()
A: Given: Reaction To find: Complete the reaction Solution: Balanced equation is a equation in which…
Q: grams of Zn(OH), will be formed in
A:
Q: A new organic material is to be examined by combustion analysis. Burning 0.09303 g of this sample in…
A: The sample contains the percentage of, C = 40.63 % H = 5.08 % O = 54•28%
Q: c
A: 0.3146 g sample of a mixture of NaCl and KBr 0.6824 g of precipitate mixture of AgCl and AgBr
Q: of Br2, in grams, is required for complete reaction with 9.56 g of A g Br2 of white, solid Al,Bre is…
A: Given, Balanced chemical reaction, 2Al + 3Br2 ----> Al2Br3 Mass of Al = 9.56 g Molar mass of Al…
Q: NH, CH, h. HCI(g) i. NaCI SiC Cu
A: Attractive intermolecular forces are categorized into the following types: 1) Ionic bonding 2)…
Q: After titration, 25 mL of a vinegar solution was found to contain .03 moles of acetic acid. Using a…
A: Volume = 25 ml Moles of acetic acid = 0.03 M
Q: A sample weighing 3.056 g is a mixture of Fe, O, (molar mass = 159.69 g/mol) and Al, O, (molar mass…
A:
Q: Which of the following processes does NOT involve an increa . Melting of gold nuggets
A: In this question we have to find correct option and also explain reasonable point for statement.
Q: Write the empirical formula for at least four ionic compounds that could be formed from the…
A: The steps to write the empirical formula are shown in the next step
Q: 1.- Calculate-the mass ff Ba3(PO4)2 produced from the reaction of 0.85 g NazPO4:12H20 with excess…
A: The relationship between moles, mass and molar mass is: Moles = MassMolar mass
Q: Give the empirical formula for the following compounds: a) b) d) Paragraph BIU A + v GO ...
A: Molecular formula is the formula of a compound that gives the total number of atoms present in that…
Q: If 100.0 mL of 0.351 M Na, SO, are added to 100.0 mL of 0.905 M Pb(N0,),, how many grams of PbS0,…
A: Given, question belong to the Limiting reagent. The limiting reagent (or limiting reactant or…
Q: If someone grabbed medium porosity filter paper instead of fine porosity paper and you noticed…
A: Limiting reagent is the reactant that limits the chemical reaction and determines the amount of the…
Q: Balance the following equation by algebraic method. Li + H20 LIOH H2
A:
Q: If You want to prepare 600 grams of 0.5%sulfuric ácido substance do You need
A: Given : Mass of solution = 600 g And mass percent of sulfuric acid = 0.5 %
Q: Q1/ What is the weight, in milligrams, of 0.250 mmol Fe2O3 (ferric oxide),F.wt (159.7 mg/mmol)?
A:
Q: 4) + ? ? H20 HO,
A: Alkyl magnesium halide ( R-Mg-X ) is called Grignard reagent. Ketones reacts with Grignard reagent…
Q: 1. Get the percentage composition of C&H9NO2.
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: excess H2, Ni 2 mol Br2, CH,Cl2 \alcoholic AgNO3 1. O3 | 2. Zn, HCI
A: Given : Reaction of alkynes with different reagents To find: Complete all the Reaction Solution :…
Q: What is the mass percentage of C in CH,CH,OH?
A:
Q: (4) Ag20, NH,OH `H
A:
Q: If you used 19.0 mL of bleach (6.5 m/m % sodium hypochlorite), what is the actual mole of NaOCl (MW=…
A:
Q: A common method for analyzing the nickel content of a sample is to use a precipitation reaction…
A:
Q: Ammonium phosphate ((NH,), PO, is an important ingredlent in many fertilizers. It can be made by…
A:
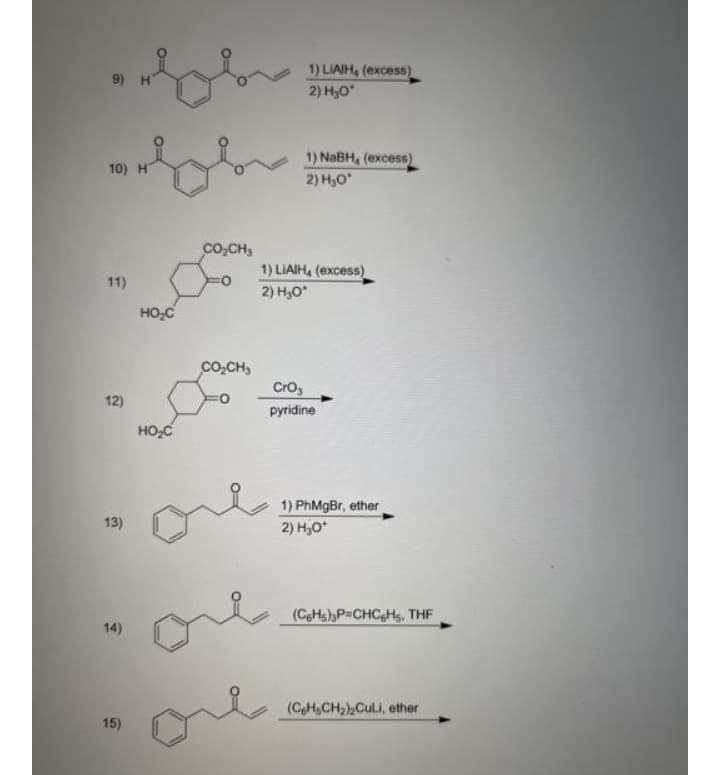
Step by step
Solved in 2 steps with 1 images

- Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2 144.3 158.9 Ca2+ (g) 1925.9 CaC2 (s) -59.8 -64.9 70.0 CaCO3 (s, calcite) -1206.9 -1128.8 92.9 CaCl2 (s) -795.8 -748.1 104.6 CaF2 (s) -1219.6 -1167.3 68.9 CaH2 (s) -186.2 -147.2 42.0 CaO (s) -635.1 -604.0 39.8 CaS (s) -482.4 -477.4 56.5 Ca(OH)2 (s) -986.1 -898.5 83.4 Ca(OH)2 (aq) -1002.8 -868.1 -74.5 Ca3(PO4)2 (s) -4126.0 -3890.0 241.0 CaSO4 (s) -1434.1 -1321.8 106.7 CaSiO3 (s) -1630.0 -1550.0 84.0 Carbon ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) C (s, graphite) 0 0 5.7 C (s, diamond) 1.9 2.9 2.4 C (g) 716.7 671.3 158.1 CCl4 (l) -135.4 -65.2 216.4 CCl4 (g) -102.9 -60.6 309.9 CHCl3 (l) -134.5 -73.7 201.7 CHCl3 (g) -103.1 -70.3 295.7 CH4 (g) -74.8 -50.7 186.3 CH3OH (g)…Please help with calulation of theoretical yield of the following: Na2CO3 + Ca(NO3)2.4H2O ---> CaCo3 + 2NaNo3 + 4H2O 2.03g Na2CO3 2.06g Ca(NO3)2.4H2OEl poder de rotación específico de la sacarosa a 20 °C es [alfa]D20 = +66,53 grados/(dm g mL-1). ¿Cuál es su poder de rotación molar, expresado en grados/(dm mol L-1)? (Peso molecular de la sacarosa: 342,3). 1). 5.14 2). 22.77 3). 194.4 4). 22773
- Recall that the formula for ΔHrxn = [(-Ccal)(moles of limiting reactant)/(ΔT)]If the actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value, will the value of Ccal increase, decrease, or remain the same? Please explain. How about if the actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value. However, the NaOH solution used in the determination of ΔHrxn was of the correct concentration. Will the magnitue of ΔHrxn increase, decrease, or remain the same? Please explain.3'-Methylphthalanilic acid is used commercially as a “fruit set” to prevent premature drop of apples, pears, cherries, and peaches from the tree. It is 70.58% carbon, 5.13% hydrogen, 5.49% nitrogen, and 18.80% oxygen. If eaten, the fruit set reacts with water in the body to produce an innocuous product, which contains carbon, hydrogen, and oxygen only, and m-toluidine (NH2C6H4CH3), which causes anemia and kidney damage. Compute the mass of the fruit set that would produce 5.23 g m-toluidine.After heating 2.000 g of MgSO4.7H2O to constant mass, the remaining residue in the crucible weighs 0.978 g. Using the data above , calculate the mass of water lost. After heating 2.000 g of MgSO4.7H2O to constant mass, the remaining residue in the crucible weighs 0.978 g. Using the data above , calculate the mass percent of water in the hydrate. 16 AND 17 PLEASE
- Na2SO3Na2SO3 reacts with HClO4HClO4 according to the reaction Na2SO3(aq)+2HClO4(aq)⟶2NaClO4(aq)+H2O(l)+SO2(g)Na2SO3(aq)+2HClO4(aq)⟶2NaClO4(aq)+H2O(l)+SO2(g) You wish to provide a 50% excess of 5.955.95 wt% aqueous HClO4HClO4 to react with 30.030.0 mL of 0.07740.0774 M Na2SO3Na2SO3. Calculate the mass of the 5.955.95 wt% aqueous HClO4HClO4 solution required to provide the 50% excess. mass:To my understanding that you are supposed to answer 3 questions per asking . Write clear and mine form as questions thank you(AH is all in kJ mol-1) AH 2 Na = 0 AH 2 H20 = -285.8 AH 2 NaOH = -470.1 AH H2 = 0
- Mass of original mixture: 0.280g Mass of naphthalene recovered: 0.160 g Mass of 3-nitroaniline recovered: 0.015 g Mass of benzoic acid recovered: 0.135 g 1. a) calculate % by mass of naphthalene, 3-nitroaniline and benzoic acid in original sample 1. b) calculate total mass of three recovered components (g) 1. c) calculate total percent recovery (%)In 6.75g of distillate >130C. After performing all necessary GC calculations, it is determined that this sample is 75.9% isopentyl acetate. What qas the actual mass(g) of isopentyl acetate that was actually in this sample? Answer to 2 decimal placesAleks data for AgBrO3 is 5.38 x 10^-5

