Q: Which is a better nucleophile? a. Br or C in H20 b. Br or CI in DMSO c. CH3O or CH;OH in H20 d. CH30…
A:
Q: Rank the nucleophiles in each group in order of increasing nucleophilicity. а. "ОН, NH, Ha0 b. "OH,…
A: The reagents which have a greater affinity for the positively charged species are known as…
Q: ?nucleophiles اختر واحدة أو أكثر a. CH4 Ob. Br c. H20 d. NH3 e. H2C=CH2 (-)f. OH (+)g. Br h. BH3
A: The electron rich species are termed as nucleophiles. Nucleophiles can be neutral or negatively…
Q: of (a) vs. (b) vs. H
A:
Q: For each of the following pairs, which compound is expected to react faster with a nucleophile in…
A:
Q: Choose all the nucleophiles OH- H3O+ NH4+ CH3OH (CH3)3CLi CH3CH2CH3 NaNH2…
A: Nucleophile :-A chemical species which have free pair of electronsor can denote pair of electrons…
Q: 46. The Strength of Nucleophile: Which one is the strongest nucleophile? (A) OH (B) -CH3 -CH3 (D)…
A: As we go to down the group, size increases, basicity decreases and Nucleophilicity increases. C…
Q: Which compound would undergo carbocation rearrangement during an Syl reaction? Br Br A B Br C H Br D
A: Which compound would undergo carbocation rearrangement during an SN1 reaction? = B would undergo…
Q: Rank these nucleophiles from best to worst Na-I I Best (II)>(1)>(III) worst Best (1)>(III)>(II)…
A: Nucleophile is species which have tendency to donate electron pair. It is electron rich species
Q: Predict the Majar products of the reaction 1.Oraw MechanisM ( Label each arrow with its Dattern) Brz…
A: Electrophilic addition :- Addition of bromine to alkene is a type of electrophilic addition…
Q: Identify the stronger nucleophile in the following pair ? CH3CO2−, CH3CH2O−
A: A nucleophile has a negative charge on it which means it consist of a lone pair of electron for…
Q: D. Is the nucleophile strong or weak? E. Is the solvent polar protic or polar aprotic? F. What type…
A: Note: polar protic solvent and weak nucleophile and secondary alkyl halide all favors a sn1…
Q: Question is attached
A: An elimination reaction is a type of chemical reaction where several atoms either in pairs or groups…
Q: F CH2CH3 B-F H3CH2C-o F
A:
Q: Determine the HOMO in the nucleophile and LUMO in the electrophile in the reaction below. NH, Me…
A:
Q: Identify the stronger nucleophile in the following pair ? NH3, −NH2
A: Nucleophilicity is defined as the tendency to give the electrons. Species with negative charge will…
Q: Which of the following is the strongest nucleophile? O H2S O NH3 O PH3 O CH3OH O H2O
A: The compounds given are H2S, NH3, PH3, CH3OH and H2O.
Q: Draw the substitution product that results when CH,CH2CH;CH,Br reacts with each nucleophile. d.…
A: Since you have asked question with multiple part we will only solve 1st three parts. If you want…
Q: Which nucleophile in each pair which would give the faster SN2 rate? (a) (CH3)2S or (CH3)2Se (b)…
A: SN2 is the Nucleophilic Substitution Bimolecular reaction. The nucleophile that attacks the…
Q: When a single compound contains both a nucleophile and a leavinggroup, an intramolecular reaction…
A: When nucleophile substitutes a leaving group, such reactions are called nucleophilic substitution…
Q: 3. Circle the nucleophile in each pair that undergoes SN2 reaction most rapidly with Ph-CH2-Cl in…
A:
Q: i) (CH3)3N: (CH3)2ö: W ii)
A:
Q: Select the strongest nucleophile from the list below. OH O CF O HS-
A: The species which have tendency to donate electron pair and are electron rich in nature is known as…
Q: Please answer this question
A: Nucleophiles are the molecules or substances that have tendency to donate electrons or react with…
Q: Which of the following will react most slowly with cyanide nucleophile (NC-) in an Sy2 reaction?…
A:
Q: Which of the following is the strongest nucleophile? OH- H20 H2S HS-
A: Answer :- HS- -------------------------------------
Q: Rank these nucleophiles from best to worst Na- I -I Best (II)>(1)>(III) worst Best (1)>(III)>(II)…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: Which is a better nucleophile? a. Br− or Cl− in H2O b. Br− or Cl− in DMSO c. CH3O− or CH3OH in H2O…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: Rank the nucleophiles from weakest to strongest. (1 = weakest, 3 = strongest) HOH
A: Nucleophiles are capable of donating electron pair for forming a bond. The measure of strength of…
Q: 5. For the following pairs of species, which is the strongest nucleophile in acetone? Explain.
A: Nucleophilicity is the tendency of the nucleophile to donate electron to an electron deficient…
Q: Fill in the blanks: Good nucleophiles are usually strong mechanism. nucleophiles and a alkyl halide…
A: A nucleophile is defined as a chemical substance that has lone pair of electron or negative charge…
Q: Consider the following groups of compounds. In each series circke the weaker (poorer) nucleophile.…
A: The nucleophilicity of a substrate depends upon many factors such as basicity, nature of…
Q: For each of the following pairs of species, which is the stronger nucleophile in ethanol? Explain.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Which of the following is the STRONGEST nucleophile? -H- -OH I II II IV а. I b. II с. Ш d. IV
A: a) The compound I is the weakest nucleophile because there is no heteroatom as well as it is…
Q: Question attached
A: A stronger base will be a stronger nucleophile.
Q: Which of the following is the strongest nucleophile ? О а. СНз O b. NH2 С. O C. HO O d. F O e. NH3
A: Nuclephile are species which are able to donate lone pair of electron
Q: Select which molecule is the better nucleophile in the following pair: a). Br− or Cl− in H2O b).…
A:
Q: From each pair, select the stronger nucleophile. Q.) CH3COO- or OH-
A: The species, that make a covalent bond by donates a pair of electrons is known as nucleophile.…
Q: Which of the following is a stronger nucleophile in a polar, protic solvent? O HS
A: The reagent has a negative charge or lone pair of electrons acts as a nucleophile. As the…
Q: 9. Rank the following nucleophiles, in a solution of DMF, in order of weakest to strongest. НО CI…
A: DMF is a solvent known as dimethylformamide. It is a polar aprotic solvent. It doesn’t form hydrogen…
Q: 5. What halide is the strongest nucleophile in a polar aprotic solvent? Why? a. F-, Br-, Cl-, I- b.…
A: The polar aprotic solvents do not contain hydrogen atom, which can take part in hydrogen bonding.…
Q: Which of the following is the strongest nucleophile? CH3S OH- H20 CH3O¯ NH3
A: Nucleophiles A Nucleophile is a chemical species that donates an electron pair to form a…
Q: For the substitution (SN2) reaction below circle the nucleophile and draw a box around the…
A:
Q: Rank the nucleophiles in each group in order of increasing nucleophilicity. a.−OH, −NH2, H2O b.−OH,…
A: a. Negatively charged nucleophiles are stronger nucleophiles than neutral ones. Hence water is the…
Q: In the E1 reaction, how is the rate affected when the concentration of attacking nucleophile is…
A: The rate of a reaction helps in the determination of the effect of the change in the concentration…
Q: ProVide the major prochuct for each following reactuns H more Than one 5 formed, Circk the major…
A: 1) first allylic cation is formed which is electrophile and it attack on Benzene .Ring and product…
Q: Drag the labels to identify the appropriate reagents for each reaction below. Each label is…
A: Double bond between two carbon atoms contain a 'pi' bond. The bonded carbons have sp2 hybridization…
Q: In the reaction CH4 + C12 --> CH3CI + HCI, which is the nucleophile? A) CH4 B) C12 C) HCI D CH3CI E…
A: Given- CH4+Cl2-->> CH3Cl + HCl
Q: *NH OCH CI 7. Which would be the least reactive to nucleophiles? 8. Which will react with SOCI2 to…
A: A question based on Nucleophilic substitution reaction, which is to be accomplished.
Q: Draw the TWO most abundant products for each reaction. Of the two products, circle the one product…
A: Given: Some of the Reactions are given To find: Products formed and circle the major product formed…
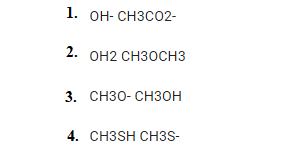
Which pair between 1, 2, 3, and 4 has a nucleophile stronger on the right than on the left?
Step by step
Solved in 2 steps with 2 images

- What is qrxn? -3661 J -146.4 J +146.4 J +3661 JUse the information provided to determine  triangle H degree rxn for the following reaction 3Fe2O3(s) +CO(g) —> 2Fe3O4(s) + CO2(g) triangle H degree rxn? Triangle H degree F (kJ/mol) Fe2O3(s) -824 Fe3O4 (s) -1118 CO (g) -111 CO2 (g) -394especially for B TO X, D to Y, and G to H, I => React with (CH3CO)2O , : N (C2H5) 3
- Plz give all answer only with little bit explanationCalculate ΔH°rxn using the following information: SO2I2(g) + 2 H2O(l) → 2 HI(g) + H2SO4(l) ΔH°rxn = ? ΔH°f (kJ/mol) SO2I2(g) -264 H2O(l) -286 HI(g) -12 H2SO4(l) -814For the Organic Chemistry topic of R and S, to make the structure R, does the prioritizing number have to be in order from 1 -> 2 -> 3 clockwise or can it be clockwise from 3 -> 2 -> 1?

