Q: Convert each of the following OH concentrations to pH. 1.4 M i eTextbook and Media 2.4 µM i 0.031 M ...
A: It can be calculated by using formula :- pH = 14 + log[OH-]
Q: what is the ΔG when - K constant = 1.2x103 - temperature = 40 degrees - R = 8.31
A: Gibbs Free Energy Equation, ∆G = ∆G° + RT(lnK) ∆G = Gibbs Free Energy ∆G° = Standard Gibbs Free Ener...
Q: sample of a gas is in a reaction vessel under a certain set of conditions such that it possesses 4.3...
A: ANSWER : (a)We know a formula that entropy relates with the microstates : S = K*lnW Here : S = ...
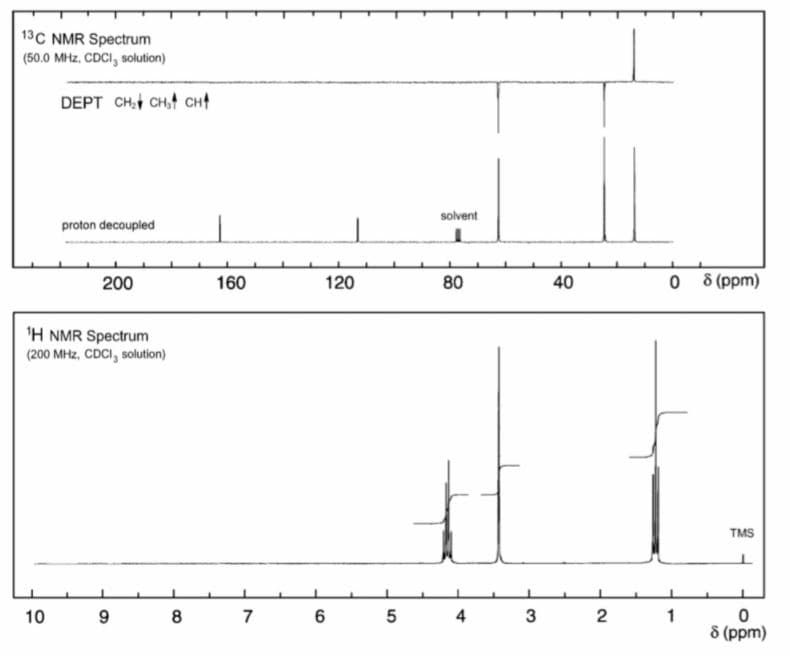
Q: 4. Determine the structure of the compound with molecular formula CaHsO2 that gives the following 'H...
A:
Q: Activity has the largest effect on an equilibrium when the solution contains a low ionic strength an...
A: Activity has the largest effect on an equilibrium when the solution contains, a low ionic strength a...
Q: ) Which of the flowing reactions will have the faster rate and give a better yield? Use drawings of ...
A: Soln
Q: An aqueous solution with 15 g sulfuric acid is dissolved to make 0.2F solution. The specific gravity...
A: The solution contains 15 g sulfuric acid.
Q: Why do we need to know the concept of chiral molecules?
A: The study of chirality in nature has played a very significant role in understanding biochemistry. C...
Q: Suggest a combination of an organic halide and a cuprate reagent appropriate for the preparation of ...
A:
Q: what are the structures for the follwoing c) benzoate reacts with methyl benzoate reacts with ...
A: Since you have posted question with multiple sub-parts, we will solve only first three sub-parts fo...
Q: The decomposition of nitramide in aqueous solution at 25 °C NH,NO2(aq)–N20(g) + H20(1) is first orde...
A:
Q: You're trying to calculate the solubility of a saturated solution of Ba(ClO)2. You start out by writ...
A: Solubility product is the indicator of solubility of a partially soluble substance in a solution.
Q: Calculate the pHpH for the following weak acid. A solution of HCOOHHCOOH has 0.18M0.18M HCOOHHCOOH ...
A: Given, the concentration of HCOOH at equilibrium = 0.18 M The Ka for HCOOH = 1.8×10−4 We have to cal...
Q: A solution of iodine in carbon tetrachloride has a density of 3.22 g/mL and a concentration of 3.00 ...
A: Answer: To calculate the volume percent, we need the volume of iodine and volume of solution. First ...
Q: What is the resulting concentration in molarity when 250.0 mL of 0.1432 M of HCl was mixed with 100....
A:
Q: [0]
A:
Q: A gas mixture is composed of 2.0 moles H2 and I mole N2. Calculate (a) the average molar mass of the...
A: Number of moles of H2 = 2 mole Number of moles of N2 = 1 mole Mole fraction of H2 = 2/3 = 0.667 Mole...
Q: Which of the following statements is/are correct about visible spectrum i. white light passes throug...
A: That portion of electromagnetic radiations that is visible to human eye is called the visible spectr...
Q: HF HO
A:
Q: Which does not belong to the group. I need answer from both questions.
A: This question is related to d-block elements. The elements from the 3rd group to the 12th group of t...
Q: If this is the procedure that we used to adapt a new reaction, what is the mass, molecular weiht, vo...
A: Amount of 2-chloro-5-nitrophenol added = 2.75 g Molar mass of 2-chloro-5-nitrophenol...
Q: In the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses...
A: Given that, lysergic acid diethylamide (LCD, C20H25N3O). It is a weak base, having Kb = 7.6×10-7. Th...
Q: molecules are unlikely to dissolve in water easily. molecules with polar and nonpolar areas polar co...
A: Answer .......... molecules are unlikely to dissolve in water easily ...
Q: What volume in liters would be occupied by 0.22777kg of a material with a density of 0.00523kg/mL?
A: Given mass = 0.22777 kg density = 0.00523 kg/mL
Q: Identify the major species (other than water) that has no effect on pH. Include charge (if any) and ...
A: Soln
Q: Al, Fe, Co *
A: We have to distinguish the ions Al , Fe and Co.
Q: Calculate the [Ag*] of a saturated solution of Agl that has an ionic strength of 0.10 M using activi...
A:
Q: Analyze and answer the problem. Show your complete solution. What mass of AgNO3 must be dissolved i...
A:
Q: Concentrated aqueous HCIO4 has a concentration of 14.8 M. Calculate the concentrations of all ions p...
A:
Q: A student is determining the density of an unknown metal with a mass of 32.56 g. The student partial...
A:
Q: What are some of the ways to ensure that fermentation is not disrupted
A:
Q: Bromine has two naturally occurring isotopes (Br-79 and Br-81) If the natural abundance of Br-79 is ...
A: Given: There are two naturally occurring isotopes of Br. The abundance of isotope 1 = 50.69 % Mass o...
Q: Calculate the buffering index for a solution containing 1 × 10–2 mol L–1 of acetic acid when the sol...
A:
Q: i-step synthetic pathway below by filling in the missing products along Synthesis B Synthesis C CH;C...
A:
Q: identify each substance Acid Base conjugate acid Conjugate base
A:
Q: Test for lipids.
A:
Q: 1. Calculate the percent yield of alkenes in your dehydration. (HINT: What role does the sulfuric ac...
A:
Q: What is the formula fortheedges, expressed as “f”, if the radius of the atom is expressed as “k”: a...
A: In general, edge length is represented by 'a' and radius is represented by 'r' Here , radius is den...
Q: エ 2. Determine which of the following would effectively deprotonate an acid with a pKa of 30. (hint:...
A: Given-
Q: Consider the compounds CH₃OCH₃, CH₃CH₂OH, CH₃CH₂CH₃. Arrange the following compounds from lowest to ...
A: Given compounds are : CH₃OCH₃, CH₃CH₂OH, CH₃CH₂CH₃
Q: ndicate the quantum numbers that describes the followin electrons indicated below: 1. 34th electron ...
A: Soln
Q: Calculate for the buffer capacity if 20 mL of 0.048 M hydrochloric acid is added to 500 mL of buffer...
A: To calculate buffer capacity of sodium acetate and acetic acid buffer when HCl is added. Given data:...
Q: n a replicated experimental procedure for phosgene formation by Bodenstein and Plaut, it was found o...
A: Answer: First of all we will calculate the equilibrium constant for the decomposition reaction with ...
Q: Write the products structures and mechanisms CO2H A = (cat.) HO (a) NC CN LOME ? solvent Ph (i) (iii...
A: The Diels-Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene ...
Q: The rate constant for this first-order reaction is 0.0246 s−1 at 300 °C. A⟶products Calculate th...
A: The given reaction is of first order. k = rate constant = 0.0246 s-1 FInal mass after t = 1.28 mins ...
Q: the volume of a cylinder is a function of its radius(r) and heigh(h) and is given by the equation V=...
A: Given that : The volume (V) is a function of the radius (r) and the height (h) can be given as : V =...
Q: Conjugate acid Conjugate base
A:
Q: The smallest bone in the human body, which is in the ear, has a mass of 0.0030 g. what is the mass ...
A: Given, The smallest bone in the human body which is the ear, has a mass of 0.0030 g mass of bone in ...
Q: Based your answer on the phase diagram of carbon dioxide a. What is the lowest temperature at which ...
A: The Phase Diagram of Carbon Dioxide: The triple point -56.60C and 511 atm. It means that liquid CO2...
Q: Please all solve the issue Sodium nitrate/nitrite stabilizes the red color, adds flavor, and it also...
A: As per the guideline, since you have asked multiple questions, we have solved the first question for...
Find the IHD, cleavage, Functional groups, frequency, base peak, fragment and unknown compound
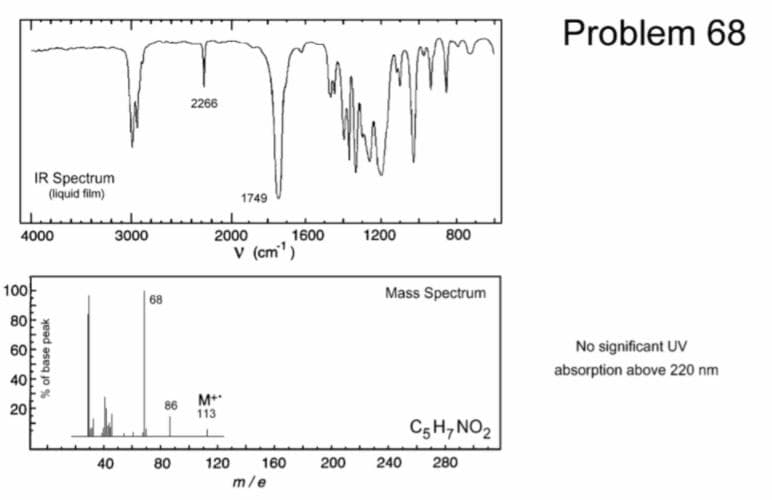
Step by step
Solved in 2 steps with 1 images

- state if true or false 1. Considering that concentration and the size of sample container are the same, a sample with a higher molar absorptivity will have a higher TRANSMITTANCE than a sample with the lower E (molar absorptivity). 2. Considering that concentration and the size of sample container are the same, a sample with a higher molar absorptivity will have a higher ABSORBANCE than a sample with the lower E (molar absorptivity). 3. The higher the concentration of a sample is the higher the transmittance will be. 4. Spectrophotometry is the study of how light interacts with matter 5. During an absorbance reading, light must pass through the opaque side of the cuvette. 6. Distilled water is an acceptable blank solution for any sampleI have 4 Questuion Q1: find the A , T , T% of a 2.4X10-3 mol L-1 solution of a substance whose molar absorptivity is 313 L M-1 cm -1 at 520 nm when measured in a cell of 2.00 cm pathlength ؟ Q2: A sample in a 1 cm cuvette transmits 79% of light at 280 nm , what is the absorbance ? if the molar extinction coefficient is 2.0 M-1 cm-1 , what is the concentration of the sample ? Q3 : the aspirin an analgesic form complex with metal ion which has a molar absorpitvity of 8000 L M-1 cm-1 at ( lenada max 435 nm ) . calculate the absorbance of 6.77 X 10-5 M solution of the complex measured in a 1.00 cm cell at 435 nm ? Q4 :Find the A and T% of a 6.5X10-4 Mol L-1 solution of a drug ( molecular weigth 200 ) whose absorpitvity is 0.026 L cm-1 g-1 at 520 nm when measured in a cell of 2.00 cm pathlength ?FYI: 37-40 is like a),b),c) and d)
- Answer is Ve = 47.79mL ; pH = 8.74 , 5.35 , 4.87 , 4.40 , 3.22 , 2.58 Please show all work detail pleaseAssuming that the path length of your cuvette is exactly 1.00 cm, calculate the molar absorptivity for each of your KMn04 solutions at the wavelength of maximum absorbance, and take the average value. Tube 8.0x10^-2M KMnO4 Distilled water Sulfuric acid %T @520nm 1 1 ml 7 ml 2ml 68 2 2 ml 6 ml 2ml 53 3 3 ml 5 ml 2ml 45 4 4 ml 4 ml 2ml 29c32 plz answer what u can
- IR Spec question: https://webbook.nist.gov/cgi/cbook.cgi?ID=B6007500&Mask=80 The spec is chloroform, deutero. I don't know what the peaks are around 3200 cm-1, 3000 cm-1 and 2300 cm-1.When we perform Bradford Assay to measure protein concentration, the spectrophotometer is set 595 nm because: a. the excitation peak of Coomassie Blue is best observed at 595 nm b. when Coomassie Blue binds to proteins, its maximum absorbance shifts to 595 nm c. the reducing effects of Coomassie Blue shifts the protein's maximum absorbance to 595 nm d. myoglobin is best observed at 595 nmThe absorbance values at 250nm of 5 standard solutions, and sample solution of a drug are given below: Conc. (ug/ml) A 250 nm10 0.16820. 0.32930 0.50840. 0.66050 0.846Sample. 0.661Calculate the concentration of the sample
- I really need help getting this question right pls pls help me asap and make sure everything’s correct 100000% pls and thank youanswer is on bracket. thank you.Calculate the concentration of lycopene in your orange fraction using using Beer's law equation A=ExBxC A is the measured absorbance, E is the molar extinction coefficient (M-1cm-1) at a particular wavelength, B is the path length (in cm) and C is the analyte concentration (M). A= 0.602. Wavelenght is 500.95nm. Molar extinction coefficients is 123,000M-1cm-1

