1:39 ll LTE Question 15 of 20 Submit nsider the balanced chemical reaction below. ow many grams of carbon monoxide are required to react with 6.4 g of iron(III) oxide? Fe2O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g) 1 2 Based on the set up your table from the previous step, determine the grams of carbon monoxide required. massco = 5 RESET 6.4 3.2 19 0.56 1.1 1.7 2.2 3.4 7.6
1:39 ll LTE Question 15 of 20 Submit nsider the balanced chemical reaction below. ow many grams of carbon monoxide are required to react with 6.4 g of iron(III) oxide? Fe2O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g) 1 2 Based on the set up your table from the previous step, determine the grams of carbon monoxide required. massco = 5 RESET 6.4 3.2 19 0.56 1.1 1.7 2.2 3.4 7.6
Chapter5: Gases
Section: Chapter Questions
Problem 73E: Consider the reaction between 50.0 mL liquid methanol, CH3OH (density = 0.850 g/mL), and 22.8 L O2...
Related questions
Question
How to solve
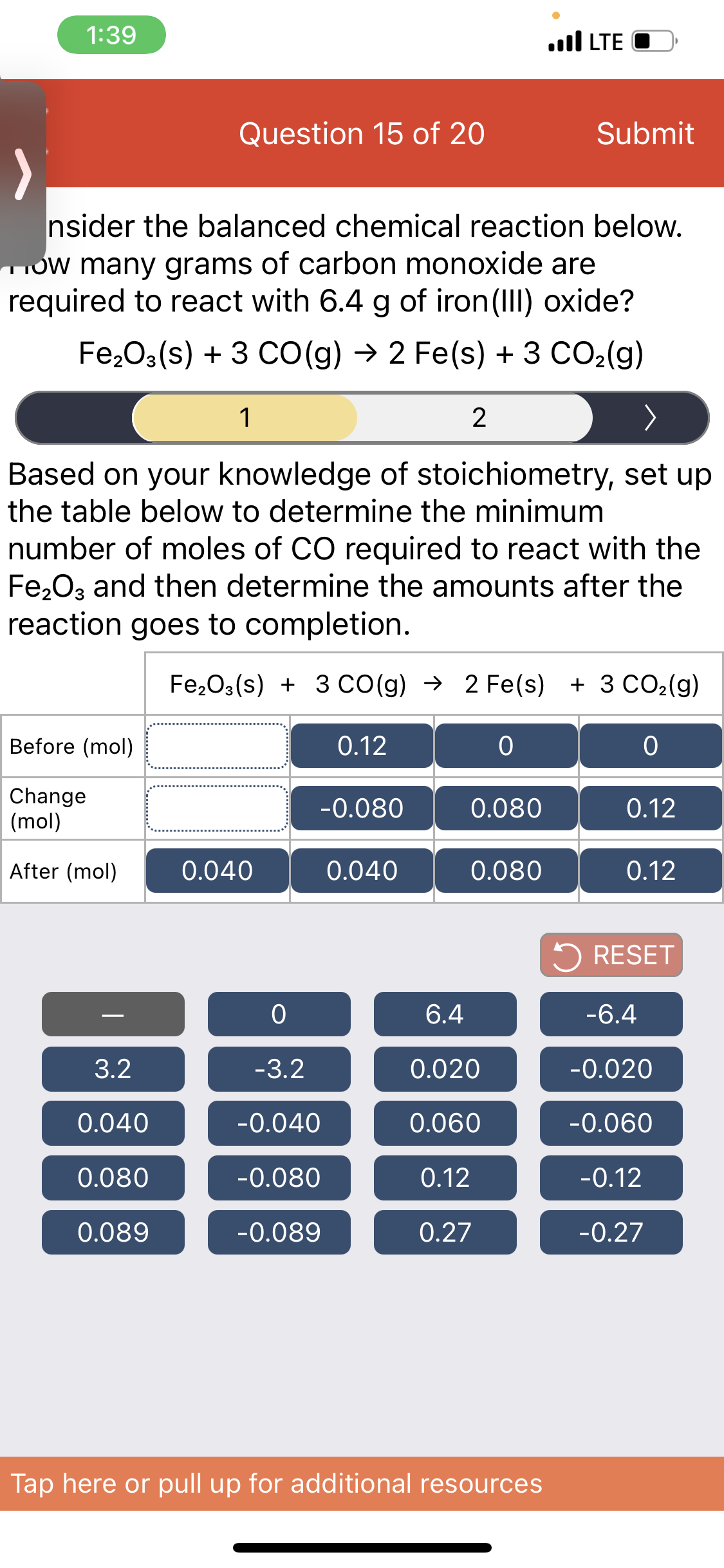
Transcribed Image Text:1:39
ull LTE
Question 15 of 20
Submit
nsider the balanced chemical reaction below.
ow many grams of carbon monoxide are
required to react with 6.4 g of iron (III) oxide?
Fe,O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g)
1
Based on your knowledge of stoichiometry, set up
the table below to determine the minimum
number of moles of CO required to react with the
Fe2O3 and then determine the amounts after the
reaction goes to completion.
Fe,O3(s) + 3 Co(g) → 2 Fe(s)
+ 3 CO2(g)
Before (mol)
0.12
Change
(mol)
-0.080
0.080
0.12
After (mol)
0.040
0.040
0.080
0.12
5 RESET
6.4
-6.4
-
3.2
-3.2
0.020
-0.020
0.040
-0.040
0.060
-0.060
0.080
-0.080
0.12
-0.12
0.089
-0.089
0.27
-0.27
Tap here or pull up for additional resources

Transcribed Image Text:1:39
ull LTE
Question 15 of 20
Submit
nsider the balanced chemical reaction below.
ow many grams of carbon monoxide are
required to react with 6.4 g of iron (III) oxide?
Fe,O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g)
1
Based on the set up your table from the previous
step, determine the grams of carbon monoxide
required.
massco =
5 RESET
6.4
3.2
19
0.56
1.1
1.7
2.2
3.4
7.6
Tap here or pull up for additional resources
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning