Q: 1. Assuming the validity of Raoult's law, do the following calculations for the benzene/toluene…
A:
Q: None
A: Step 1:Valency of Te atom = 6Valency of S atom = 6so each S atom share 2 electrons to become octet.…
Q: A gas occupies 15.5 L at 0°C and 879 torr. What temperature would be required to increase the…
A: Given:Volume = 15.5LInitial temperature T1 = 0°CInitial pressure P1 = 879 torrFinal pressure P2 =…
Q: For 1 and 2, show the products of the following reaction:
A: Step 1:Step 2:
Q: 71. How much heat (in kJ) is required to convert 25.0 g of liquid CCl4, initially at 35 °C, to…
A: Step 1:To solve this problem, we'll use the formula: q=m×C×ΔTWhere:q is the…
Q: Which of the following gas samples would have the largest volume, if all samples are at the same…
A: The objective of the question is to determine which of the given gas samples would occupy the…
Q: A student dissolves 12.2 g of lithium chloride (LiC1) in 200. g of water in a well-insulated open…
A: Step 1: Step 2: Step 3: Step 4:
Q: 2. Predict the products. (Hint: for the 2rd one, remember the driving force for a Claisen reaction,…
A: The first reaction involves treating a compound with LDA (lithium diisopropylamide) followed by MeI…
Q: Considering which of the OH groups should be more accessible to MsCl, suggest a mechanism for the…
A: The objective of the question is to propose a mechanism for the rearrangement of cyclobutane to…
Q: Considering which of the OH groups should be more accessible to MsCl, suggest a mechanism for the…
A: Step 1: Step 2: Step 3: Step 4:
Q: match the corresponding molecule to the spectrum (only one molecule per spectrum )
A: Step 1:Given Two infrared spectra of organic compounds. Match the spectrum with compounds. Step…
Q: 45) Determine whether each pair of substances forms a homogeneous solution when combined. For those…
A: A) LiF and C6H14 (Lithium Fluoride and Hexane)LiF is an ionic compound (metal and non-metal).C6H14…
Q: 3
A: the number of 1,2,4-triazines with the molecular formula C₃HFIN₃, we need to consider the possible…
Q: A student reacts a sample of Alka-Seltzer (the reactive ingredient is NaHCO3)with hydrochloric acid…
A: The sodium bicarbonate in Alka-Seltzer reacts with stomach acid and neutralize the acid forming…
Q: Please don't provide handwritten solution ....
A: Step 1:Step 2:Step 3:Step 4:
Q: Present a synthesis for the transformation depicted
A: Step 1: Step 2: Step 3: Step 4:
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Ni(s) + O2(g) → 2NiO(s)…
A: if any further issues regarding writing or some doubt let me know I will clarify you again.Thank you…
Q: dont provide handwriting soliution ......
A: Step 1: given line segment with endpoint (-4, -1) and (0, -2) We have…
Q: Please answer the following chem question
A: Step 1:Step 2:Step 3:
Q: from the list, match a molecule (only one for each case) to the IR spectrum A and B.
A: Spectrum AI only encircle the necessary peaks to identify the molecule. #1 indicate a peak for O-H…
Q: Given the standard enthalpy changes for the following two reactions: (1) N2(g) + O2(g) → 2NO(g) (2)…
A:
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: Non metal like N and O and Cl and C can form only covalent bonds with each other as these are…
Q: explain please
A: Step 1:Step 2: Step 3: Step 4:
Q: Draw a triacylglycerol with 12 carbon chains that are monounsaturated (you can choose the site of…
A: Step 1:Hydrogenation is a reaction where hydrogen atoms are added to the unsaturated compounds…
Q: None
A: Step 1:Step 2:Step 3:
Q: None
A: To calculate the percent of NaCl in the mixture, we first need to determine the mass of NaCl in the…
Q: Enter electrons as e*. Use smallest possible integer coefficients for ALL reactions. If a box is not…
A: Step 1:Galvanic cell is an electrochemical cell that converts chemical energy into electrical energy…
Q: Show work, thank you!
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: F3 Modify the structure to draw the organic product of the following reaction. Note that [O] is…
A: The given structure is CH2CHOHCH3, which is a secondary alcohol. The presence of [O] indicates that…
Q: A sample of krypton gas at a pressure of 1.12 atm and a temperature of 28.5 °C, occupies a volume of…
A: The objective of the question is to find the pressure of a sample of krypton gas after it is allowed…
Q: + H-Br Energy I 3. H-Br addition to 3-methylbut-1-ene forms 2-bromo-2-methylbutane as the major…
A: Step 1: Step 2: Step 3: Step 4:
Q: A sample of krypton gas at a pressure of 794 mm Hg and a temperature of 71 °C, occupies a volume of…
A: The objective of the question is to find the volume of a sample of krypton gas when it is cooled…
Q: Debye and Hückel were able to show that in dilute solutions the activity coefficient i of an ionic…
A: Step 1: Step 2: Step 3: Step 4:
Q: solve step by step
A: Step 1: Step 2: Step 3: Step 4:
Q: need help with major product
A:
Q: None
A: To find out how many grams of H2C2O4 are needed to react with 29.3 grams of KOH, you'll need to use…
Q: Using chemistry how do you find/choose the right spf
A: Step 1:To find or choose the right SPF (sun protection factor) using chemistry, we need to…
Q: Please classify and explain the following as either aromatic (A) or nonaromatic (NA)
A: Step 1:To answer this question, we must be familiar with the 4 laws of aromaticity. 1. It must be…
Q: None
A: The balanced reaction for the same will be given as:- HCL + NaOH = NaCl + H2O The change in enthalpy…
Q: Finish the reaction by drawing the initial Diels-Alder product. H3C CH3 H₂C CH3 H CH
A: Diels- alder reaction is 4π+2 cycloaddition reaction cyclization is possible in tharmal condition.…
Q: Construct the phase diagram for for gold and thallium. Pure gold melts at 1063°C and pure thallium…
A: a) Eutectic Phase Composition: 27% Au (calculable if enthalpy of melting is present). (b) Melt:…
Q: Solubility of Ca(OH)2a) Give a balanced reaction equation for the reaction of Ca(OH)2 with…
A: The objective of the question is to understand the chemical reactions involving calcium hydroxide…
Q: 1. A pure solvent freezes at 6.2oC. Determine the molar mass of a 0.8712 g nonelectrolyte dissolved…
A: Step 1: Step 2: Step 3: Step 4:
Q: How much energy must be supplied to break a single aluminum-27 nucleus into separated protons and…
A: The objective of this question is to calculate the energy required to break a single aluminum-27…
Q: Draw a triacylglycerol with 12 carbon chains that are monounsaturated (you can choose the site of…
A: Step 1: Any unsaturated fatty acid's carbon-carbon double bonds can be converted to carbon-carbon…
Q: How many grams of water can be heated from 20.00C to 78.00C using 12500.0 J? The specific heat…
A: GUIDE: (1) Heat Energy Formula Q=mc(Tf−Ti)where:Q = energy m = mass of the substancec = specific…
Q: Convert each pressure measurement to atm. 129 psi
A: The objective of the question is to convert the pressure measurement from psi (pounds per square…
Q: Chemistry
A: Step 1:In an electrochemical cell, oxidation takes place at the anode electrode while reduction…
Q: A 'H NMR spectrum is shown for a molecule with the molecular formula of C5H10O2. Draw the structure…
A: Step 1:
Q: Please don't provide handwritten solution.
A: reaction uses phosphoric acid (H3PO4)to dehydrate 2-methylcyclohexanol, eliminating water to form an…
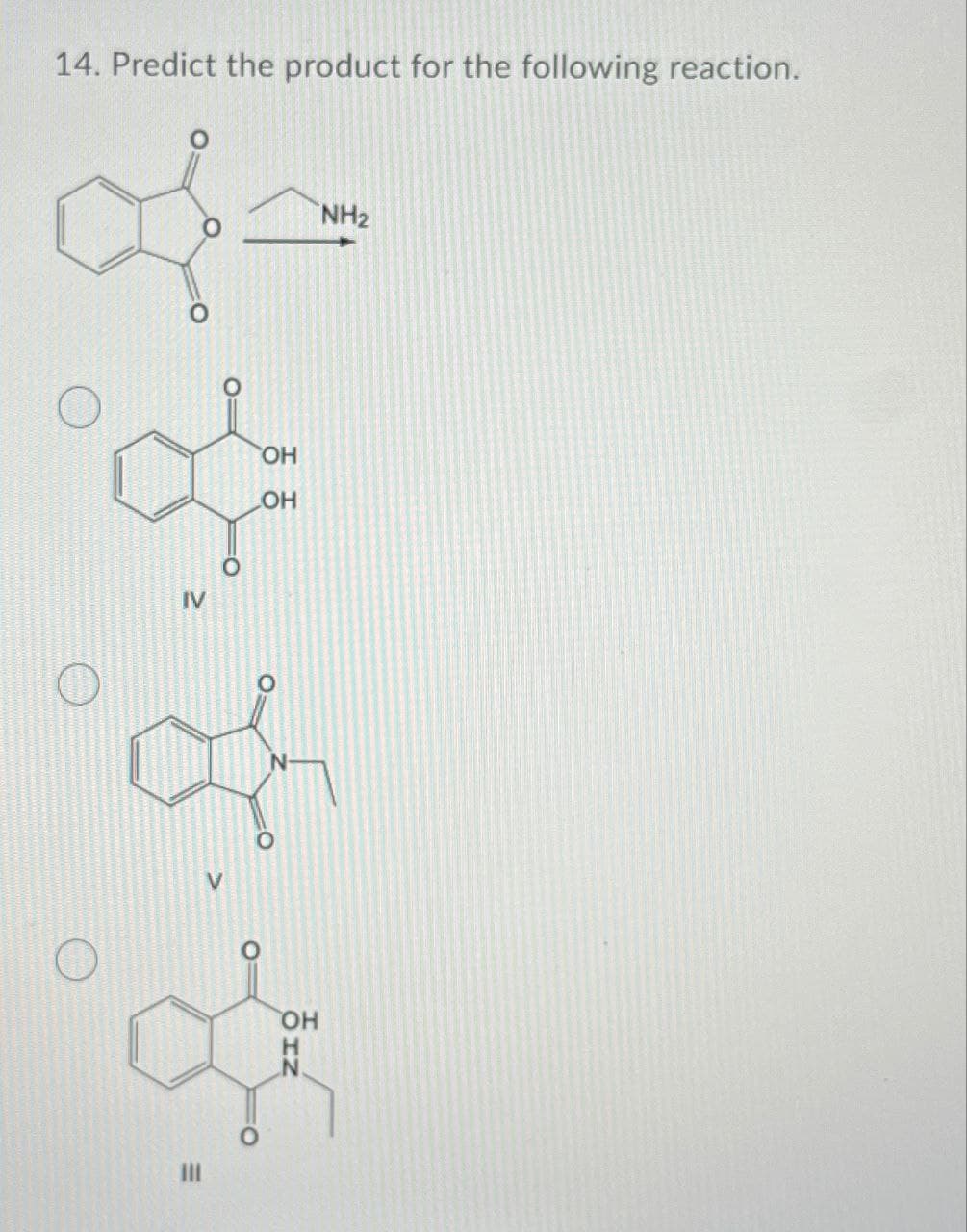
Step by step
Solved in 2 steps with 2 images

- predict the major product of the reaction below15. provide the missing reagents for the following reactionThe enolate derived from diethyl malonate reacts with a variety ofelectrophiles (not just alkyl halides) to form new carbon–carbon bonds.With this in mind, draw the products formed when Na+ −CH(CO2Et)2reacts with each electrophile, followed by treatment with H2O.

