1. You received the following absorbance sketch from your lab partner, and he/she asks you for help in identifying the absorbance maximum. What is the estimated Amax in the spectrum for Standard No. 5 Blue #1 solution (see Figure 1)? 2.00- 1.95 1.50- 1.25 1.00t 75 .50 .25 O300 10 400 450 500 550 600 650 700 Wavekngth Cnm) Figure 1: Absorbance vs. Wavelength of Blue #1 2. What color is associated with this Amax? Explain why this color association makes sense for this dye solution. Assume your lab partner described that the solution looked hlue Absorbome o CAu)
1. You received the following absorbance sketch from your lab partner, and he/she asks you for help in identifying the absorbance maximum. What is the estimated Amax in the spectrum for Standard No. 5 Blue #1 solution (see Figure 1)? 2.00- 1.95 1.50- 1.25 1.00t 75 .50 .25 O300 10 400 450 500 550 600 650 700 Wavekngth Cnm) Figure 1: Absorbance vs. Wavelength of Blue #1 2. What color is associated with this Amax? Explain why this color association makes sense for this dye solution. Assume your lab partner described that the solution looked hlue Absorbome o CAu)
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section: Chapter Questions
Problem 77PS
Related questions
Question
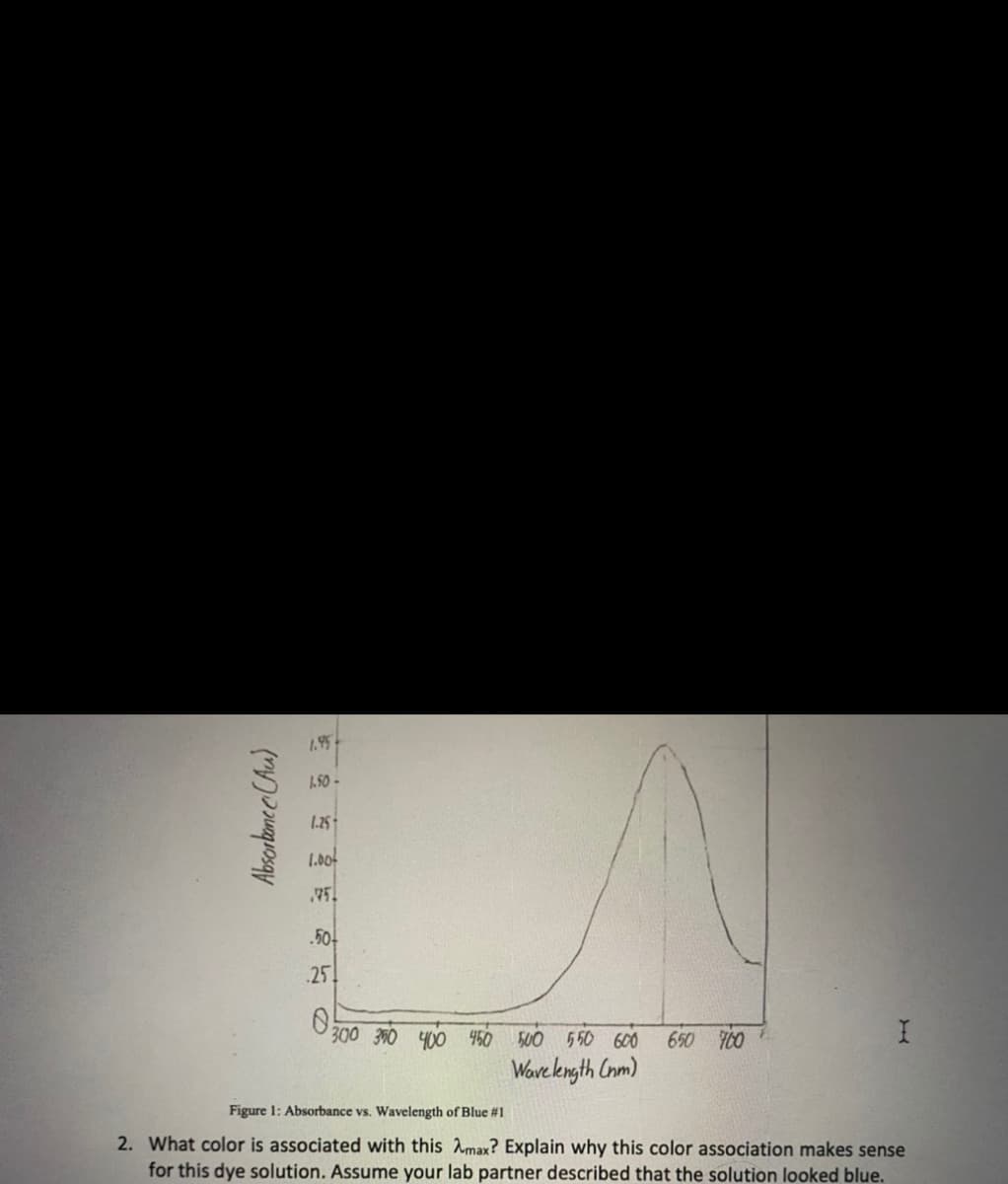
Transcribed Image Text:1.95
1.50 -
1.25-
1.00
75
.50-
.25
300 360 400 460 500 550 600
650 700
Wave kngth Cnm)
Figure 1: Absorbance vs. Wavelength of Blue #1
2. What color is associated with this Amax? Explain why this color association makes sense
for this dye solution. Assume your lab partner described that the solution looked blue.
Absortone e Chu)
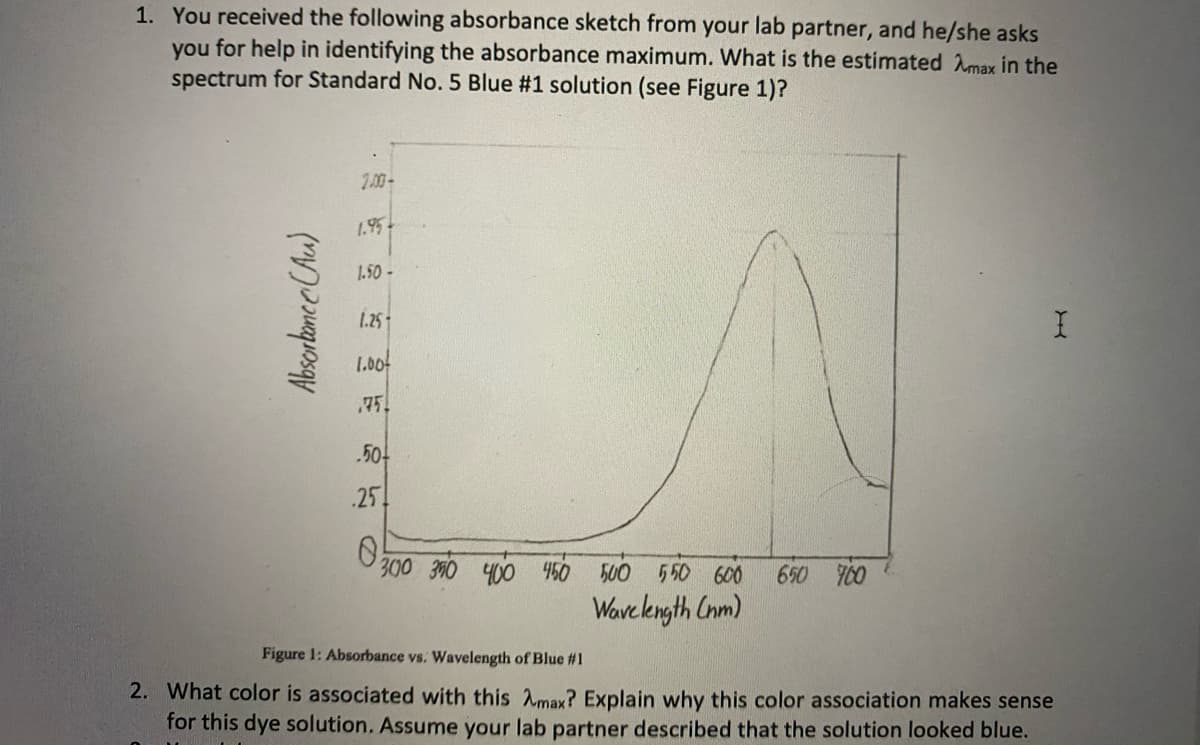
Transcribed Image Text:1. You received the following absorbance sketch from your lab partner, and he/she asks
you for help in identifying the absorbance maximum. What is the estimated Amax in the
spectrum for Standard No. 5 Blue #1 solution (see Figure 1)?
2.00-
1.95
1.50-
1.25
1.00
75
.50
.25
300 300 400
450 500 550 600
650 700
Wavekngth Cnm)
Figure 1: Absorbance vs. Wavelength of Blue #1
2. What color is associated with this Amax? Explain why this color association makes sense
for this dye solution. Assume your lab partner described that the solution looked blue.
Absorbame o CAu)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole