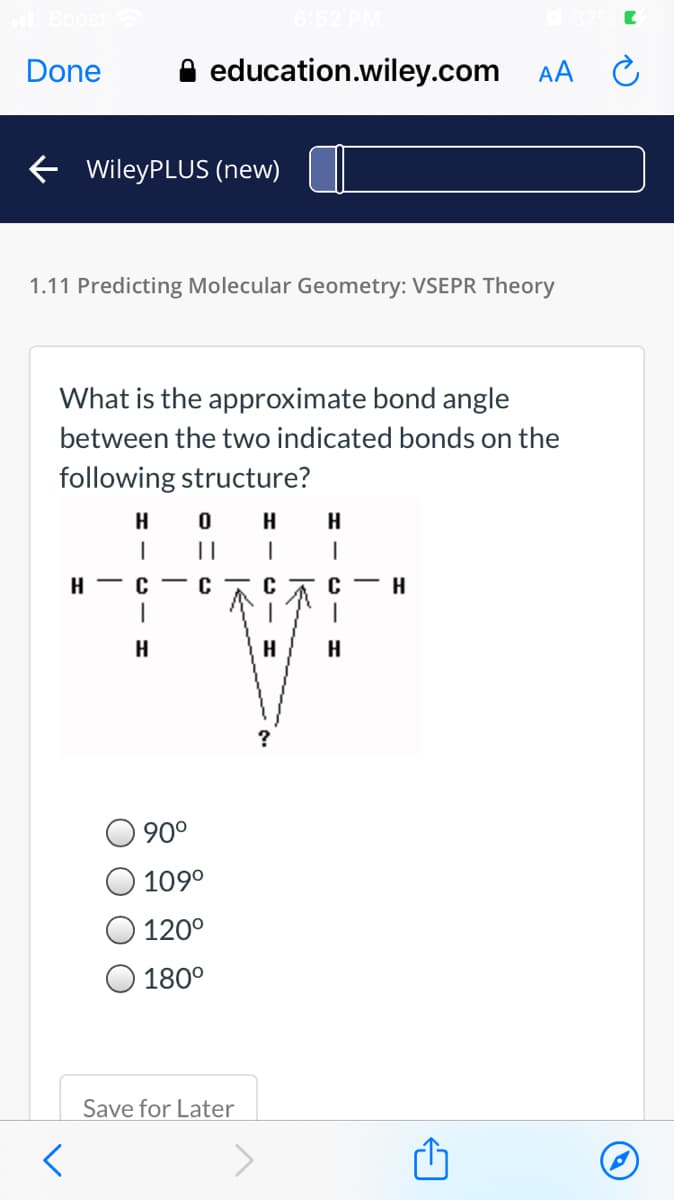
1.11 Predicting Molecular Geometry: VSEPR Theory What is the approximate bond angle between the two indicated bonds on the following structure? H H || н — с C с — н | H H 90° 109° 120° 180°
1.11 Predicting Molecular Geometry: VSEPR Theory What is the approximate bond angle between the two indicated bonds on the following structure? H H || н — с C с — н | H H 90° 109° 120° 180°
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter1: Bond Angles And Shape
Section: Chapter Questions
Problem 14CTQ: A student draws the picture of ammonia (NH3) in the box below, left, and predicts it will be a...
Related questions
Question
Transcribed Image Text:Done
A education.wiley.com
AA C
E WileyPLUS (new)
1.11 Predicting Molecular Geometry: VSEPR Theory
What is the approximate bond angle
between the two indicated bonds on the
following structure?
H
H
H
||
H - C - C
с— н
H
H
H
?
90°
109°
120°
180°
Save for Later
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning