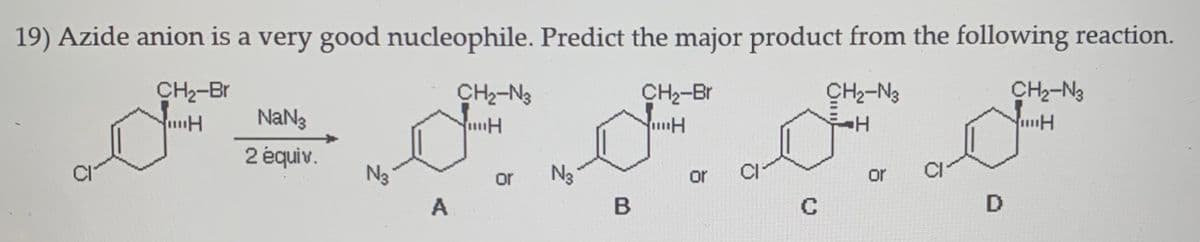
19) Azide anion is a very good nucleophile. Predict the major product from the following reaction. CH2-Br CH2-N3 CH-Br CH2-N3 CH2-N3 NaNg 2 équiv. N3 or N3 or or C
Q: Which is a better nucleophile? a. Br or C in H20 b. Br or CI in DMSO c. CH3O or CH;OH in H20 d. CH30…
A:
Q: 7. Which nucleophile would you expect to add reversibly to an a,ß-unsaturated carbonyl? а. NaBHa b.…
A: Addition of a nucleophile to an α,β-unsaturated carbonyl compound Takes place in two ways; one is…
Q: Show which compound can act as a nucleophile: (A) (В) H H. H (D) H-C-H (C) H -N- H H -m
A: Introduction: Nucleophile means nucleus loving. That means nucleophile are the molecules which are…
Q: Which molecule can loose a hydrogen and become a nucleophile? Select one: a. H3C– ECH b. H;C- -CH3…
A: The terminal alkynes or 1-alkynes are weak acids and loose a hydrogen when treated with strong base…
Q: 5:03 LTE O 9. Benzene is not a very reactive nucleophile and must be provided with a strong…
A:
Q: 1. Show how each of the following compounds could be prepared in one step using an Sy reaction.…
A: The Sn2 reaction takes place in one step and hence the halide must be primary
Q: Which of the following anions is the most nucleophilic in polar protic solvents? а. F- b. Cl- С. Br-…
A:
Q: 46. The Strength of Nucleophile: Which one is the strongest nucleophile? (A) OH (B) -CH3 -CH3 (D)…
A: As we go to down the group, size increases, basicity decreases and Nucleophilicity increases. C…
Q: Which structure is the best nucleophile for an SN² reaction? A. NH3 B. NH2 C. NH4* D. HCN O B
A: The question is based on the concept of reactive intermediates. We have to identify which is the…
Q: Which nucleophile in each pair which would give the faster SN2 rate? (a) (CH3)2S or (CH3)2Se (b)…
A: SN2 is the Nucleophilic Substitution Bimolecular reaction. The nucleophile that attacks the…
Q: In an SN2 reaction, which of the following nucleophiles will produce an ether? "C=CH "OH "O-C-CH3…
A:
Q: Which of the following are not examples of nucleophiles (1) F (2) H* (3) H2O (4) NH3 (5) Grignard…
A:
Q: Which member of each pair is a better nucleophile in methanol? a. H2O or HO- b. NH3 or H2O c. H2O or…
A: Any atom with a negative charge is a stronger base and a better nucleophile than the same atom but…
Q: The SN2 reaction proceeds with 100% inversion of configuration as a consequence of backside attack…
A: SN2 - Nucleophilic substitution bimolecular reaction. In SN2 reaction the rate of the reaction…
Q: Which of the following cannot react as a nucleophile? I. CH3NH2 II. (CH3)2N III. (CH3)3N IV.…
A: Nucleophiles: A nucleophile is defined as a substance that donates the electron pair to…
Q: Reaction A Reaction B Reaction C Br Br Br 1 2 3 Br2, FeBr3 NBS Br2, A
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Q: Which of the following is the least reactive towards a nucleophile? O C A Н3С о A Н Н С O B CH3 C…
A:
Q: Rank the following alkyl chlorides below in terms of increasing reactivity with a nucleophile via…
A: We have to arrange alkyl halides in order of increasing reactivity in SN2 reaction.
Q: Which of the following species is likely to be a nucleophile and which an electrophile? (a) CH3CI…
A: Electrophile is species which loves negative charge and nucleophilehile is species which loves…
Q: 7. For each of the following pairs of species, encirele the stronger nucleophile in acetone? (a)…
A: Since the question you have posted contains many subpart so I am solving first three for you.If you…
Q: Same reaction as in Q1. H3PO4 Identify the nucleophile: H2PO4t- H20 H30* H3PO4 А В D E O A O B O E
A: The nucleophile in the given reaction = ?
Q: SN2 Reaction lodide ion is an effective nucleophile in S2 displacements. In acetone solution, other…
A: Above reaction is 2° R-X.
Q: In the presence of a nucleophile, which of the following will most likely undergo conjugate…
A:
Q: Which of the two is the stronger nucleophile? Select one: a CH3OH b. CH3O Which is a polar aprotic…
A:
Q: A nucleophilic substitution reaction is shown below. Identify the nucleophile. I A) CH:S- B) Na II…
A: Nucleophile: In a chemical reaction a nucleophile is a species that forms bonds with electrophiles…
Q: Which is the correct order of reactivity towards a nucleophile, from most reactive to least? H3CO F…
A: Given compounds:
Q: Arrange the following ketones in order of increasing reactivity towards nucleophiles with the least…
A: The carbonyl compounds give nucleophilic addition reactions. The carbon atom of the carbonyl carbon…
Q: For each of the following pairs of species, which is the stronger nucleophile in acetone? Explain.…
A: Stronger nucleophiles are :
Q: Rank the following alkyl halides in order of increasing reactivity in an E2 reaction. Be sure to…
A: E2 reaction : E2 reactions are typically seen with secondary and tertiary alkyl halides, but a…
Q: Which of the following is the best (strongest) nucleophile? NH3 H20 NH2 CH30 II II IV Select one:…
A:
Q: Identify the stronger nucleophile in each pair. a.NH3, −NH2 b.CH3NH2, CH3OH c.CH3CO2−, CH3CH2O−
A: -NH2 is a conjugate base of NH3. The amine-based compound is a stronger base than the alcohol-based…
Q: Select which molecule is the better nucleophile in the following pair: a). Br− or Cl− in H2O b).…
A:
Q: Rank the following species in each set from best nucleophile to poorest nucleophile.
A: A good nucleophile has a very good tendency to donate electrons.
Q: Which of the following haloalkanes undergoes the fastest SN2 reaction with NaN3 (nucleophile)? a)…
A:
Q: In an SN2 reaction, which of the following nucleophiles will produce an ether? O "C=CH HO. "O-C-CH3…
A:
Q: Which one of these provides the strongest nucleophile in an SN2 reaction? a) HC=N b) NaC=N c) NH3 d)…
A: The nucleophilicity of the above mentioned compounds depends on the extent of the dissociation of…
Q: Which of the listed reagents (in-sequence) will accomplish the following synthetic transformation? I…
A: Answer: This reaction is based on the chemical properties of alkene and alkyl halide.
Q: s the best choice of reagent to effect the following transformation? SCH3 ? ..... (A) NaSCH3 (B) 1.…
A: According to the question, we need to identify the correct reagent for the conversion.
Q: Identify the stronger nucleophile in each pair. a. NH3,−NH2 b. CH3NH2, CH3OH c. CH3CO2−, CH3CH2O−
A: Nucleophile : Molecule which easily donates electron pair are called nuclephile.
Q: Which of the following is the strongest nucleophile? CH3S OH- H20 CH3O¯ NH3
A: Nucleophiles A Nucleophile is a chemical species that donates an electron pair to form a…
Q: Which molecule can loose a hydrogen and become a nucleophile? Select one: a. H3C- ECH b. H3C- -CH3…
A: Solution:
Q: Which one of the following nucleophiles will give mainly direct (1,2) addition in its reaction with…
A: b) HBr : HBr is acid and it's released H+ ion this ion attack on oxygen lone pair and then Br- ion…
Q: A set of three nucleophilic displacement reactions is shown below: CH3 CH3 CH,CHCH,CH,CCH,CI CH3 CH3…
A: tertiary alkyl halide tends to undergo the SN1 faster than secondary or primary alkyl halide because…
Q: H-S-CH,CH3 (HINT: "S" is a nucleophile ) S-CH2CH3 -S-CH2CH3 C А ОН S-CH2CH3 B D HO CH3CH2-S Select…
A: Here S is a nucleophile,attack at carbon and remove at the oxygen site,so oxygen become -ve charge…
Q: What sequence of reactions will convert benzene to o-bromoethylbenzene ? Br Select one: А.…
A:
Q: What nucleophile would be required to effect the following reactions? (a) HO. Br (b) Br
A:
Q: Which is the least reactive towards nucleophilic acyl substitution? a) H₂C-C-Cl c) H₂C-C-NH₂ 1₂C-E-N…
A: Reactivity of the carbonyl compound depends on the electrophilicity of (C=O) and the leaving…
Q: Identify the nucleophile in the reaction below: ÇH3 C-OH ÇH3 ČH3 + 2 H20 ČH3 + H30 O CH3 I. III. H3O…
A: Answer : II. H2O
Q: Rank these substrates in order of increasing ability to undergo a nucleophilic substitution aromatic…
A:
Q: Strong bases are good nucleophiles. Which reaction/s can occur on tertiary alkyl halides when a…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (d) Sodium cyanide in dimethyl sulfoxide (e) Sodium azide in aqueous ethanol (f) Sodium hydrogen sulfide in ethanolWhich species is likely to undergo a reaction with the nucleophile readily? A. (ch3)3cl b. (Ch3)2HCI c. Ch3ch2cl d. CH3CI1. SN1 or SN2 2. Include major product and stereochemistry 3. Add detailed reaction mechanisms
- Select which molecule is the better nucleophile in the following pair: a). Br− or Cl− in H2O b). CH3O− or CH3OH in H2O c). HO− or HS− in H2O d). I− or Br− in H2OMatch the questions with the correct descriptions below: Sn2, E2 E1cb Sn1, E1 (anti) E2, E1cb Sn1, E1 2 steps poor leaving group 2 carbons removed from a carbonyl group carbanion intermediate 3°>allylic, benzylic>2°>1°>CH₃ carbocation intermediate Ist order reaction (r=k[RX]) H-X anti no intermediate 2nd order reaction (r=k[RX][Nu]) polar aprotic solventIf anhydrides react like acid chlorides with the nucleophiles, draw the products formed when each of the following nucleophiles reacts with benzoic anhydride [(C6H5CO)2O]: (a) CH3MgBr (2 equiv), then H2O; (b) LiAlH4, then H2O; (c) LiAlH[OC(CH3)3]3, then H2O.
- 1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic centerFor problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (g) Sodium methanethiolate (NaSCH3) in ethanolRank the following in increasing order of reactivity towards nucleophilic acyl substitutiona. CH3COCl, CH3COOCH3, CH3CONH2b. CH3COOCH3, CH3COOCH2CCl3, CH3COOCH(CF)3
- Pleas explain how this process occurs. Identify SN1, SN2, E2, E1, nucleophiles and electrophiles.Rank the following compounds in order of increasing SN2 reaction rate with a good nucleophile. Explain yourreasoning. (CH3)2CHBr (CH3)3CBr CH3CH2Br CH3CH2IConsider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:

