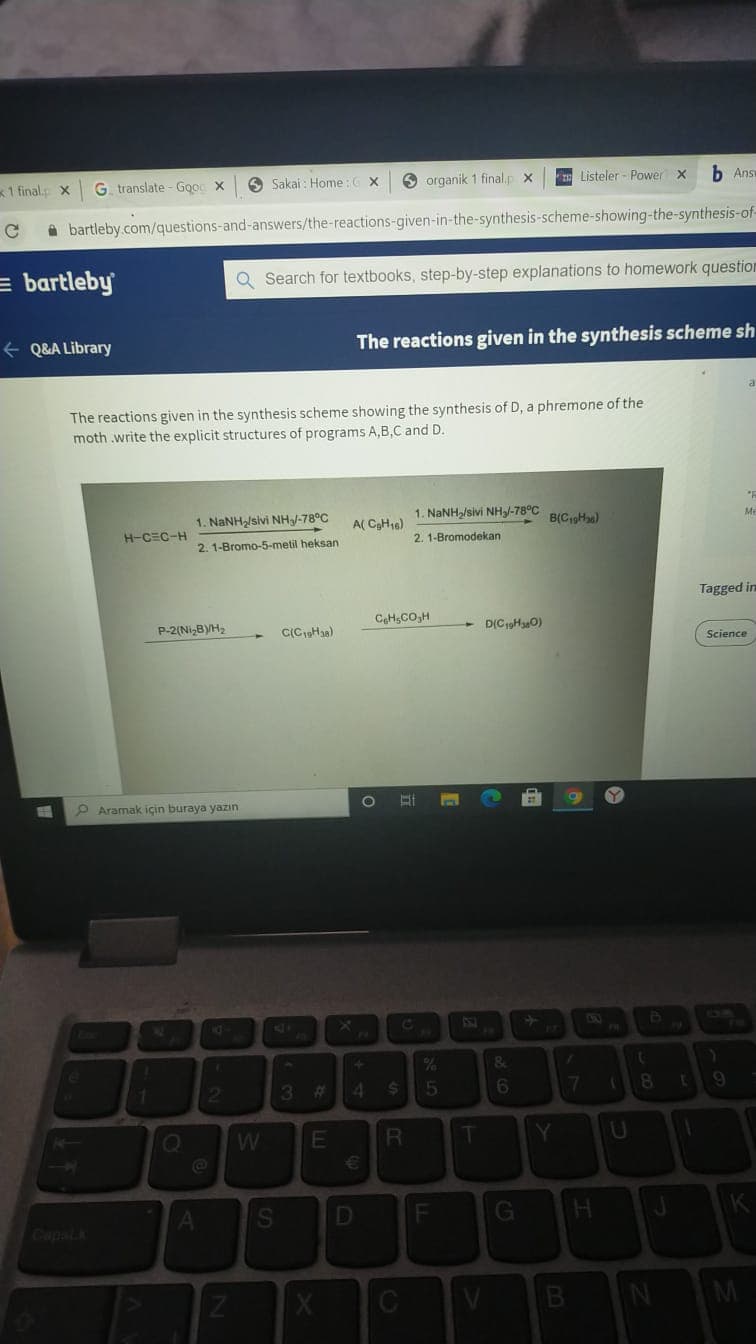
1. NaNH/sivi NHy-78°C 1. NANH/sivi NH-78°C B(CHa) A( CHa) H-CEC-H 2. 1-Bromodekan 2.1-Bromo-5-metil heksan CH,CO,H P-2(Ni,BYH2 C(CHsa) D(CHaO)
1. NaNH/sivi NHy-78°C 1. NANH/sivi NH-78°C B(CHa) A( CHa) H-CEC-H 2. 1-Bromodekan 2.1-Bromo-5-metil heksan CH,CO,H P-2(Ni,BYH2 C(CHsa) D(CHaO)
Chapter5: Equilibrium, Activity And Solving Equations
Section: Chapter Questions
Problem 10P
Related questions
Question
The reactions given in the synthesis scheme showing the synthesis of D, a phremone of the moth.write the explicit structures of programs A,B,C and D.
Transcribed Image Text:1 final. X
G. translate - GQo X
O Sakai : Home:C X
O organik 1 final.p x
Listeler - Power x
b Ans
i bartleby.com/questions-and-answers/the-reactions-given-in-the-synthesis-scheme-showing-the-synthesis-of-
= bartleby
Q Search for textbooks, step-by-step explanations to homework question
+ Q&A Library
The reactions given in the synthesis scheme sh
The reactions given in the synthesis scheme showing the synthesis of D, a phremone of the
moth .write the explicit structures of programs A,B,C and D.
1. NANH/sivi NH/-78°C
1. NaNH/sivi NHJ-78°C
A( CH10)
B(CpHa)
Me
H-CEC-H
2. 1-Bromodekan
2, 1-Bromo-5-metil heksan
Tagged in
CH,CO,H
P-2(Ni,BYH2
D(C19H30)
C(CpHa)
Science
9 Aramak için buraya yazin
27
&
%23
%24
17
Y.
Capalk
FL
+4
DI
E.
93
w/
AL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

