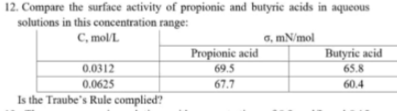
12. Compare the surface activity of propionic and butyric acids in aqueous solutions in this concentration range: C, mol/L. a, mN/mol Propionic acid Butyric acid 0.0312 69.5 65.8 0.0625 Is the Traube's Rule complied? 67.7 60.4
Q: how does the common ion effect affect the precision or accuracy of lead analysis? an example…
A: In a common ion effect, the concentration of making of that ion will be less.
Q: In non-aqueous volumetric methods: I. Carbon dioxide and water is excluded to prevent these from…
A: Non aqueous titration
Q: 13.53 For the cell (13.71), the observed emf at 25°C was 612 mV. When solution X was replaced by a…
A: pH of the solution X is given as follows; pH(X)=pH(S)+ξx-ξsRTF-1ln10 Here, pH(X) is the pH of…
Q: What causes the increase in conductivity of glacial acetic acid upon dilution with distilled water?…
A: Glacial acetic acid means it contains 99.5% of pure acetic acid and the rest water.
Q: A 1.00-L solution contains 2.50×10-4 M Cu(NO3)2 and 1.00×10-3 M ethylenediamine (en). The Kf for…
A:
Q: Regarding Fluoride experiment, determine which are is/true? A. Sodium ion is capable of reacting…
A: Solutions: Fluoride experiment
Q: Calculate the concentration of SCN in each of the following standard solutions. Use 0.0500 M Fe3+…
A: a) Initial molarity of SCN-, M1 = 0.002 M Initial volume, V1 = 1.00 mL Final molarity of SCN-, M2 =…
Q: Calculate the concentration of oxalate ions [C2O42-] in a solution that contains solid silver…
A: The solubility reaction of Ag2C2O4 is => Ag2C2O4 (s) -----> 2 Ag+ (aq) + C2O42- (aq) And the…
Q: To 20,00 ml of 0,0500 mol/l solution of potassium bromide – 19,90 ml of solution of silver nitrate…
A:
Q: SOLVE mathematically, then choose the correct answers: Calculate the pH of solution made by…
A:
Q: The landfill manager suggested using H2CO3 with Na2CO3 as a buffer to minimize pH changes. Do you…
A:
Q: What is the ionic strength of a 0.050 M solution of sodium carbonate? 0.30 M 0.10 M 0.20 M 0.050 M…
A: Here, we have to calculate the ionic strength of a 0.050 M solution of sodium carbonate.
Q: Calculate the concentration in ppm for 0.362 molar of magnesium hydroxide. MM: Mg: 24.30; OH: 17
A:
Q: 0.02 M CH3COOH solution has a resistance of 240 ohm with a cell constant of 0.366 cm'. Calculate the…
A: According to Kohlrausch's law, limiting molar conductivity of weak electrolyte is equal to the sum…
Q: The selectivity of ions towards the ion exchanger increases in concentrated solutions: 1- According…
A: The ion exchange is based on the adsorption of molecules to the chromatographic column driven by the…
Q: he Affect of Metal Degradation Based on Acid Type Four acids are put into varying test tubes and…
A: To determine which acid provides the highest reaction rate. Test Tube-1 (H2SO4 3M) time: 3.68seconds…
Q: (c) Suppose this 0.05 M stock solution was left on the counter, open, overnight before generating…
A: The Solution of CoCl2 is stored at 4 to -20°C for the stability of the concentration. As the…
Q: A 0.9550 g sample of ASA was dissolved in 40.1 mL of standard NaOH. The excess NaOH was neutralized…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: The overall formation constant for HgI42- is 1.0x1030. That is, 1.0x1030 = [HgI42-] / [Hg2+] [I-]4…
A: Given: Initial concentration of Hg2+ ion = 0.016 M Initial concentration of I- ion = 0.72 M…
Q: How many milliliters of lactic acid must be added to 1L of water to produce a solution of 0.477 mM?…
A: The correct answer is given below
Q: The pH of 0.65 M aqueous solution of a weak base (monoprotic) is 10.24. Determine the pKp of this…
A:
Q: Balance the following reaction in basic solution Cr20,2(aq) + SO2(aq) → Cr³*(aq) + SO3(aq)…
A:
Q: Which of the following will be insoluble with sulfates but soluble with halides? CATIONS High…
A: Solubility rule- For halides : They form solution with all cations except Ag+ , Pb+2 , Hg+2 . For…
Q: you will titrate 2.00 mL of 0.0100 M KMnO4 in base with 0.0200 M NaHSO3. Using the correct redox…
A:
Q: A 150-ml sample of water containing Ca2+ and Mg2+ was treated with sodium hydroxide solution to…
A: EDTA complexometric titrations are based on the fact that many metallic ions form stable complexes…
Q: For Kw, the product of [H3O+] and [OH-] is
A:
Q: Salinity of seawater is S% = 3.35%, density if seawater is d = 1.025 g/mL. Titration volume of…
A: Answer: Data given- Salinity of sea water = 3.35% density of sea water = 1.025 g/ml. Concentration…
Q: If two water samples were collected as follows: Sample #1 with a pH = 8.8 and the concentration of…
A: The alkalinity of water is responsible due to the presence of a concentration of ions like…
Q: The OH– concentration in a solution of 0.0100 M NaClO is 5.9×10–5 M. What is the value of Kb for…
A: Given that the concentration of OH- is 5.9×10-5 M and initial concentration of NaClO is 0.0100 M.…
Q: A saturated solution of magnesium hydroxide, Mg(OH)2, has a pH of 10.42 at 25oC. Calculate the Ksp…
A:
Q: 26. What concentration of lead (II) iodide can dissociate in 3.00 M potassium iodide? ksp=[Pb²+][1]…
A: The answer is 9.4×10-10M
Q: A 26.50 mL solution of Fe2+ required 17.50 mL of 0.175 M KMNO, to reach the end point. 5 Fe2 + (aq)…
A:
Q: 95.Reaction of acid and base always produce and 96. The concentrations of standard solution in…
A: 95. Solutions of acid and bas always produces salt and water. 96. The concentration in…
Q: solution of 0.100 M HCl and a solution of 0.100 M NaOH are prepared. A 40.0 mL sample of one of the…
A:
Q: A 150-ml sample of water containing Ca2+ and Mg2+ was treated with sodium hydroxide solution to…
A: Many metal ions form stable complexes with Ethylenediaminetetraacetic acid (EDTA) so EDTA is used to…
Q: An industrial sewage must be discharged into a receiving stream with a pH of 8,3 and alkalinity =…
A: Given: When pH = 8.3 Carbonates = 100% HCO3 - Conc of [HCO3-] = 2 x 10-3 eq/L When pH = 6.3…
Q: A standard solution is a solution of accurately known concentration. What is a standard solution for…
A: Titration is the qualitative method to determine the concentration of unknown solution by adding the…
Q: We use distilled deionized water for making up NaOH solutions in order to minimize or eliminate the…
A: A question based on stoichiometry that is to be accomplished.
Q: 1.00-L solution contains 3.00×10-4 M Cu(NO3)2 and 2.00×10-3 M ethylenediamine (en). The Kf for…
A: Given data: The volume of solution = 1 lt.Morality of Cu(NO3)2 = 3.00×10-4MMorality of…
Q: What is the [NH4] in the solution?
A: Given, Formation constant for Cu(NH3)42+ = 5.0*1012. The given reaction is
Q: Calculate the percent dissoclation of acetic acid (CH,CO,H) In a 0.95 mM aqueous solution of the…
A:
Q: Which of the following is the best way to test the acidity or alkalinity of an unknown liquid sample…
A: a species which liberates hydrogen ion on dissolution it is said to be acid a species which accept…
Q: The activity coefficient of Ni²+ ion (a Ni2* =0.6 nm) in a solution that has an ionic strength of…
A:
Q: Calculate the ionic strength of a 100mM sodium phosphate, 200mM sodium chloride, pH 7.0 buffer. You…
A: Introduction Ionic compounds when dissolved in water dissociate into ions. Ionic strength is the…
Q: Aluminum hydroxide Al(OH)3 is insoluble in water but is soluble in both acidic and basic solutions.…
A: Given : Reaction and some statements To find : correct statement Solution: HSAB stands for hard and…
Q: Problem is attached
A: The Le Chatelier principle tells us that when an equilibrium is modified, it will shift in the…
Q: *76. Using data from Table 16.3, calculate the concentrations of Hg²+(aq), HgCl*(aq), and HgCl2(aq)…
A: We have find out concentration of Hg2+, HgCl+ and HgCl2.
Q: droxide was required to neutralize 25.0 mL of malonic acid to a phenolphthalein end point. Calculate…
A: The equation to calculate the unknown from known values is given by, M1V1=M2V2 where M is the…
Q: What is the concentration of Be(OH)2 (aq) in a solution in which the concentrations of Be2+ and OH−…
A: The equilibrium constant can be written as the ratio of concentration of products to the…
Q: Calculate AG for the following reaction. 2 Cr(s) +3 Cl2(aq) → 2 Cr** (aq) + 6 CI (aq) Hall-Reaction…
A: The expression for the standard Gibbs free energy change can be given as follows:…
Step by step
Solved in 2 steps with 2 images

- Sketch a plot of the fraction of species, f, vs pH for Tyr. Show your calculations for several points on the plot. You want to calculate the fraction of the species for more than 3 of the points. pKa’s for Tyr is 2.24, 9.04, and 10.10.Calculate the alkaline strength of pearl ash (impure potassium carbonate) in terms of percent K2O from the following data: Sample = 0.3500 g; HCl used = 48.03 mL; NaOH used for back titration = 2.02 mL; 1.000 mL HCl = 0.005300 g Na2CO3; 1.000 mL NaOH = 0.02192 g KHC2O4.H2O. (Note: Use the least significant digits in your answer.)A 370.00 mL solution of 0.00185 M A3B4 is added to a 390.00 mL solution of 0.00100 M C3D4. What is pQsp for A3D4?
- Calculate the concentration of A when chemical AB is dissolved in pure water. Ksp = 0.00001331 AB2(s) ⇌ A2+(aq) + 2B¯(aq) Enter answer to 3 sig figs using exponential format.Calculate the alkaline strength of pearl ash (impure potassium carbonate) in terms of percent K2O from the following data: Sample = 0.3500 g; HCl used = 48.03 mL; NaOH used for back titration = 2.02 mL; 1.000 mL HCl = 0.005300 g Na2CO3; 1.000 mL NaOH = 0.02192 g KHC2O4.H2O.Which of the following statements is advantageous? A). The main differences between potentiometric and conductometric titrations are that potentiometric titrations exemplify the analyte sample-by-sample while conductometric titrations show the potential of the analyte B). A conductivity is analyl from the people of the charge. C.) Conductivity increases rapidly as close as equivalence, as a strong acid and a strong base will decrease D). The conductor remains constant according to the viscosity of the pixel, the decomposer and its size, which consists of design modelling.
- A 200.00 mL solution of 0.00105 M AB4 is added to a 270.00 mL solution of 0.00245 M CD5. What is pQsp for AD4? pQsp = -log(Qsp)The salt K2HPO4 (potassium hydrogen phosphate) is a strong electrolyte but not a strong acid or base. Consider an F = 0.010 M solution of K2HPO4. a) Add the acid/base behavior of hydrogen phosphate by writing a “sequential dissociation scheme” for phosphoric acid (H3PO4). Then write mass balance equations for total phosphate and total potassium and give their numerical values. b) Write the charge balance equation for this solution. How may total species are there? [Count species in the charge balance equation and add any neutrals.] How many additional equations are needed for systematic treatment of equilibrium? Name them (give their usual symbols) c) Consider the KHPO4−(aq) ion pair. This is a new species, so what additional equation would you need? Tell how you would alter each charge or mass balance equation (if necessary) to include this species.The Henderson-Hasselbalch equation: a. relates the pH of a solution to the pKa and the concentration of acid and salt. b. allows the graphical determination of the molecular weight of a weak acid from its pH alone. c. is equally useful with solutions of acetic acid and of hydrochloric acid. d. employs the same value for pKa for all weak acids.
- 4. The molar absorptivities of the indicator weak acid HIn (K_{a} = 1.42 * 10 ^ - 5) and its conjugate base In at 570 were determined as epsilon(HIn) = 7120 and ε(In) = 961. The optical length b = 1.00 cm. (a) What is the absorbance of an unbuffered indicator solution having total indicator concentration 8.0 * 10 ^ - 5 (b) What is the absorbance of a buffered indicator solution having total indicator concentration 8.0 * 10 ^ - 5 and pH = 6.5 .Calculate the alkaline strength of pearl ash ( impure potassium carbonate) in terms of percent K2O from the following data: Sample=0.3500 g; HCl used=48.03 m; NaOH used for back titration=2.02 mL; 1.000 mL HCl = 0.005300 g Na2CO3; 1.000 mL NaOH = 0.02192 g KHC2O4·H2OYou have performed an iodimetric titration using a commercial vitamin C tablet. Based on the following information below, calculate the %(w/w) of vitamin C(MM=176.16 g/mol) in the tablet: Mass of tablet dissolved in 250.0 mL: 5.422 g Aliquot volume of sample titrated: 25.00 mL Concentration of KIO3: 0.023 M Final burrette volume: 41.31 mL Initial burrette volume: 8.89 mL Blank volume: 0.14 mL



