11. Complete the chart. **You should not have the same answer for all the IMF in the last column** Molecular Formula Lewis Structure(1) Polarity of Molecule Justify your Polarity of Bond. Justify your answer. What type of Intermolecular Forces exist in the molecule. Justify your answer. answer. 02 N2 Between the H and the O H2O NH3 Between the H and the N
11. Complete the chart. **You should not have the same answer for all the IMF in the last column** Molecular Formula Lewis Structure(1) Polarity of Molecule Justify your Polarity of Bond. Justify your answer. What type of Intermolecular Forces exist in the molecule. Justify your answer. answer. 02 N2 Between the H and the O H2O NH3 Between the H and the N
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 28E: At 25 C, how high will water rise in a glass capillary tube with an inner diameter of 0.63 mm? Refer...
Related questions
Question
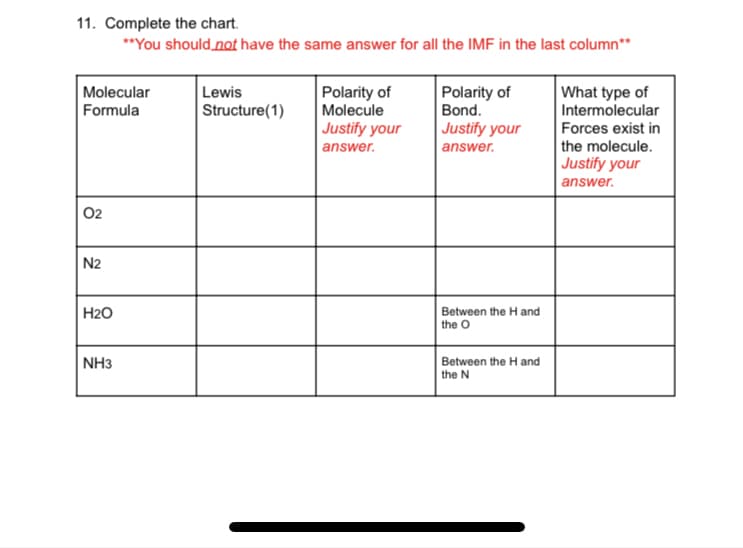
Transcribed Image Text:11. Complete the chart.
**You should not have the same answer for all the IMF in the last column**
Polarity of
Molecular
Formula
Lewis
Structure(1)
Polarity of
Molecule
Justify your
answer.
What type of
Intermolecular
Forces exist in
the molecule.
Justify your
Bond.
Justify your
answer.
answer.
02
N2
H2O
Between the H and
the O
NH3
Between the H and
the N
Expert Solution
Step 1
The Lewis structures were given by Lewis who depicts the bonds and electrons present in the molecule.
For O2, the Lewis structure is
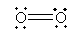
For N2, the Lewis structure is
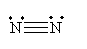
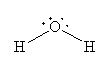
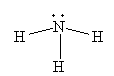
For H2O, the Lewis structure is
For NH3, the Lewis structure is
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning