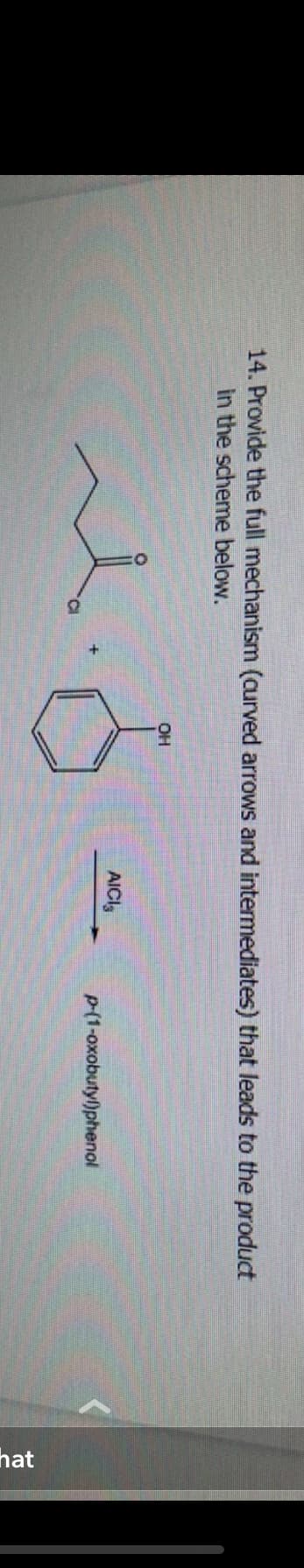
14. Provide the full mechanism (curved arrows and intermediates) that leads to the product in the scheme below. C OH AICI p-(1-oxobutyl)phenol
Q: Draw structural formulas for the major organic product of the reagents shown. CH3 NO₂ + HNO3 H₂SO4
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: NH₂ The following reaction will give 3 different organic products. Sketch the three products. CH3Br…
A: The Gabriel synthesis is a chemical reaction that transforms primary alkyl halides into primary…
Q: Identify the intermediates in the above reaction energy diagram.
A:
Q: The boiling point of a substance is affected by atmospheric pressure.
A: (A) Hydrogen bonding cause substances to have higher melting points than predicted by periodic…
Q: What is the major organic product of the following Claisen Condensation reaction? excess OMe 1. Na…
A: Question belongs to mixed condensation reaction. Condensation reaction between aldehyde and ester.
Q: calculate. V AH₂ for: lattice IE, елегду IE₂ e affinity of F bond energy of F₂ sublimation E Mg(e) +…
A:
Q: Pick the drawing to show a hydrogen bonding interaction A B C D E
A:
Q: 8. Provide the structure of the 1,4 addition product for the reaction of 1,3-hexadiene with Br₂/…
A:
Q: Draw the missing products and/or reagents in the following multistep synthesis. Ignore any inorganic…
A: Here, we have to find the missing products and/or reactants for the given multistep synthesis.
Q: QUESTION 2 ATO The following mechanism has been proposed for the gas-phase exothermic reaction of…
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: Briefly explain the theory of Clark and Lyons about the first enzyme-based electrode in detecting…
A: To explain the theory of Clark and Lyons about the first enzyme-based electrode in detecting glucose
Q: Calculate the molarity of the acid using the titration data. b) If we assume that the titrated…
A: We are given two methods titration method and pH method We have to find molarity of acid, pH if…
Q: Which of the following statements is INCORRECT concerning ideal gases? Select one: a.Particles…
A:
Q: A buffered solution containing dissolved aniline, C, H, NH,, and aniline hydrochloride, CH₂NH₂Cl,…
A: A buffer solution of aniline, C6H5NH2 and aniline hydrochloride, C6H5NH3Cl has a pH of 5.33 . The…
Q: H
A:
Q: 15. Draw the full cleavage of 2-ethoxypropane by HC scheme on your paper, and then draw in the…
A:
Q: Determine the electron geometry and explain your answer for one of the C atoms in H₂CCH2.
A:
Q: Airbags used in automobiles are inflated with nitrogen gas (28.01 g/mol) produced from the reaction…
A:
Q: Draw the products of the following reaction sequence. Ignore any inorganic byproducts formed. 1.…
A:
Q: An acid-base reaction based on the Bronsted-Lowry definition involves: O the transfer of a…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: In the titration of 20.00 mL of 0.1000 M AlCl3 with 0.1500 M AgNO3, what is the volume of titrant…
A:
Q: What is the assumption we can make to simplify the Charge Balance equation if we have a solution of…
A: The correct option is option (3).
Q: Provide a synthesis of the following molecule starting from toluene. CH3 ber CH3 Draw the 4 possible…
A:
Q: In Experiment 4, we used titration to determine the total acid content of samples, which we reported…
A: #A: The balanced equation for the reaction of the monoprotic acid, HA(aq) with NaOH(aq) is: HA(aq) +…
Q: What is the volume (in liters) of 0.245 mol of a gas that is heated to 88.3 °C and is subjected to a…
A:
Q: 5. NO₂¹ Total # of Valence Electrons: 18 Lewis Structure (show all resonance structures if…
A: In the above question NO2- and H3O+ are given and Lewis structure of these molecules is asked, and…
Q: 7. Provide the major organic product formed when butanoic acid reacts with SOCI₂.
A:
Q: Convert each hydrogen ion concentration into a pH (list pH and pOH answers to 2 decimals). Identify…
A:
Q: (b) The critical constants of Pc, T. and Vme of ethane, C₂He, were given as 48.2 atm, 305.4 K and…
A: Molar mass of ethane is 30 g/mol. Given value of Pc ,Vc and Tc are respectively 48.2 atm, 148…
Q: relationship between concentration and time
A: Given statement is : What is the relationship between concentration and time ? Concentration is…
Q: 21. Which of the following is anti-aromatic (select all that apply)? Å O @0:
A: By using this we can identify the Anti aromatic compound .
Q: A 14.09 g sample of NaBr contains 22.34% Na by mass. Considering the law of constant composition…
A: The law of constant composition is : samples of a pure compound always contains same elements in…
Q: a) Cation exchange is an important aspect of soil fertility (i) Explain what is involved in CEC (ii)…
A: i) CEC stands for Cation exchange capacity. By determining CEC we can understand how much calcium,…
Q: For each chemical reaction in the table below, decide whether the highlighted reactant is a…
A: The answer to the following question is-
Q: Corn can be made into alcohol fuel that is burned in an automobile’s engine to make it move. Put the…
A: Here we have to predict how the energy is converted from one form into other form when alcohol fuel…
Q: (a-b) (c-d) (e-f) OH CI CI HORE ? NH pyridine -OH HAM S CI LIAIH[OC(CH3)3]3 1. LIAIH4 2. H₂O+ 1.…
A: Lithiumaluminiumhydride is a reducing agent. It release hydride ion (H-). According to Q&A…
Q: During and acid/base neutralization titration, 20.23 mL of 0.225 M NaH is required to completely…
A: Given data Volume of NaOH solution = 20.23 mL molarity (M) of the NaOH solution = 0.225 M Volume…
Q: Determine the number of Na* and SO, ions required to form a neutral ionic compound. E D C 312 M N…
A: We have to calculate the numbers of Na+ and SO42- ions required to form neutral compound.
Q: The rate of effusion of an unknown gas is about 1.323 times higher than that of N2. What is the…
A:
Q: On a piece of scratch paper, draw the Lewis structure for the SO32 ion. Then choose the correct…
A: Lewis structure is representation of valence shell electrons of an atom.
Q: For the following reversible reaction, A (g) + B (g)-C (g) Kc = x What is the Kc for the following?…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the correct IUPAC name of the following compound? Problem viewing the image, Click Preview…
A: To give the IUPAC name to the compound , first we will see the long carbon chain and then the…
Q: Which of (a)-(d) indicates the correct order of chemical shifts of the four carbons in the 13C-NMR…
A:
Q: The molecules in a sample of liquid propionitrile, CH3CH2C≡N, are attracted to each other by which…
A: we have to determine the intermolecular forces present in propionitrile
Q: Which of the following statements about ideal gases is INCORRECT? Select one: a.Ideal gases…
A:
Q: Provide a name for the following heterocycles.
A: The name for the given heterocycles are given below. Pyrrole, furan and thiophene are aromatic in…
Q: Given K+ values: NiY2 = 4.2 x 10¹8, FeY2 = 2.1 x 10¹4, CoY2 = 2.0 x 10¹6. In the separate…
A: Complexometric titration is used for the determination of a mixture of different metal ions in…
Q: The vapor pressures of pure benzene and pure toluene at 20 ºC are 76 torr and 21 torr, respectively.…
A: Given, The vapor pressure of pure benzene = 76 torr. The vapor pressure of pure toluene = 21 torr…
Q: Br₂ FeBr3
A: Methyl (CH3) group has a +I (inductive effect) which affects the attack on the nucleophilic centre…
Q: 211 values from your textbook. TOP the reaction shown below using standard heat of formation Fe(s) +…
A:
Step by step
Solved in 2 steps with 2 images

- Under second-order conditions (strong base/nucleophile), SN2 and E2 reactions may occur simultaneously and compete witheach other. Show what products might be expected from the reaction of 2-bromo-3-methylbutane (a moderately hindered 2°alkyl halide) with sodium ethoxide.Indicate the complete mechanisms for each step of the synthesis below.Illustrate detailed mechanisms and draw Product Z that could be obtained from the reaction below.
- write out a complete mechanism for the reaction below1. Discuss the role of the Aldol condensation reaction in the synthesis below. What specific reaction was used in the synthesis? What is the importance of the aldol reaction in the entire synthetic approach? 2. Show the detailed reaction mechanism involved in their corresponding specific parts in this syntheses.Please describe the difference in the outcome of treating a terminal alkyne with HgSO4, H2SO4, H2O and with 9-BBN, followed by H2O2 and NaOH.
- Please provide a mechanism with curved arrows by replacing MgBr with 3-methoxyphenylmagnesium bromide and the final product should be 3-methoxybenzoic acidProvide the synthetic route for the following reaction? (CH3)2CHCH2Br ----> (CH3)2CHCH2CH2CH2NH2Devise a detailed mechanism for formation of the major product of the elimination reaction below.
- Propose a reasonable mechanism for dihydroxylation with oxone (you may start with dimethyldioxirane, DMDO) that would lead to formation of a mixture of the cis- and trans-diols.What is the main product from the reaction sequence below?Fill in the missing reagent/product(s) for the following reactions.

