1-Phenylethanol undergoes an esterification with propionic anhydride in the presence of the enantioselective catalyst (S)-HBTM. The products of the reaction are analyzed by thin-layer chromatography (TLC). Which of the given statements about TLC are correct? The silica stationary phase is nontoxic, so TLC plates can be handled by their faces with bare hands. A comparison of the relative areas of the developed spots of the ester and unreacted alcohol can lead to an estimate of the amount of conversion. Assuming that there is 1.2 cm of ethyl acetate/hexanes solvent system at the bottom of the TLC tank, the ideal height at which to spot your samples on the TLC plate is 1.1 cm. The phosphomolybdic acid stains the spots green. The alcohol has a lower R; value than the ester product. The samples in lanes 1 and 2 should be spotted so close to each other that they are almost touching.
1-Phenylethanol undergoes an esterification with propionic anhydride in the presence of the enantioselective catalyst (S)-HBTM. The products of the reaction are analyzed by thin-layer chromatography (TLC). Which of the given statements about TLC are correct? The silica stationary phase is nontoxic, so TLC plates can be handled by their faces with bare hands. A comparison of the relative areas of the developed spots of the ester and unreacted alcohol can lead to an estimate of the amount of conversion. Assuming that there is 1.2 cm of ethyl acetate/hexanes solvent system at the bottom of the TLC tank, the ideal height at which to spot your samples on the TLC plate is 1.1 cm. The phosphomolybdic acid stains the spots green. The alcohol has a lower R; value than the ester product. The samples in lanes 1 and 2 should be spotted so close to each other that they are almost touching.
Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.SE: Something Extra
Problem 79AP
Related questions
Question
Please help me with both these questions. i am trying to study

Transcribed Image Text:1-Phenylethanol undergoes an esterification with propionic anhydride in the presence of the enantioselective catalyst
(S)-HBTM. The products of the reaction are analyzed by thin-layer chromatography (TLC).
Which of the given statements about TLC are correct?
The silica stationary phase is nontoxic, so TLC plates can be handled by their faces with bare hands.
A comparison of the relative areas of the developed spots of the ester and unreacted alcohol can lead to an estimate of
the amount of conversion.
Assuming that there is 1.2 cm of ethyl acetate/hexanes solvent system at the bottom of the TLC tank, the ideal height at
which to spot your samples on the TLC plate is 1.1 cm.
The phosphomolybdic acid stains the spots green.
The alcohol has a lower R; value than the ester product.
The samples in lanes 1 and 2 should be spotted so close to each other that they are almost touching.
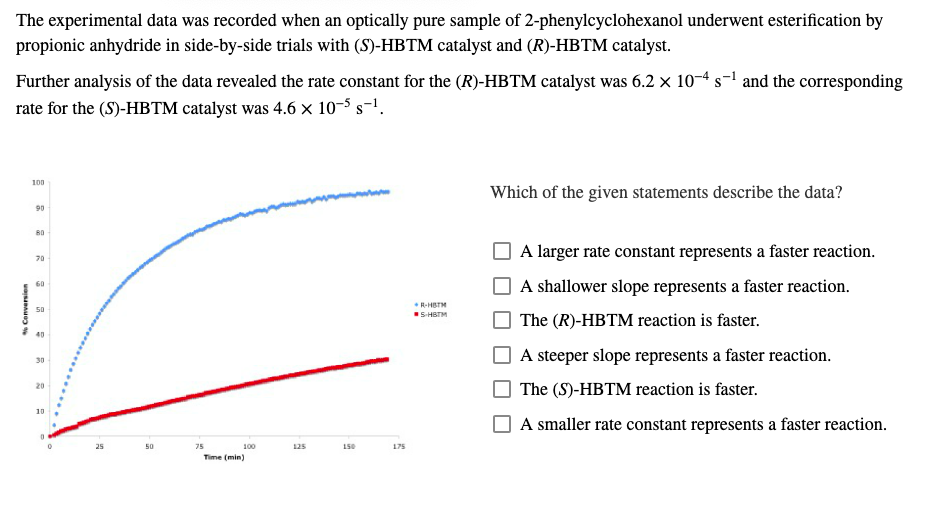
Transcribed Image Text:The experimental data was recorded when an optically pure sample of 2-phenylcyclohexanol underwent esterification by
propionic anhydride in side-by-side trials with (S)-HBTM catalyst and (R)-HBTM catalyst.
Further analysis of the data revealed the rate constant for the (R)-HBTM catalyst was 6.2 × 10-4 s-1 and the corresponding
rate for the (S)-HBTM catalyst was 4.6 × 10-5 s-1.
100
Which of the given statements describe the data?
90
B0
A larger rate constant represents a faster reaction.
70
A shallower slope represents a faster reaction.
60
R-HBTM
50
S-HBTM
The (R)-HBTM reaction is faster.
A steeper slope represents a faster reaction.
30
20
The (S)-HBTM reaction is faster.
10
A smaller rate constant represents a faster reaction.
25
50
75
100
125
150
175
Time (min)
Conversion
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning