1113 Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) AH? (kJ/mol) AG (kJ/mol) S° AG (kJ/mol) Şubstance (kJ/mol) U/mol-K) Substance U/mol-K) -569.6 26.8 -205.9 -156.1 MgO(s) Mg(OH)2(s) 108.1 -601.8 CuCla(s) CuO(s) Cuz0(s) - 161.7 -128.3 42.59 -924.7 -833.7 63.24 -170.7 -147.9 92.36 Manganese Mn(g) Mn(s) MnO(s) Fluorine 280.7 238.5 173.6 80.0 61.9 158.7 32.0 F(g) F"(aq) F2lg) HF(g) -362.9 -464.8 -332.6 -278.8 -13.8 -385.2 59.7 -519.6 -541.4 202.7 MnO2(s) 53.14 -268.61 -270.70 173.51 MnO, (ag) --447.2 191.2 AG? AG? (kJ/mol) AH) AH) (kJ/mol) Hydrogen H(g) H*(aq) H*) H2) Mercury Hg(g) Hg() HgCl2(s) Hg2Cl2(s) (kj/mol) (kJ/mol) U/mol-K) Substance 0/mol-K) Substance 217.94 203.26 114.60 60.83 31.76 174.89 52.30 68.11 219.4 77.40 Сан(8) C3H3) C,Ha(8) CH16(8) CH10(1) CHol8) Aluminum 28.32 -84.68 -32.89 229.5 1536.2 1517.0 108.9 -230.1 -184.0 144.5 Al(s) AICly(s) AlzOs(s) -705.6 -630.0 109.3 - 103.85 -23.47 269.9 130.58 -264.9 -210.5 192.5 -1669.8 -1576.S 51.00 -124.73 -15.71 310.0 Iodine Nickel -147.6 --15.0 231.0 106.60 70.16 180.66 429.7 384.5 182.1 I(g) r(aq) Ig) 2(s) HI(g) Ni(g) Ni(s) NIC2(s) NiO(s) Barium 82.9 129.7 269.2 29.9 63.2 -55.19 -51.57 111.3 Ba(s) BaCO(s) BaO(s) 49.0 124.5 172.8 62.25 260.57 -305.3 -259.0 97.65 19.37 -1216.3 -1137.6 112.1 CнHоне) CHОН() CаHоне) CHSOH() CH12O6(s) COg) CO2{8) CH,COОН() -201.2 -161.9 237.6 116.73 -239.7 -211.7 37.99 -553.5 -525.1 70.42 -238.6 --166.23 126.8 25.94 1.30 206.3 Nitrogen N(g) Nzg) NH3(aq) NH3(8) NH, (aq) N2H4(g) NH,CN(6) NH,CI(s) NH,NO3(4) NO(g) NO2(8) N2O(g) NO,(8) NOCI(g) HNO3(aq) HNO,(g) -168.5 282.7 Berylium Be(s). BeO(s) Be(OH)2(s) -235.1 9.44 -277,7 174.76 160.7 Iron 472.7 455.5 153.3 415.5 369.8 180.5 191.50 Felg) Fe(s) Fe2"(aq) Fe"(aq) -608.4 -579.1 13.77 -1273.02 -910.4 212.1 27.15 113.4 -905.8 -817.9 50.21 -110.5 -137.2 197.9 --80.29 -26.50 111.3 -87.86 -84.93 -46.19 - 16.66 192.5 -393.5 -487.0 -394.4 213.6 Bromine -47.69 - 10.54 293.3 -132.5 -79.31 113.4 -392.4 159.8 111.8 82.38 174.9 Br(g) Br"(aq) Br2(g) -341.8 -302.3 117.9 159.4 238.5 FeCh(s) FeCly(s) FeO(s) Fe,O,(s) Fe,O(s) FeS2(s) 95.40 -120.9 -102.8 80.71 Cesium -400 -334 142.3 0.0 30.71 3.14 245.3 76.50 49.53 175.6 Cslg) Cs() -271.9 -255.2 60.75 -314.4 -203.0 94.6 92.07 85.15 152.3 2.09 0.03 -822.16 -1117.1 -740.98 89.96 -365.6 --184.0 151 ABr(g) -36.23 -53.22 198.49 Cs(s) CSCI(s) -1014.2 146.4 90.37 86.71 210.62 --442.8 -414.4 101.2 -160.1 52.92 240.45 Calcium -171.5 33.84 51.84 179.3 145.5 154.8 81.6 103.59 220.0 Ca(g) Ca(s) CaCO,(s, calcite) CaCla(s) CaF2(s) CaO(s) Ca(OH)2(s) CasO,(s) Chlorine Lead 41.4 121.7 105.7 165.2 9.66 98.28 304.3 CI(g) CI"(aq) C2(g) НClag) HC(g) 68.85 Pb(s) РЪВrs(9) РЬСО,) -1207.1 -1128.76 92.88 -167.2 -131.2 56.5 -277.4 -260.7 161 52.6 66.3 264 -795.8 -748.1 104.6 222.96 -206.6 -110.5 146 699.1 -625.5 131.0 -1219.6 -1167.3 68.87 -167.2 -131.2 56.5 --246.9 303.3 -134.3 -73.94 266.4 Pb(NOsh(aq) Pb(NO,)2(s) PbO(s) -421.3 -635.5 -604.17 39.75 -92.30 -95.27 186.69 -451.9 Oxygen Og) Olg) Os(8) OH (aq) -986.2 -898.5 83.4 Chromium -2173 -187.9 68.70 247.5 230.1 161.0 -1434.0 -1321.8 106.7 397.5 352.6 174.2 205.0 Cr(g) Cr(s) CryOs(s) Lithium Carbon 23.6 Li(g) 159.3 126.6 138.8 142.3 163.4 237.6 718.4 672.9 158.0 -1058.1 81.2 -230.0 -157.3 -10.7 Cg) C(s, diamond) C(s, graphite) CCL(8) CC4() CF(g) CH,8) Сн8) -1139.7 29.09. Li(s) Li*(aq) Li"(g) LICI(6) 1.88 2.84 2.43 -241.82 -228.57 188.83 69.91 Н.Os) Н.О() HO28) H2O2(g) -278.5 685.7 Cobalt -273.4 12.2 5.69 -285.83 -136.10 -237.13 -105.48 439 393 179 648.5 133.0 Co(g) Co(s) -106.7 -64.0 309.4 28.4 -408.3 -384.0 59.30 232.9 - 139.3 -68.6 214.4 - 187.8 -120.4 109.6 Copper Cutg) Cu(s) Magnesium Mg(g) Mg(e) MgCl2(s) -679.9 635.1 262.3 148.6 166.3 33.30 Phosphorus P(g) P2(g) -74.8 -50.8 186.3 338.4 298.6 147.1 112.5 32.51 316.4 280.0 163.2 226.77 209.2 200.8 -641.6 -592.1 89.6 144.3 103.7 218.1 1112 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 1114 Ане APPENDIX APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 1114 AG} (kJ/mol) S° AH (kJ/mol) (kJ/mol) AG} ану (kJ/mol) Substance U/mol-K) Substance U/mol-K) 96.11 A8CI(s) Ag20(s) AGNO,(s) -127.0 -109.70 Phosphorus (cont.) Plg) Pa(s, red) Pa(s, white) PCly(g) PC() PFs(g) PH3(g) P,O6(s) P,O10(s) POCI(g) -31.05 -11.20 121.3 58.9 24.4 280 -124.4 -33.41 140.9 -17.46 -12.03 22.85 41.08 Sodium 77.3 153.7 -288.07 -319.6 -269.6 311.7 107.7 Na(g) Na(s) Na*(aq) Na*(g) NaBr(aq) NaBr(s) NazCOs(s) NaCl(aq) NaCl(g) NaCl(s) NAHCO,(s) NANO3(ag) NANO3(6) NaOH(aq) NaOH(s) -272.4 217 51.45 -1594.4 -1520.7 300.8 -240.1 -261.9 59.0 5.4 13.4 210.2 609.3 574.3 148.0 -1640.1 -360.6 -364.7 141.00 -2940.1 -2675.2 228.9 -361.4 -349.3 86.82 136.0 1130.9 -407.1 -542.2 -502.5 325 -1047.7 -597.0 -520.9 222 -393.0 115.5 POCI() НРОдаg) -1288.3 -1142.6 158.2 -181.4 -201.3 229.8 -410.9 -384.0 72.33 Potassium -947.7 -851.8 102.1 89.99 61.17 160.2 K(g) K(s) KCI(s) KCIO,(s) KCIO,(ag) KĄCO(s) KNO,(s) KO(s) KO,(s) KO2(s) КОН(6) коНод) -446.2 -372.4 207 64.67 -467.9 --367.0 116.5 435.9 --408.3 82.7 49.8 -469.6 -425.6 -419.2 -391.2 -289.9 143.0 -379.5 64.46 --349.5 --284.9 265.7 -1150.18 -1064.58 155.44 Strontium -592.0 561.9 54.9 -492.70 -363.2 -393.13 132.9 SrO(s) --322.1 94.14 Sr(g) 164.4 110.0 164.6 -284.5 -240.6 122.5 Sulfur S(s, rhombic) S(g) SO(3) SO,(8) So,*"(aq) SOCI;() HSg) HSO,(ag) HSO,() --495.8 -429.8 113.0 31.88 -424.7 -378.9 78.91 162.3 49.7 430.9 -482.4 -440.5 91.6 -296.9 -300.4 248.5 --395.2 -370.4. 256.2 Rubidium -909.3 -744.5 20.1 170.0 76,78 85.8 55.8 Rb(g) Rb(s) RЬCI(:) -245.6 -20.17 -909.3 -33.01 205.6 -430.5 -412.0 92 -744.5 20.1 RBCIO,(s) -392.4 -292.0 152 -814.0 -689.9 156.1 Scandium Titanium 377.8 336.1 174.7 Scg) Sc(s) 468 422 180.3 Ti(g) Ti(s) 34.6 30.76 354.9 221.9 -763.2 -726.8 ПC48) TICL4() TIO2(8) Selenium H,Selg) 29.7 15.9 219.0 -804.2 -728.1 -944.7 -889.4 50.29 Silicon 368.2 323.9 167.8 Vanadium Si(g) Si(s) SIC(S) SICL() SIO2(s, quartz) 18.7 514.2 453.1 182.2 -73.22 --70.85 16.61 V(s) 28.9 239.3 41.84 -640.1 -572.8 Zinc -910.9 -856.5 130.7 95.2. 160.9 Zng Zn(s) ZnClą(s) ZnO(s) 41.63 Silver --415.1 --369.4 111.5 42.55 Ag(s) Ag (aq) -348.0 -318.2 43.9 105.90 77.11 73.93
1113 Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) AH? (kJ/mol) AG (kJ/mol) S° AG (kJ/mol) Şubstance (kJ/mol) U/mol-K) Substance U/mol-K) -569.6 26.8 -205.9 -156.1 MgO(s) Mg(OH)2(s) 108.1 -601.8 CuCla(s) CuO(s) Cuz0(s) - 161.7 -128.3 42.59 -924.7 -833.7 63.24 -170.7 -147.9 92.36 Manganese Mn(g) Mn(s) MnO(s) Fluorine 280.7 238.5 173.6 80.0 61.9 158.7 32.0 F(g) F"(aq) F2lg) HF(g) -362.9 -464.8 -332.6 -278.8 -13.8 -385.2 59.7 -519.6 -541.4 202.7 MnO2(s) 53.14 -268.61 -270.70 173.51 MnO, (ag) --447.2 191.2 AG? AG? (kJ/mol) AH) AH) (kJ/mol) Hydrogen H(g) H*(aq) H*) H2) Mercury Hg(g) Hg() HgCl2(s) Hg2Cl2(s) (kj/mol) (kJ/mol) U/mol-K) Substance 0/mol-K) Substance 217.94 203.26 114.60 60.83 31.76 174.89 52.30 68.11 219.4 77.40 Сан(8) C3H3) C,Ha(8) CH16(8) CH10(1) CHol8) Aluminum 28.32 -84.68 -32.89 229.5 1536.2 1517.0 108.9 -230.1 -184.0 144.5 Al(s) AICly(s) AlzOs(s) -705.6 -630.0 109.3 - 103.85 -23.47 269.9 130.58 -264.9 -210.5 192.5 -1669.8 -1576.S 51.00 -124.73 -15.71 310.0 Iodine Nickel -147.6 --15.0 231.0 106.60 70.16 180.66 429.7 384.5 182.1 I(g) r(aq) Ig) 2(s) HI(g) Ni(g) Ni(s) NIC2(s) NiO(s) Barium 82.9 129.7 269.2 29.9 63.2 -55.19 -51.57 111.3 Ba(s) BaCO(s) BaO(s) 49.0 124.5 172.8 62.25 260.57 -305.3 -259.0 97.65 19.37 -1216.3 -1137.6 112.1 CнHоне) CHОН() CаHоне) CHSOH() CH12O6(s) COg) CO2{8) CH,COОН() -201.2 -161.9 237.6 116.73 -239.7 -211.7 37.99 -553.5 -525.1 70.42 -238.6 --166.23 126.8 25.94 1.30 206.3 Nitrogen N(g) Nzg) NH3(aq) NH3(8) NH, (aq) N2H4(g) NH,CN(6) NH,CI(s) NH,NO3(4) NO(g) NO2(8) N2O(g) NO,(8) NOCI(g) HNO3(aq) HNO,(g) -168.5 282.7 Berylium Be(s). BeO(s) Be(OH)2(s) -235.1 9.44 -277,7 174.76 160.7 Iron 472.7 455.5 153.3 415.5 369.8 180.5 191.50 Felg) Fe(s) Fe2"(aq) Fe"(aq) -608.4 -579.1 13.77 -1273.02 -910.4 212.1 27.15 113.4 -905.8 -817.9 50.21 -110.5 -137.2 197.9 --80.29 -26.50 111.3 -87.86 -84.93 -46.19 - 16.66 192.5 -393.5 -487.0 -394.4 213.6 Bromine -47.69 - 10.54 293.3 -132.5 -79.31 113.4 -392.4 159.8 111.8 82.38 174.9 Br(g) Br"(aq) Br2(g) -341.8 -302.3 117.9 159.4 238.5 FeCh(s) FeCly(s) FeO(s) Fe,O,(s) Fe,O(s) FeS2(s) 95.40 -120.9 -102.8 80.71 Cesium -400 -334 142.3 0.0 30.71 3.14 245.3 76.50 49.53 175.6 Cslg) Cs() -271.9 -255.2 60.75 -314.4 -203.0 94.6 92.07 85.15 152.3 2.09 0.03 -822.16 -1117.1 -740.98 89.96 -365.6 --184.0 151 ABr(g) -36.23 -53.22 198.49 Cs(s) CSCI(s) -1014.2 146.4 90.37 86.71 210.62 --442.8 -414.4 101.2 -160.1 52.92 240.45 Calcium -171.5 33.84 51.84 179.3 145.5 154.8 81.6 103.59 220.0 Ca(g) Ca(s) CaCO,(s, calcite) CaCla(s) CaF2(s) CaO(s) Ca(OH)2(s) CasO,(s) Chlorine Lead 41.4 121.7 105.7 165.2 9.66 98.28 304.3 CI(g) CI"(aq) C2(g) НClag) HC(g) 68.85 Pb(s) РЪВrs(9) РЬСО,) -1207.1 -1128.76 92.88 -167.2 -131.2 56.5 -277.4 -260.7 161 52.6 66.3 264 -795.8 -748.1 104.6 222.96 -206.6 -110.5 146 699.1 -625.5 131.0 -1219.6 -1167.3 68.87 -167.2 -131.2 56.5 --246.9 303.3 -134.3 -73.94 266.4 Pb(NOsh(aq) Pb(NO,)2(s) PbO(s) -421.3 -635.5 -604.17 39.75 -92.30 -95.27 186.69 -451.9 Oxygen Og) Olg) Os(8) OH (aq) -986.2 -898.5 83.4 Chromium -2173 -187.9 68.70 247.5 230.1 161.0 -1434.0 -1321.8 106.7 397.5 352.6 174.2 205.0 Cr(g) Cr(s) CryOs(s) Lithium Carbon 23.6 Li(g) 159.3 126.6 138.8 142.3 163.4 237.6 718.4 672.9 158.0 -1058.1 81.2 -230.0 -157.3 -10.7 Cg) C(s, diamond) C(s, graphite) CCL(8) CC4() CF(g) CH,8) Сн8) -1139.7 29.09. Li(s) Li*(aq) Li"(g) LICI(6) 1.88 2.84 2.43 -241.82 -228.57 188.83 69.91 Н.Os) Н.О() HO28) H2O2(g) -278.5 685.7 Cobalt -273.4 12.2 5.69 -285.83 -136.10 -237.13 -105.48 439 393 179 648.5 133.0 Co(g) Co(s) -106.7 -64.0 309.4 28.4 -408.3 -384.0 59.30 232.9 - 139.3 -68.6 214.4 - 187.8 -120.4 109.6 Copper Cutg) Cu(s) Magnesium Mg(g) Mg(e) MgCl2(s) -679.9 635.1 262.3 148.6 166.3 33.30 Phosphorus P(g) P2(g) -74.8 -50.8 186.3 338.4 298.6 147.1 112.5 32.51 316.4 280.0 163.2 226.77 209.2 200.8 -641.6 -592.1 89.6 144.3 103.7 218.1 1112 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 1114 Ане APPENDIX APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 1114 AG} (kJ/mol) S° AH (kJ/mol) (kJ/mol) AG} ану (kJ/mol) Substance U/mol-K) Substance U/mol-K) 96.11 A8CI(s) Ag20(s) AGNO,(s) -127.0 -109.70 Phosphorus (cont.) Plg) Pa(s, red) Pa(s, white) PCly(g) PC() PFs(g) PH3(g) P,O6(s) P,O10(s) POCI(g) -31.05 -11.20 121.3 58.9 24.4 280 -124.4 -33.41 140.9 -17.46 -12.03 22.85 41.08 Sodium 77.3 153.7 -288.07 -319.6 -269.6 311.7 107.7 Na(g) Na(s) Na*(aq) Na*(g) NaBr(aq) NaBr(s) NazCOs(s) NaCl(aq) NaCl(g) NaCl(s) NAHCO,(s) NANO3(ag) NANO3(6) NaOH(aq) NaOH(s) -272.4 217 51.45 -1594.4 -1520.7 300.8 -240.1 -261.9 59.0 5.4 13.4 210.2 609.3 574.3 148.0 -1640.1 -360.6 -364.7 141.00 -2940.1 -2675.2 228.9 -361.4 -349.3 86.82 136.0 1130.9 -407.1 -542.2 -502.5 325 -1047.7 -597.0 -520.9 222 -393.0 115.5 POCI() НРОдаg) -1288.3 -1142.6 158.2 -181.4 -201.3 229.8 -410.9 -384.0 72.33 Potassium -947.7 -851.8 102.1 89.99 61.17 160.2 K(g) K(s) KCI(s) KCIO,(s) KCIO,(ag) KĄCO(s) KNO,(s) KO(s) KO,(s) KO2(s) КОН(6) коНод) -446.2 -372.4 207 64.67 -467.9 --367.0 116.5 435.9 --408.3 82.7 49.8 -469.6 -425.6 -419.2 -391.2 -289.9 143.0 -379.5 64.46 --349.5 --284.9 265.7 -1150.18 -1064.58 155.44 Strontium -592.0 561.9 54.9 -492.70 -363.2 -393.13 132.9 SrO(s) --322.1 94.14 Sr(g) 164.4 110.0 164.6 -284.5 -240.6 122.5 Sulfur S(s, rhombic) S(g) SO(3) SO,(8) So,*"(aq) SOCI;() HSg) HSO,(ag) HSO,() --495.8 -429.8 113.0 31.88 -424.7 -378.9 78.91 162.3 49.7 430.9 -482.4 -440.5 91.6 -296.9 -300.4 248.5 --395.2 -370.4. 256.2 Rubidium -909.3 -744.5 20.1 170.0 76,78 85.8 55.8 Rb(g) Rb(s) RЬCI(:) -245.6 -20.17 -909.3 -33.01 205.6 -430.5 -412.0 92 -744.5 20.1 RBCIO,(s) -392.4 -292.0 152 -814.0 -689.9 156.1 Scandium Titanium 377.8 336.1 174.7 Scg) Sc(s) 468 422 180.3 Ti(g) Ti(s) 34.6 30.76 354.9 221.9 -763.2 -726.8 ПC48) TICL4() TIO2(8) Selenium H,Selg) 29.7 15.9 219.0 -804.2 -728.1 -944.7 -889.4 50.29 Silicon 368.2 323.9 167.8 Vanadium Si(g) Si(s) SIC(S) SICL() SIO2(s, quartz) 18.7 514.2 453.1 182.2 -73.22 --70.85 16.61 V(s) 28.9 239.3 41.84 -640.1 -572.8 Zinc -910.9 -856.5 130.7 95.2. 160.9 Zng Zn(s) ZnClą(s) ZnO(s) 41.63 Silver --415.1 --369.4 111.5 42.55 Ag(s) Ag (aq) -348.0 -318.2 43.9 105.90 77.11 73.93
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter10: Entropy And The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 10.78PAE: Why is it usually easier to use G to determine the spontaneity of a process rather than Su ?
Related questions
Question
Calculate the enthalpy change for the complete combustion of 25.0 g ethanol
C2H5OH(l) + 3O2(g) --> 2CO2(g) + 3H2O(l) deltaHrxn = -555kJ/mol
Using appendix C values, calculate the delta H
for the following reactions:
4NH3(g) + O2(g) -->2N2H4(g) + 2H2O(l)
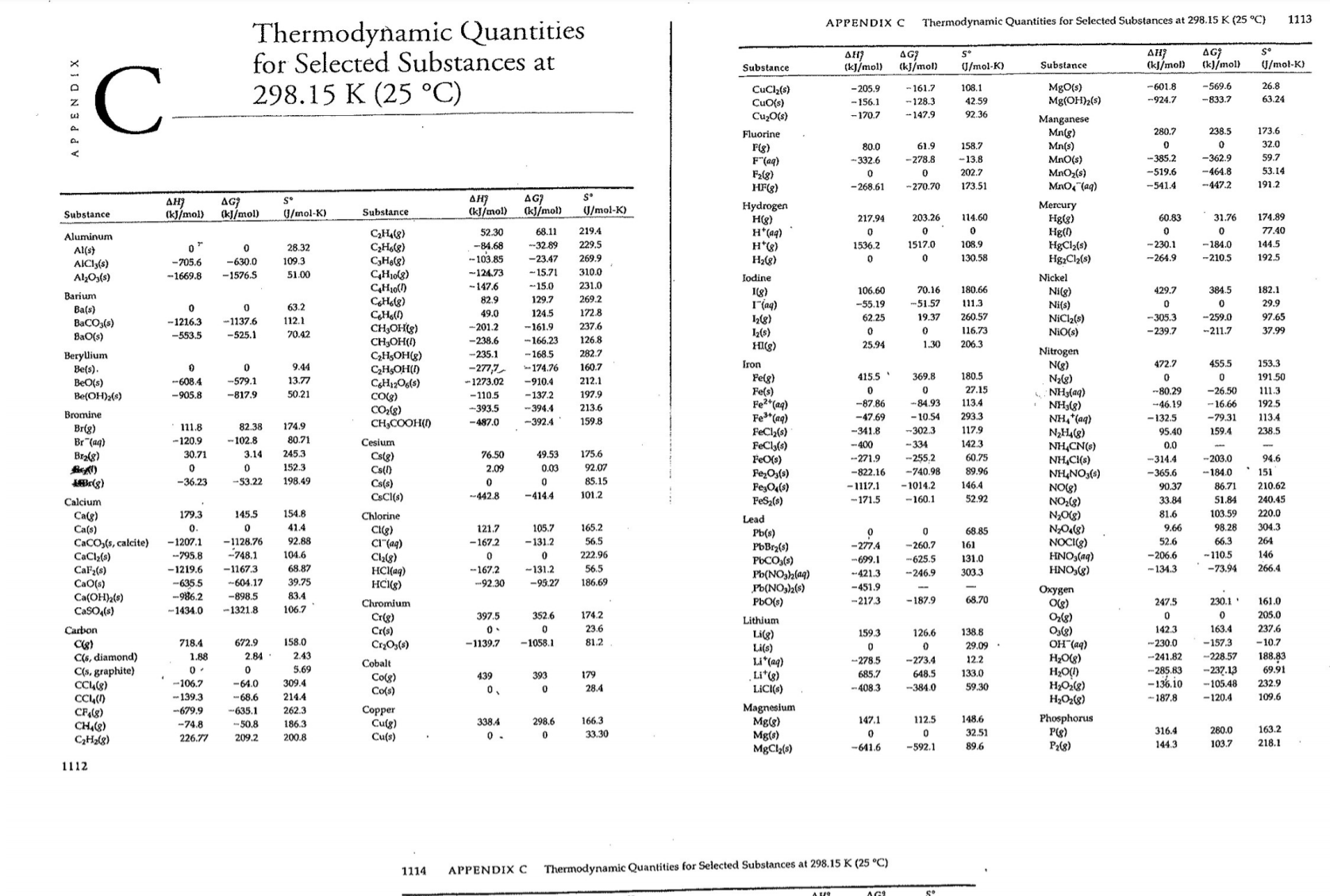
Transcribed Image Text:1113
Thermodynamic Quantities
for Selected Substances at
298.15 K (25 °C)
APPENDIX C
Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C)
AH?
(kJ/mol)
AG
(kJ/mol)
S°
AG
(kJ/mol)
Şubstance
(kJ/mol)
U/mol-K)
Substance
U/mol-K)
-569.6
26.8
-205.9
-156.1
MgO(s)
Mg(OH)2(s)
108.1
-601.8
CuCla(s)
CuO(s)
Cuz0(s)
- 161.7
-128.3
42.59
-924.7
-833.7
63.24
-170.7
-147.9
92.36
Manganese
Mn(g)
Mn(s)
MnO(s)
Fluorine
280.7
238.5
173.6
80.0
61.9
158.7
32.0
F(g)
F"(aq)
F2lg)
HF(g)
-362.9
-464.8
-332.6
-278.8
-13.8
-385.2
59.7
-519.6
-541.4
202.7
MnO2(s)
53.14
-268.61
-270.70
173.51
MnO, (ag)
--447.2
191.2
AG?
AG?
(kJ/mol)
AH)
AH)
(kJ/mol)
Hydrogen
H(g)
H*(aq)
H*)
H2)
Mercury
Hg(g)
Hg()
HgCl2(s)
Hg2Cl2(s)
(kj/mol)
(kJ/mol)
U/mol-K)
Substance
0/mol-K)
Substance
217.94
203.26
114.60
60.83
31.76
174.89
52.30
68.11
219.4
77.40
Сан(8)
C3H3)
C,Ha(8)
CH16(8)
CH10(1)
CHol8)
Aluminum
28.32
-84.68
-32.89
229.5
1536.2
1517.0
108.9
-230.1
-184.0
144.5
Al(s)
AICly(s)
AlzOs(s)
-705.6
-630.0
109.3
- 103.85
-23.47
269.9
130.58
-264.9
-210.5
192.5
-1669.8
-1576.S
51.00
-124.73
-15.71
310.0
Iodine
Nickel
-147.6
--15.0
231.0
106.60
70.16
180.66
429.7
384.5
182.1
I(g)
r(aq)
Ig)
2(s)
HI(g)
Ni(g)
Ni(s)
NIC2(s)
NiO(s)
Barium
82.9
129.7
269.2
29.9
63.2
-55.19
-51.57
111.3
Ba(s)
BaCO(s)
BaO(s)
49.0
124.5
172.8
62.25
260.57
-305.3
-259.0
97.65
19.37
-1216.3
-1137.6
112.1
CнHоне)
CHОН()
CаHоне)
CHSOH()
CH12O6(s)
COg)
CO2{8)
CH,COОН()
-201.2
-161.9
237.6
116.73
-239.7
-211.7
37.99
-553.5
-525.1
70.42
-238.6
--166.23
126.8
25.94
1.30
206.3
Nitrogen
N(g)
Nzg)
NH3(aq)
NH3(8)
NH, (aq)
N2H4(g)
NH,CN(6)
NH,CI(s)
NH,NO3(4)
NO(g)
NO2(8)
N2O(g)
NO,(8)
NOCI(g)
HNO3(aq)
HNO,(g)
-168.5
282.7
Berylium
Be(s).
BeO(s)
Be(OH)2(s)
-235.1
9.44
-277,7
174.76
160.7
Iron
472.7
455.5
153.3
415.5
369.8
180.5
191.50
Felg)
Fe(s)
Fe2"(aq)
Fe"(aq)
-608.4
-579.1
13.77
-1273.02
-910.4
212.1
27.15
113.4
-905.8
-817.9
50.21
-110.5
-137.2
197.9
--80.29
-26.50
111.3
-87.86
-84.93
-46.19
- 16.66
192.5
-393.5
-487.0
-394.4
213.6
Bromine
-47.69
- 10.54
293.3
-132.5
-79.31
113.4
-392.4
159.8
111.8
82.38
174.9
Br(g)
Br"(aq)
Br2(g)
-341.8
-302.3
117.9
159.4
238.5
FeCh(s)
FeCly(s)
FeO(s)
Fe,O,(s)
Fe,O(s)
FeS2(s)
95.40
-120.9
-102.8
80.71
Cesium
-400
-334
142.3
0.0
30.71
3.14
245.3
76.50
49.53
175.6
Cslg)
Cs()
-271.9
-255.2
60.75
-314.4
-203.0
94.6
92.07
85.15
152.3
2.09
0.03
-822.16
-1117.1
-740.98
89.96
-365.6
--184.0
151
ABr(g)
-36.23
-53.22
198.49
Cs(s)
CSCI(s)
-1014.2
146.4
90.37
86.71
210.62
--442.8
-414.4
101.2
-160.1
52.92
240.45
Calcium
-171.5
33.84
51.84
179.3
145.5
154.8
81.6
103.59
220.0
Ca(g)
Ca(s)
CaCO,(s, calcite)
CaCla(s)
CaF2(s)
CaO(s)
Ca(OH)2(s)
CasO,(s)
Chlorine
Lead
41.4
121.7
105.7
165.2
9.66
98.28
304.3
CI(g)
CI"(aq)
C2(g)
НClag)
HC(g)
68.85
Pb(s)
РЪВrs(9)
РЬСО,)
-1207.1
-1128.76
92.88
-167.2
-131.2
56.5
-277.4
-260.7
161
52.6
66.3
264
-795.8
-748.1
104.6
222.96
-206.6
-110.5
146
699.1
-625.5
131.0
-1219.6
-1167.3
68.87
-167.2
-131.2
56.5
--246.9
303.3
-134.3
-73.94
266.4
Pb(NOsh(aq)
Pb(NO,)2(s)
PbO(s)
-421.3
-635.5
-604.17
39.75
-92.30
-95.27
186.69
-451.9
Oxygen
Og)
Olg)
Os(8)
OH (aq)
-986.2
-898.5
83.4
Chromium
-2173
-187.9
68.70
247.5
230.1
161.0
-1434.0
-1321.8
106.7
397.5
352.6
174.2
205.0
Cr(g)
Cr(s)
CryOs(s)
Lithium
Carbon
23.6
Li(g)
159.3
126.6
138.8
142.3
163.4
237.6
718.4
672.9
158.0
-1058.1
81.2
-230.0
-157.3
-10.7
Cg)
C(s, diamond)
C(s, graphite)
CCL(8)
CC4()
CF(g)
CH,8)
Сн8)
-1139.7
29.09.
Li(s)
Li*(aq)
Li"(g)
LICI(6)
1.88
2.84
2.43
-241.82
-228.57
188.83
69.91
Н.Os)
Н.О()
HO28)
H2O2(g)
-278.5
685.7
Cobalt
-273.4
12.2
5.69
-285.83
-136.10
-237.13
-105.48
439
393
179
648.5
133.0
Co(g)
Co(s)
-106.7
-64.0
309.4
28.4
-408.3
-384.0
59.30
232.9
- 139.3
-68.6
214.4
- 187.8
-120.4
109.6
Copper
Cutg)
Cu(s)
Magnesium
Mg(g)
Mg(e)
MgCl2(s)
-679.9
635.1
262.3
148.6
166.3
33.30
Phosphorus
P(g)
P2(g)
-74.8
-50.8
186.3
338.4
298.6
147.1
112.5
32.51
316.4
280.0
163.2
226.77
209.2
200.8
-641.6
-592.1
89.6
144.3
103.7
218.1
1112
APPENDIX C
Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C)
1114
Ане
APPENDIX
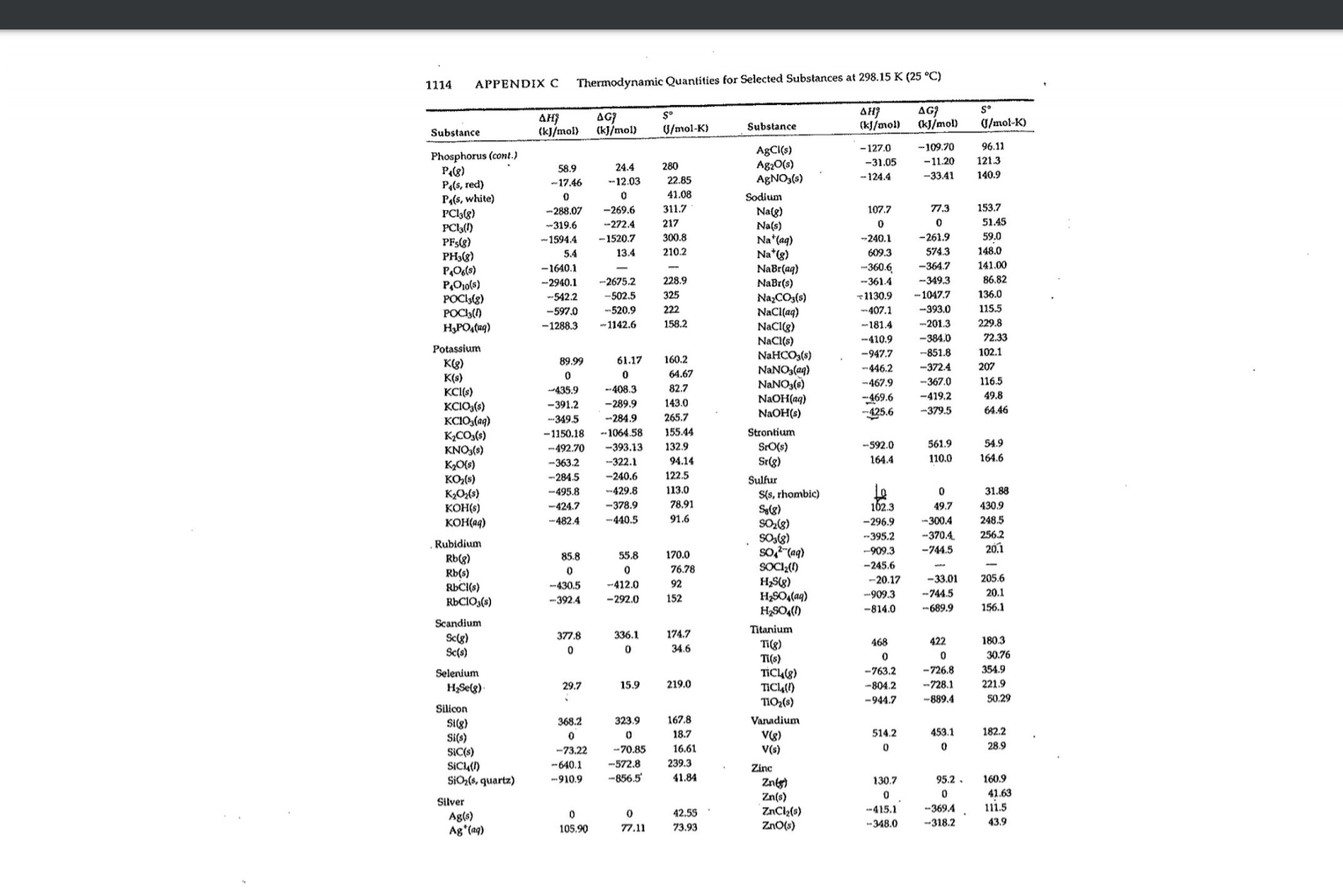
Transcribed Image Text:APPENDIX C
Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C)
1114
AG}
(kJ/mol)
S°
AH
(kJ/mol) (kJ/mol)
AG}
ану
(kJ/mol)
Substance
U/mol-K)
Substance
U/mol-K)
96.11
A8CI(s)
Ag20(s)
AGNO,(s)
-127.0
-109.70
Phosphorus (cont.)
Plg)
Pa(s, red)
Pa(s, white)
PCly(g)
PC()
PFs(g)
PH3(g)
P,O6(s)
P,O10(s)
POCI(g)
-31.05
-11.20
121.3
58.9
24.4
280
-124.4
-33.41
140.9
-17.46
-12.03
22.85
41.08
Sodium
77.3
153.7
-288.07
-319.6
-269.6
311.7
107.7
Na(g)
Na(s)
Na*(aq)
Na*(g)
NaBr(aq)
NaBr(s)
NazCOs(s)
NaCl(aq)
NaCl(g)
NaCl(s)
NAHCO,(s)
NANO3(ag)
NANO3(6)
NaOH(aq)
NaOH(s)
-272.4
217
51.45
-1594.4
-1520.7
300.8
-240.1
-261.9
59.0
5.4
13.4
210.2
609.3
574.3
148.0
-1640.1
-360.6
-364.7
141.00
-2940.1
-2675.2
228.9
-361.4
-349.3
86.82
136.0
1130.9
-407.1
-542.2
-502.5
325
-1047.7
-597.0
-520.9
222
-393.0
115.5
POCI()
НРОдаg)
-1288.3
-1142.6
158.2
-181.4
-201.3
229.8
-410.9
-384.0
72.33
Potassium
-947.7
-851.8
102.1
89.99
61.17
160.2
K(g)
K(s)
KCI(s)
KCIO,(s)
KCIO,(ag)
KĄCO(s)
KNO,(s)
KO(s)
KO,(s)
KO2(s)
КОН(6)
коНод)
-446.2
-372.4
207
64.67
-467.9
--367.0
116.5
435.9
--408.3
82.7
49.8
-469.6
-425.6
-419.2
-391.2
-289.9
143.0
-379.5
64.46
--349.5
--284.9
265.7
-1150.18
-1064.58
155.44
Strontium
-592.0
561.9
54.9
-492.70
-363.2
-393.13
132.9
SrO(s)
--322.1
94.14
Sr(g)
164.4
110.0
164.6
-284.5
-240.6
122.5
Sulfur
S(s, rhombic)
S(g)
SO(3)
SO,(8)
So,*"(aq)
SOCI;()
HSg)
HSO,(ag)
HSO,()
--495.8
-429.8
113.0
31.88
-424.7
-378.9
78.91
162.3
49.7
430.9
-482.4
-440.5
91.6
-296.9
-300.4
248.5
--395.2
-370.4.
256.2
Rubidium
-909.3
-744.5
20.1
170.0
76,78
85.8
55.8
Rb(g)
Rb(s)
RЬCI(:)
-245.6
-20.17
-909.3
-33.01
205.6
-430.5
-412.0
92
-744.5
20.1
RBCIO,(s)
-392.4
-292.0
152
-814.0
-689.9
156.1
Scandium
Titanium
377.8
336.1
174.7
Scg)
Sc(s)
468
422
180.3
Ti(g)
Ti(s)
34.6
30.76
354.9
221.9
-763.2
-726.8
ПC48)
TICL4()
TIO2(8)
Selenium
H,Selg)
29.7
15.9
219.0
-804.2
-728.1
-944.7
-889.4
50.29
Silicon
368.2
323.9
167.8
Vanadium
Si(g)
Si(s)
SIC(S)
SICL()
SIO2(s, quartz)
18.7
514.2
453.1
182.2
-73.22
--70.85
16.61
V(s)
28.9
239.3
41.84
-640.1
-572.8
Zinc
-910.9
-856.5
130.7
95.2.
160.9
Zng
Zn(s)
ZnClą(s)
ZnO(s)
41.63
Silver
--415.1
--369.4
111.5
42.55
Ag(s)
Ag (aq)
-348.0
-318.2
43.9
105.90
77.11
73.93
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning