1. Knowing that these 2 molecules are always floating in the air, hypothesize why your distilled water ( which indeed was pure when put in the container and therefore should have been pH 7), instead had a pH between 5 and 6 (i.e. acidic, not neutral). 2. Hypothesize why the addition of sucrose did not change the pH of distilled water (a big hint to the answer is in the lab section on Acids & Bases).
1. Knowing that these 2 molecules are always floating in the air, hypothesize why your distilled water ( which indeed was pure when put in the container and therefore should have been pH 7), instead had a pH between 5 and 6 (i.e. acidic, not neutral). 2. Hypothesize why the addition of sucrose did not change the pH of distilled water (a big hint to the answer is in the lab section on Acids & Bases).
Chapter8: Forms Of Drugs And How They Act
Section: Chapter Questions
Problem 30RQ
Related questions
Question
100%
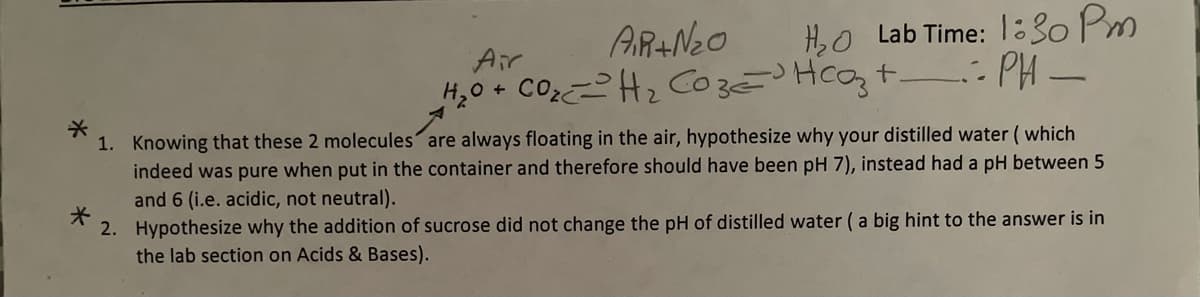
Transcribed Image Text:H,O Lab Time: 1:30 Pm
H,0 + COH2 CozHcozt : PH -
AR+Neo
Air
1. Knowing that these 2 molecules are always floating in the air, hypothesize why your distilled water ( which
indeed was pure when put in the container and therefore should have been pH 7), instead had a pH between 5
and 6 (i.e. acidic, not neutral).
2. Hypothesize why the addition of sucrose did not change the pH of distilled water (a big hint to the answer is in
the lab section on Acids & Bases).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you



Health Safety And Nutrition F/Young Child
Health & Nutrition
ISBN:
9781305144767
Author:
MAROTZ
Publisher:
Cengage



Health Safety And Nutrition F/Young Child
Health & Nutrition
ISBN:
9781305144767
Author:
MAROTZ
Publisher:
Cengage

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Cardiopulmonary Anatomy & Physiology
Biology
ISBN:
9781337794909
Author:
Des Jardins, Terry.
Publisher:
Cengage Learning,