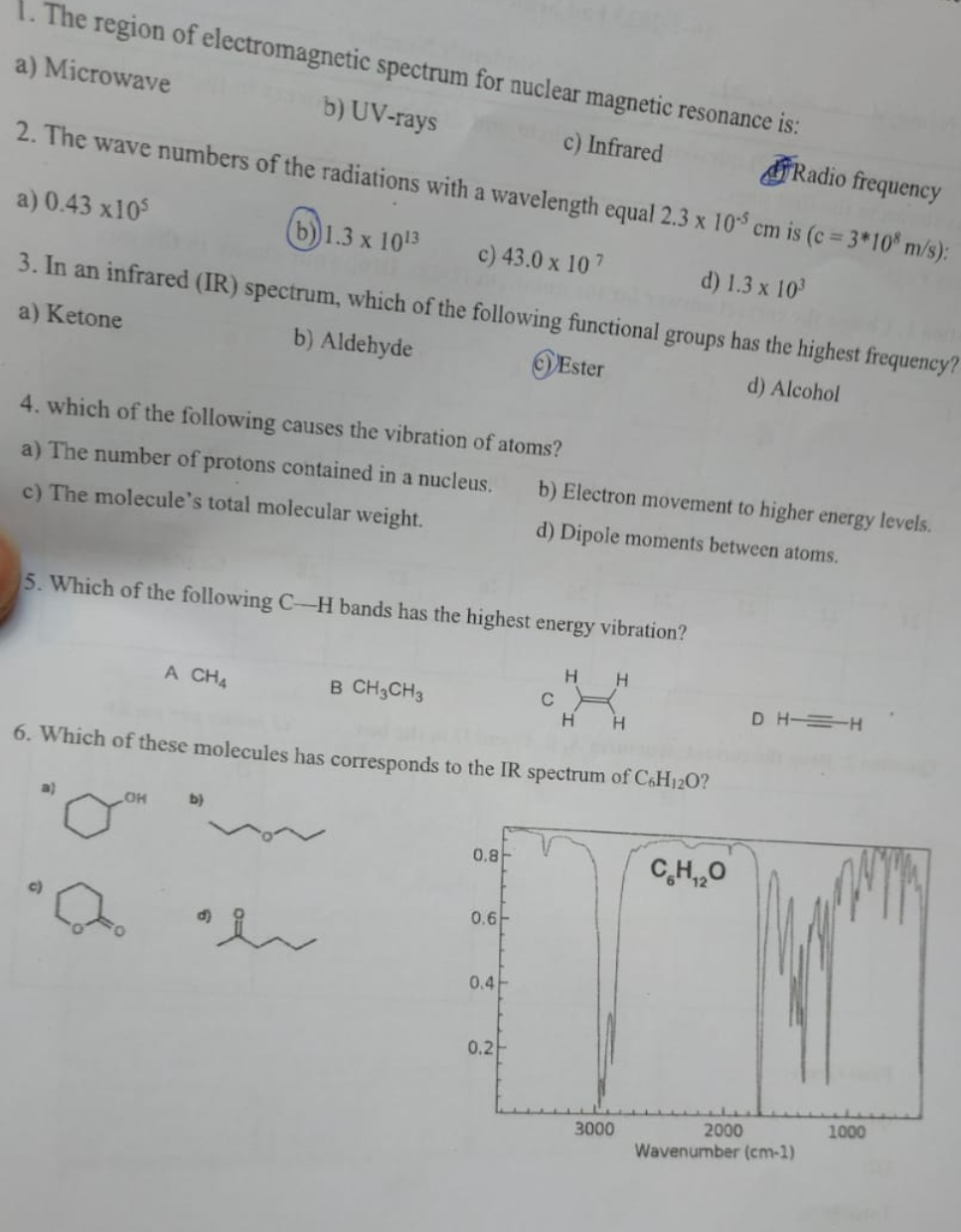
1. The region of electromagnetic spectrum for nuclear magnetic resonance is: a) Microwave b) UV-rays c) Infrared Radio frequency 2. The wave numbers of the radiations with a wavelength equal 2.3 x 105 cm is (c = 3*108 m/s): a) 0.43 x105 b) 1.3 x 1013 c) 43.0 x 107 d) 1.3 x 10³ 3. In an infrared (IR) spectrum, which of the following functional groups has the highest frequency? a) Ketone b) Aldehyde Ester d) Alcohol 4. which of the following causes the vibration of atoms? a) The number of protons contained in a nucleus. c) The molecule's total molecular weight. 5. Which of the following C-H bands has the highest energy vibration? a) A CH4 LOH b) B CH3CH3 a h 6. Which of these molecules has corresponds to the IR spectrum of C6H120? 0.8 0.6 b) Electron movement to higher energy levels. d) Dipole moments between atoms. 0.4 0.2 C H H H H 3000 CH,,O DHH 2000 Wavenumber (cm-1) 1000
Electronic Transitions and Spectroscopy
The term “electronic” connotes electron, and the term “transition” implies transformation. In a molecule, the electrons move from a lower to a higher energy state due to excitation. The two energy states, the ground state and the excited state are the lowest and the highest energy states, respectively. An energy change is observed with this transition, which depicts the various data related to the molecule.
Photoelectron Spectroscopy
Photoelectron spectroscopy (PES) is a part of experimental chemistry. It is a technique used in laboratories that involves projecting intense beams of radiation on a sample element. In response, the element ejects electrons for which the relative energies are measured.
Step by step
Solved in 3 steps









