1. cannot be separated by physical or chemical means. 2.Elements are abbreviated with which consist of one or two 3. Elements are organized on the according to their 4. Two or more elements chemically combined make up a 5.. is an example of an element and is an example of a compound. 6. Elements and compounds are called have a unique set of chemical and physical substances because they
1. cannot be separated by physical or chemical means. 2.Elements are abbreviated with which consist of one or two 3. Elements are organized on the according to their 4. Two or more elements chemically combined make up a 5.. is an example of an element and is an example of a compound. 6. Elements and compounds are called have a unique set of chemical and physical substances because they
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter12: The Alkaline Earths And The Halongens-two Families In The Periodic Table
Section: Chapter Questions
Problem 1ASA: As pure elements, all of the halogens are diatomic molecular species. Their melting points are: F2....
Related questions
Question
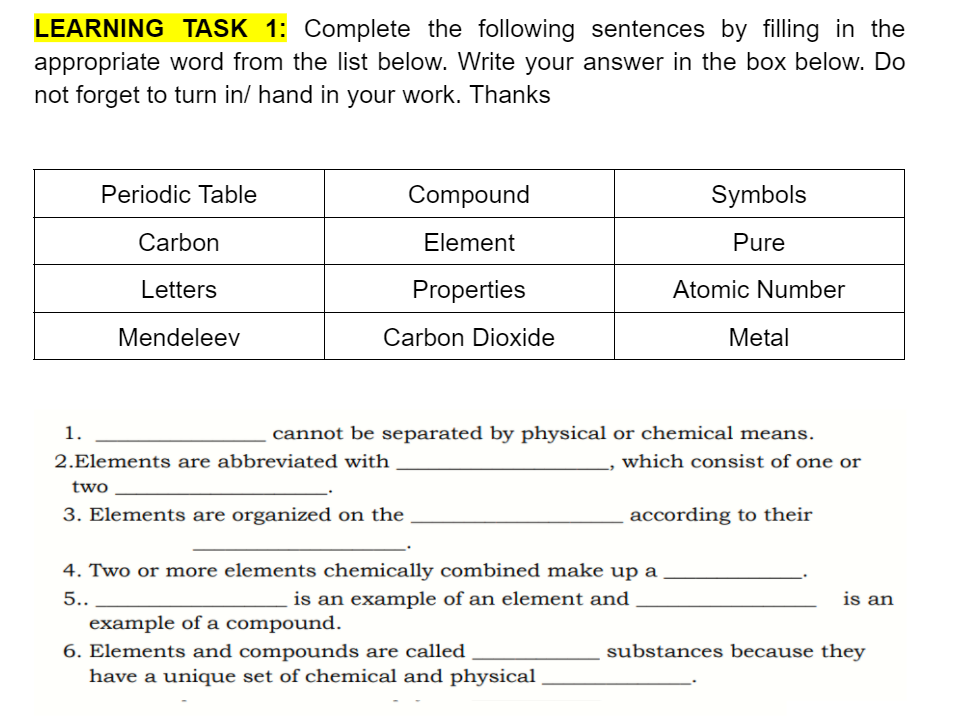
Transcribed Image Text:LEARNING TASK 1: Complete the following sentences by filling in the
appropriate word from the list below. Write your answer in the box below. Do
not forget to turn in/ hand in your work. Thanks
Periodic Table
Compound
Symbols
Carbon
Element
Pure
Letters
Properties
Atomic Number
Mendeleev
Carbon Dioxide
Metal
1.
cannot be separated by physical or chemical means.
2.Elements are abbreviated with
which consist of one or
two
3. Elements are organized on the
according to their
4. Two or more elements chemically combined make up a
5..
is an example of an element and
is an
example of a compound.
6. Elements and compounds are called
have a unique set of chemical and physical
substances because they
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning
