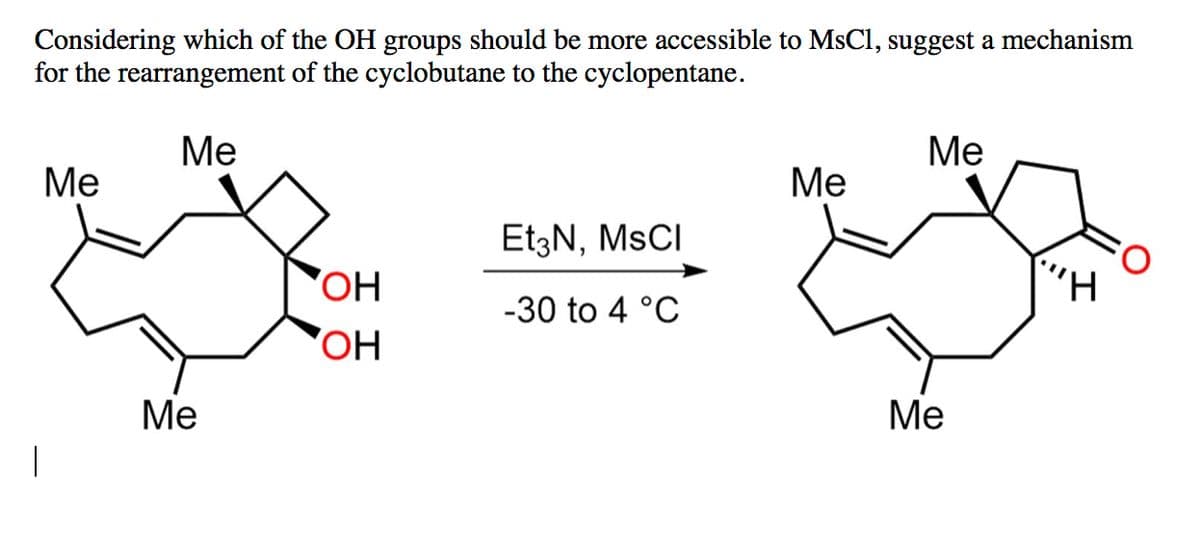
Considering which of the OH groups should be more accessible to MsCl, suggest a mechanism for the rearrangement of the cyclobutane to the cyclopentane. Me Me Me Me Et3N, MSCI OH -30 to 4 °C OH Me Me
Q: Answer the following question correctly
A:
Q: The peaks in a UV-Visible spectrum are associated with which molecular changes? electronic state…
A: In a UV-Visible spectrum, the peaks are associated with electronic state changes within molecules.…
Q: What is the chemistry involved in sunscreens? What is the process of making sunscreens? Chemicals…
A: EXPLAINED ABOVE IN DETAILED.
Q: Draw products
A:
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: Approach to solving the question:NoneDetailed explanation:NoneExamples:NoneKey references:None
Q: Please answer the following question correctly
A:
Q: Calculate the G°rxn using the following information. 2 HNO3(aq) +NO(g) → 3 NO2(g) + H2O(l) AG'rxn =?…
A:
Q: 137Cs has a half-life of 30.2 years. How many years will it take for a 100.0-g sample to decay to…
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: None
A: EXPLAINED ABOVE IN DETAILED
Q: n What would be the expected result from the following reaction? (cy is an abbreviation of…
A: Step 1: The expected result from the following reaction is So, pentanal is the correct answer.
Q: How many protons are present in Pb-206? How many neutrons are present in 17N? Give text symbol of…
A: Use a periodic table to see the values1. Atomic Number = ProtonsPb atomic number is 82Protons=822.…
Q: Heeeeeeeeee solve alllll
A: The IUPAC names for the given anhydrides are:For the first structure, it consists of two pentanoic…
Q: d. H3N H₂C!!!! H3CW H H-N 'N' Ph 4|1!!! HCl(aq)
A: Step 1: Identify the Central Atom and ChargeThe central atom is Nitrogen (N) with three single…
Q: Consider the following reaction: H₂O: OH + HCI What is the rate-determining step in this reaction?…
A:
Q: Predict the products of the following reactions. Show stereochemistry when applicable.
A: The second reaction, the addition may occur via syn or anti pathway. So both cases are possible
Q: None
A: Step 1:We must be familiar with the different monosaccharide to be able to answer this question. One…
Q: 1. A pure solvent freezes at 6.2oC. Determine the molar mass of a 0.8712 g nonelectrolyte dissolved…
A: Step 1: Step 2: Step 3: Step 4:
Q: What is the enegry numbers for the table?1 calorie (cal) = 4.184 J1 kWh = 3.600 × 106 J
A: Please see the attached image for the solution. If there are queries,please do not hesitate to ask.…
Q: Determine the predominant type of reaction: SN1, SN2, E1 and E2. Draw the major products only. Show…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Aromatic resonance is a theory that explains why aromatic compounds are so stable.This idea entails…
Q: Consider a voltaic cell that consists of a Ag+/Ag half-cell and a standard hydrogen electrode (SHE)…
A:
Q: given the iupac name ... dont provide handwriting solution....
A: An amide is usually an organic compound that contains a functional group consisting of an acyl group…
Q: 3. Propose starting materials and reagents to achieve the following synthesis. Carbonyls with 3…
A: I can help you with understanding the general approach to this question based on the information in…
Q: Question 1 Which set of Newman projections represents correct the most stable and the least stable…
A: There are two kinds of Newman projections: Staggered and Eclipsed. Staggered conformation is always…
Q: Give the major organic product or missing starting material for the following reaction do either…
A:
Q: Help me solve this.
A: Given: pH=3.444;[HA]=0.222M;T=25°C=298.15KStep 1: Write the dissociation of…
Q: ABCD Question 2 Please predict the product for each of the following reactions. Make sure to clearly…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Include sterioisomers
A:
Q: Question 27 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: Determine the predominant type of reaction: SN1, SN2, E1 and E2. Draw the major products only.…
A:
Q: Question 17 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: The generic acid HA has a Ka = 3.5 x 10-5. Determine the pH of a solution that is 0.22 M in A- (the…
A: Given: [A−]=0.22M;Ka=3.5x10−5Step 1: Solve for pKb and Kb.…
Q: The "particle in a ring" is another relatively simple 1-D quantum mechanics problem: The Schrödinger…
A: PART A: PART B:PART C:PART D:
Q: balance C6H12O6+CO2+H2O with explanation
A:
Q: 1. Compare ... M 2. Standard ... 3. AS surroundi... 4. AG° = AH°... 5. AG: Pre... 1req 6. AG:…
A: Step 1:First, we need to find the standard free energy change of the reaction, ΔG°. We can do this…
Q: What is the mechanism of this aldol reaction? Thank you!
A: Thank you.
Q: dont provide handwriting solution .....
A: References Smith, M. B. (2020). March's advanced organic chemistry: reactions, mechanisms, and…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the alkoxide side product.…
A: LiAlH4 is a reducing agent. Here LiAlH4 is a source of H+ which attacks carbonyl carbon and then…
Q: Infertility issues in both men and women can prompt hormonal testing to determine the cause.…
A: Approach to solving the question: Detailed explanation:True.Hormone testing plays a crucial role in…
Q: Solve all complete solutions
A: Here are the IUPAC names and structures of the esters provided:Propyl propanoate:…
Q: Predict the major product for the following reaction. Ans: OH H3O+
A: Step 1 :
Q: Draw the most stable chair conformation of each of the following compounds. (a)cis-1,1,4 -…
A: Step 1: Step 2: Step 3: Step 4:
Q: Help answer.
A: Step 1: We know that, ΔG=−RTlnKspThe Ksp for CaF2 is 3.9 × 10-11 at 25 °C. Step 2:…
Q: The wow expert Hand written solution is not allowed please
A: Step 1:Given, decomposition reaction is2 NOBr (g) ⇌ 2 NO (g) + 1 Br2 (g) Step 2:Kp is an equilibrium…
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: if any doubt regarding the mechanism then let me know I will send again .
Q: 1) A beaker containing calcium chloride (CaCl2) is divided by a membrane. Initially, the…
A:
Q: I would appreciate any help on how to solve this problem :)
A: Given: masNO2=10.2g;MN=14.01g/mol;MO=16.00g/mol2NO2(g)→N2O4(g)ΔH°=−55.3kJStep 1: Is the…
Q: Predict the major products of this organic reaction. NBS ? hv
A:
Q: Don't use hend raiting and Step by step solutions
A: Total answers posted by the expert is: 6759 Answer:The peak labelled 1 matches with the proton sets…
Q: Describe the three main mechanisms for transport across membranes?
A: Reference:https://life.nthu.edu.tw/~b830473/transmechanism.htmlhttps://www.inspiritvr.com/mechanisms…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4A solution of pure (S)-2-iodobutane ([a] = +15.90°) in acetone is allowed to react with radioactive iodide, 131I -, until 1.0% of the iodobutane contains radioactive iodine. The specific rotation of this recovered iodobutane is found to be +15.58°. (a) What does this result suggest about the mechanism of the reaction of 2-iodobutane with iodide ion?8 Give logical fragmentation reactions to account for the following ions observed in these mass spectra.(a) n-octane: 114, 85, 71, 57 (b) methylcyclohexane: 98, 83 (c) 2-methylpent-2-ene: 84, 69(d) pentan-1-ol: 70, 55, 41, 31
- Qa 18. Bromination of 5a-cholestan-3-one occurs at C-2 or C-4 to give 2 regioisomers. Two unsaturated ketones with lambdamax = 230 nm and lambdamax = 241 nm are yielded during dehydrobromination. Use Woodward-Fieser rules to distinguish between the two.at 25C, which of the following reactions of PbS and H2O2 is more favourable?1) PbS + 4H2O2 -> SO2 + PbO2 + 4H2O2) PbS + H2O2 -> PbSO4 + H2OVirgin coconut oil contains about 46-48% of lauric acid (CH3(CH2)10COOH). In thepresence acid catalyst and an alcohol, propose the mechanism of this reaction.
- The reaction of 2,2-dimethyl-1-propanol [(CH3)3CCH2OH], also known by the common name neopentyl alcohol, with HBr is very slow and gives 2-bromo-2-methylbutane as the major product.Give a mechanistic explanation for these observations.Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained: Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.Conformation control of reaction mechansims. Consider the reaction below: Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.
- I need help proposing a detailed electron pushing mechanism showing the cyclinization of the amide intermediate to benzimidazole. I must show all resonance stabilized carbocations with RSCC letters. Please help!Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answerGive logical fragmentation reactions to account for the following ions observed in these mass spectra. (a) 2-methylpent-2-ene: 84, 69 (b) pentan-1-ol: 70, 55, 41, 31

