1. Often, an enzyme requires a cofactor or prosthetic group in its active site in order to be an efficient catalyst. Many such cofactors are synthesized by plants and obtained in the diet as vitamins. Find a description in your text of a particular B vitamin. Give its name and describe its biological function in terms of the name(s) and function(s) of the enzyme(s) that require it. 2. Identify the category of enzyme which would catalyze the following reactions. H2 ? + FAD + FADH2 a. + HPO, *H,N-C-coo + H20 CH2 OPO,2 *H,N-C-coo CH2 b. он H H он CH3CH CH3 + H20 CH3 C. H.
Electron Transport Chain
The electron transport chain, also known as the electron transport system, is a group of proteins that transfer electrons through a membrane within mitochondria to create a gradient of protons that drives adenosine triphosphate (ATP)synthesis. The cell uses ATP as an energy source for metabolic processes and cellular functions. ETC involves series of reactions that convert redox energy from NADH (nicotinamide adenine dinucleotide (NAD) + hydrogen (H)) and FADH2(flavin adenine dinucleotide (FAD)) oxidation into proton-motive force(PMF), which is then used to synthesize ATP through conformational changes in the ATP synthase complex, a process known as oxidative phosphorylation.
Metabolism
Picture a campfire. It keeps the body warm on a cold night and provides light. To ensure that the fire keeps burning, fuel needs to be added(pieces of wood in this case). When a small piece is added, the fire burns bright for a bit and then dies down unless more wood is added. But, if too many pieces are placed at a time, the fire escalates and burns for a longer time, without actually burning away all the pieces that have been added. Many of them, especially the larger chunks or damp pieces, remain unburnt.
Cellular Respiration
Cellular respiration is the cellular process involved in the generation of adenosine triphosphate (ATP) molecules from the organic nutritional source obtained from the diet. It is a universal process observed in all types of life forms. The glucose (chemical formula C6H12O6) molecules are the preferred raw material for cell respiration as it possesses a simple structure and is highly efficient in nature.
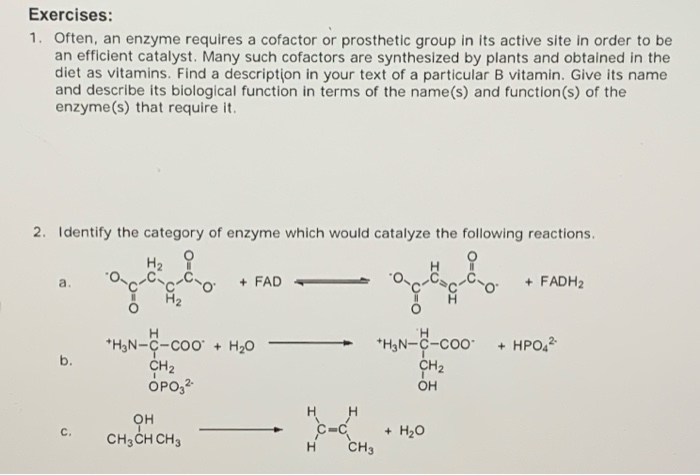
Enzyme act as a catalyst, which is made up of protein (apoprotein) and non-protein (prosthetic ) part. The prosthetic part can be either organic and inorganic. These act as a cofactor, which helps the protein to perform efficient catalysis.
There are 13 vitamins, out of which vitamin B is one of them. It consists of the following types of B vitamins:
- B1 (thiamine)
- B2 (riboflavin)
- B3 (niacin)
- B5 (pantothenic acid)
- B6.
- B7 (biotin)
- B12.
- Folic acid.
1. Thiamine: The enzyme TPP (thiamine pyrophosphate) contains thiamine. It is used to catalyze decarboxylase reaction and degradation as well as the formation of hydroxy ketones.
2. Riboflavin: It is the main constituent of FADH (flavin adenine dinucleotide hydrogen) and FMN (Flavin mononucleotide). It helps in the catalysis of redox reactions.
3. Niacin: It is the main component of nicotinamide adenine (NADH) dinucleotide hydrogen. It acts to serve oxidation-reduction reactions.
4. Biotin: It helps to perform carboxylation reactions. Some enzymes such as pyruvate carboxylase and acetyl CoA carboxylase involve this vitamin.
5. Pantothenic acid: It is the component of CoA (coenzyme A). It works to transfer acyl groups.
6. Folic acid: It performs a one-carbon transfer reaction. It is required by the dihydrofolate reductase enzyme.
7. Cobalamine: It performs intra molecular rearrangement reactions as well as methyl group transfer reaction. methyl malonyl (MCM) CoA mutase, and methionine (MS) synthase.
8. Pyridoxine: It performs transamination, alpha, and beta decarboxylation. The enzyme glutamic acid decarboxylase requires pyridoxine.
Step by step
Solved in 3 steps






