1. Consider the reaction: SO: + O1 -- SO. + 02. A rate study of this reaction was conducted at 298 K. The data that were obtained are shown in the table. (So mal. (0,). mall. Inisial Rute, mol(l. s) 0.25 0.40 0.25 0.20 0.118 0.75 0.20 L062 a. What is the over-all order with respect to SO, and 0? b. Write the rate law equation for this reaction. c. Determine the value and units of the rate constant, k.
1. Consider the reaction: SO: + O1 -- SO. + 02. A rate study of this reaction was conducted at 298 K. The data that were obtained are shown in the table. (So mal. (0,). mall. Inisial Rute, mol(l. s) 0.25 0.40 0.25 0.20 0.118 0.75 0.20 L062 a. What is the over-all order with respect to SO, and 0? b. Write the rate law equation for this reaction. c. Determine the value and units of the rate constant, k.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.33PAE: The following experimental data were obtained for the reaction of \'I14* and NOf in acidic solution....
Related questions
Question
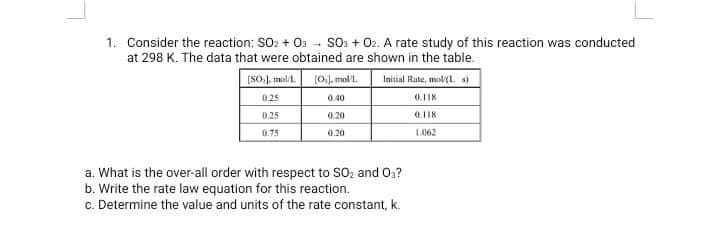
Transcribed Image Text:1. Consider the reaction: SO: + 01 - SO: + 02. A rate study of this reaction was conducted
at 298 K. The data that were obtained are shown in the table.
[SO). mal/
(0), mal.
Initial Rute, mal(l. s)
025
0.40
0.118
0.25
0.20
0.118
0.75
0.20
L062
a. What is the over-all order with respect to SOz and Oa?
b. Write the rate law equation for this reaction.
c. Determine the value and units of the rate constant, k.

Transcribed Image Text:2. One method for the destruction of ozone in the upper atmosphere is:
03 + NO → NO2 + 02 (slow)
NO2 + 0 + NO + 02 (fast)
overall rxn: 03 +0 → 202
a. Which species is an intermediate?
b. Which species is a catalyst?
c. Which is the rate-determining step (rds)?
d. Write the rate law for the reaction?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 18 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
