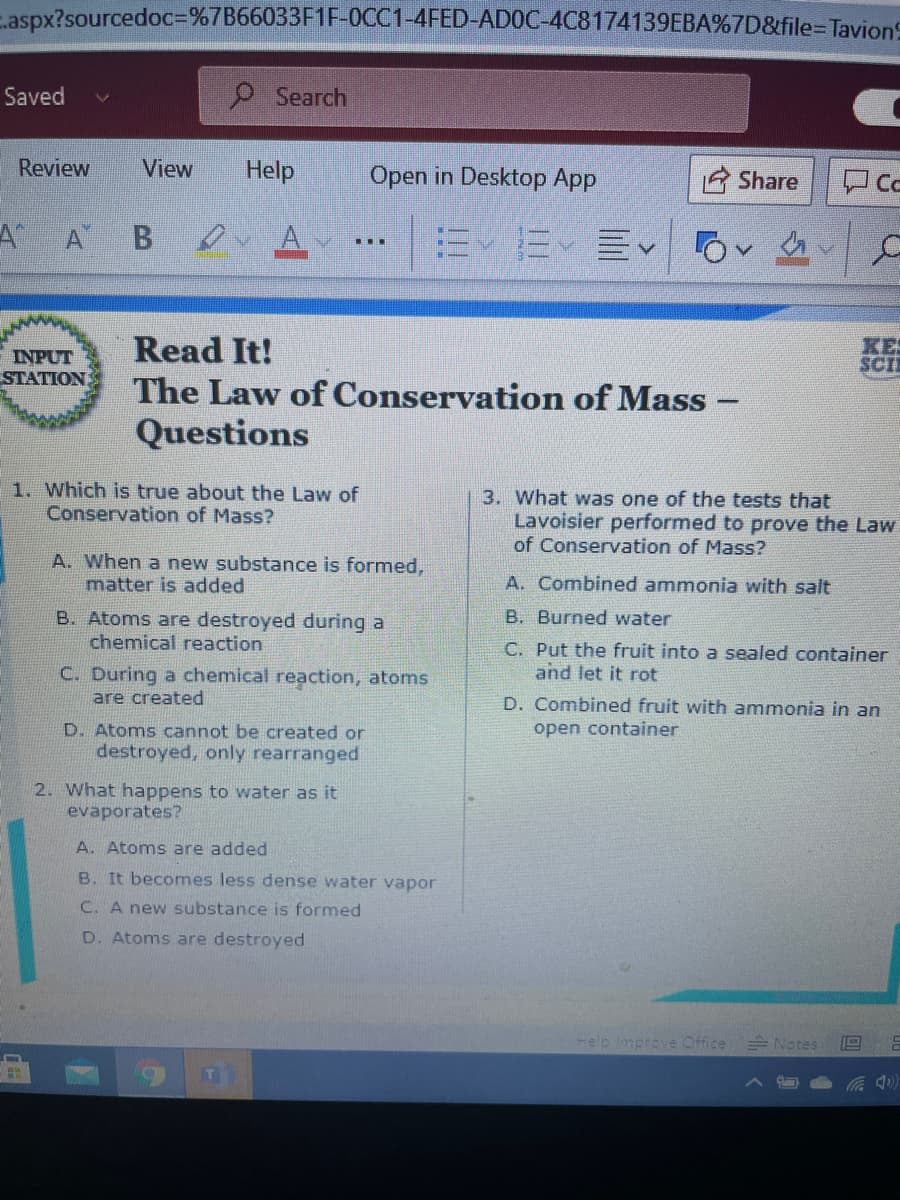
1. Which is true about the Law of Conservation of Mass? 3. What was one of the tests that Lavoisier performed to prove the Law of Conservation of Mass? A. When a new substance is formed, matter is added A. Combined ammonia with salt B. Atoms are destroyed during a chemical reaction B. Burned water C. Put the fruit into a sealed container and let it rot C. During a chemical reaction, atoms are created D. Combined fruit with ammonia in an D. Atoms cannot be created or open container destroyed, only rearranged 2. What happens to water as it evaporates? A. Atoms are added B. It becomes less dense water vapor C. A new substance is formed D. Atoms are destroyed
1. Which is true about the Law of Conservation of Mass? 3. What was one of the tests that Lavoisier performed to prove the Law of Conservation of Mass? A. When a new substance is formed, matter is added A. Combined ammonia with salt B. Atoms are destroyed during a chemical reaction B. Burned water C. Put the fruit into a sealed container and let it rot C. During a chemical reaction, atoms are created D. Combined fruit with ammonia in an D. Atoms cannot be created or open container destroyed, only rearranged 2. What happens to water as it evaporates? A. Atoms are added B. It becomes less dense water vapor C. A new substance is formed D. Atoms are destroyed
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter3: Chemical Reactions
Section3.9: Classifying Reactions In Aqueous Solution
Problem 2.2ACP
Related questions
Question
Help
Transcribed Image Text:- aspx?sourcedoc%3D%7B66033F1F-0CC1-4FED-ADOC-4C8174139EBA%7D&file=DTavion9
Saved
O Search
Review
View
Help
Open in Desktop App
Share
A A B ou A
Read It!
The Law of Conservation of Mass -
INPUT
STATION
KES
SCI
Questions
1. Which is true about the Law of
Conservation of Mass?
3. What was one of the tests that
Lavoisier performed to prove the Law
of Conservation of Mass?
A. When a new substance is formed,
matter is added
A. Combined ammonia with salt
B. Burned water
B. Atoms are destroyed during a
chemical reaction
C. Put the fruit into a sealed container
and let it rot
C. During a chemical reaction, atoms
are created
D. Combined fruit with ammonia in an
D. Atoms cannot be created or
destroyed, only rearranged
open container
2. What happens to water as it
evaporates?
A. Atoms are added
B. It becomes less dense water vapor
C. A new substance is formed
D. Atoms are destroyed
-elp Improve Office
E Notes
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning