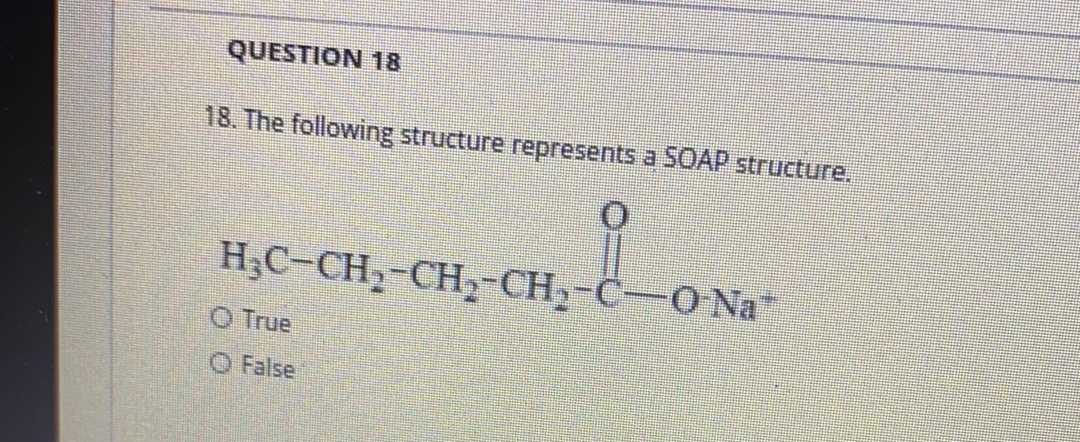
19. The following structure represents a SOAP structure. H;C-CH2-CH2-CH2-CH2-CH2-CH,-CH,-CH,-CH;-CH2- O-K+ O True O False
Q: 4. The following rules on aromaticity promotes a planar structure of nitrogenous bases EXCEPT: A.…
A: DNA and RNA are nucleic acids composed of a nitrogenous base, sugar molecule, and phosphate groups.…
Q: An alcohol can react with an aldehyde to form a a. Hemiketal b. O b. Peptide bond C. Carboxylic acid…
A: Introduction: The correct choice is option d. Hemiacetal The correct choice is option c. Oxidation
Q: 3. On the DIPEPTIDE, label the following: Two central carbons, N-term, C-term, two Rgroups A…
A: Amino acids are the building blocks of proteins where several amino acids are linked together by…
Q: 4. Which of the following structure is a B-D-glucose? * CH2OH ОН ОН Н H. ОН Option 1 CH2OH H. ОН Н 1…
A: Glucose is a monosaccharide molecule and is the most abundant sugar of the carbohydrate family. The…
Q: 1. The following structure IS SOLUBLE in water. CH3 H3C O True O False
A: An ester is a chemical compound which is derived from acid in which at least one -Oh group is…
Q: 5. In a portion of polypeptide shown below, which bond is described by o O0 0? А. 1 В. 2 С.3 D. 4 E.…
A: A polypeptide chain is a linear chain of many amino acids.
Q: Identify the chirality center (sometimes called chiral atom) in each molecule. If the molecule does…
A: A chiral center is defined as an atom in a molecule that is bonded to four different chemical…
Q: 1. Which are diastereomers CHO and сно CHO and CHO H• OH но H- H OH OH но- H- OH но CH;OH CH,OH…
A: Diastereomers are stereoisomers that are non-identical or non-mirror images whereas enantiomers are…
Q: 12. Identify the zwitterions in the list of substances (a) (CH3);NH(CH,);N_ (b)…
A: A zwitterion is a compound that contains both positive and negative charge areas in different…
Q: 1. Complete the given table below. What properties do each of the following R groups have?…
A: Basic-Positively charged, alcohol group Acidic - Negatively charged Neutral - No net charge Polar -…
Q: 5) Decide on the most likely mode of denaturation for each of the following compounds... a) hexane…
A: Denaturation is the process of the breaking the linkages or bonds that are formed in the compounds…
Q: ◆ Arginine Phenylalanine ◆ Cysteine ◆ Serine Glutamic acid ◆ Glutamine ◆ Isoleucine Leucine A.…
A: There are 20 naturally occurring amino acids. In a polypeptide chain, the alpha amino and the alpha…
Q: 4) The result above occurred because the potassium permanganate crystals have a weight than the…
A: As per our company's guidelines we are supposed to answer a single at a time, please repost the rest…
Q: NH O purine O pyridine H. Consider compound 2. Compound 2 is classified as a purine NH pyrimidine…
A: Introduction: Compound one is pyrimidine. (Uracil) Compound two is a purine. (Guanine) Compound…
Q: 5. Which of the following structure is the not stable structure of hexoses? 6 Он ОН4 HO HO. 5 HO- 2…
A: In the hexose structure or any structure when the position of OH group is in equatorial postion it…
Q: sub= 18 help
A: The reaction given in the question involves the conversion of dihydroxyacetone phosphate to…
Q: Which of the following sub
A: To assign " R " and "S " configurations to chiral carbons in optically acive compound , Cohn…
Q: _7. Which of the following FA has the lowest melting point? A. Stearic acid B. Palmitic acid C.…
A: A fatty acid is a molecule that is composed of a long hydrocarbon chain attached to the carboxylic…
Q: 7. The only carbohydrate which is not having any chiral carbon atom is (in 1 to 3 sentences, explain…
A: Carbohydrates are essential biomolecules which are also sugar molecules. Carbohydrates, along with…
Q: 2. Identify the following bonds as ionic or covalent. a) NaF b) MgCl2 c) C6H1206 d) NO2 e) CaF2 f)…
A: a )= Na+-F- b)= Mg2+ - Cl- = MgCl2 C)= d)= O - N -O = NO2 e)= F- - Ca2+ -F- f) =Li +- F-
Q: 2. Draw the structure of the tripeptide Gly-Ala-Tyr H H. N-C-C H N-C-C + N-C-C + H он он CH3 H. 0-H…
A: Proteins have four levels of structural organization including Primary, secondary,…
Q: 2. Which of the following forms of lysine would you expect to predominate at low pH, neutral pH, and…
A: Lysine is a positively charged polar amino acid with ε-amino group as the functional group. Amino…
Q: Converts to iso-parafFins Isomerization Alkylation O polymerization @ none o 1. para ffins
A: There are a number of reactions taking place within a cell. In order to understand the complexity of…
Q: Which of the following does NOT contain 1,4 glycosidic linkage? వార్ CH,OH CH,OH CH;OH CHOH о он HO…
A: Glycosidic linkage refers to C-O-C linkage between two carbon atoms of different monosaccharide…
Q: 1. Draw the condensed structural formulas for A and B. [0] [0] CH3 A B CH,-C I-CH,-C-OH
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: Monomer 1 Monomer 2 ОН НО- Monomers linked by covalent bond H,0 Monomer 1 Monomer 2 Monomers linked…
A: Biological macromolecules are crucial cellular components that execute a variety of functions that…
Q: CH3 me Left VVilaL Lype oI ISOmer is snown belUW: CHBR, CH, c=C C=C H. H. CHBR, H. cis-trans these…
A: First, let's discuss the term isomer. The isomers are the compounds which have same molecular…
Q: 1. Choose the formula for threonine: a) CH₂ -CH-COOH OH NH₂ b) CH3 -CH-CH-COOH OHNH,
A: Amino acids are essential for the growth and development of the body. Amino acids are considered to…
Q: VII. Identify the functional groups present. No need to name the compounds. H нн нн H H…
A: Functional groups: These are the atoms or group of atoms attached with the chemical compounds and…
Q: The C-C bond angles for the ring in M2 is: 60º 109.5º 120º 180º
A: Asked : C-C bond angles for the ring in M2
Q: A solution in which ["OH] = 6.3 × 10-/ M has a higher pH than a solution with a [-OH] = 4.3 x 10-2…
A: Acids are substances that can release an H+ ion. Bases are substances that accept an H+ ion. There…
Q: 3. Identify the monosaccharide represented by each of the following structures. Name each compound H…
A: Monosaccharide or simple sugars are the most basic unit of carbohydrates that can not be hydrolyzed…
Q: 4. Which of the following would be considered a basic solution? A. Solution with [OH-] = 3.8 x 10 M…
A: We can find out this using formula pH + pOH = 14 pOH = -log[OH-] and pH = -log [H+]
Q: II. Arrange the following compounds in order of increasing: A. boiling point B. acidity C.…
A:
Q: 1. Classify each pair of of structures as enantiomer, diastereomers or neither enantiomers or…
A: Hi! Thank you for posting the question. As, you have not specified with which question you need help…
Q: 2. Write the formula for the conjugate acid for each of the following bases: a) CO3- b) H:O c) H:POr
A: A conjugate acid–base pair, according to the Bronsted–Lowry definition of acids and bases, consists…
Q: your hospital pharmacy makes a fortified tobramycin eyedrops that is 13.6% in a 10 ml bottle. How…
A: The ratio of mass of solute present in a solution relative to the volume of the solution represents…
Q: 4. Determine if the following compound is chiral or not. OH Но. HO Me ZOH ZoH HO HO, OH Ме CI CI OH…
A: The parameters based on which chirality can be determined are: 1. Visualise the mirror image of the…
Q: Titration of alanine by a strong base, for example NaOH, reveals two pk's. Which is the titration…
A: A titration curve of an amino acid is the plot of the pH of the amino acid solution versus the…
Q: 2. Which of the following is an aldotriose? (in 1 to 3 sentences, explain why this is your answer).…
A: Sugars are referred to as saccharides and are the generic term that refer to carbohydrates.…
Q: In the figure below, the bond labeled "X" is a (an) X bond H. Y bond
A: Q. A water molecule is formed when two atoms of hydrogen covalently bonded with one atom of oxygen .…
Q: 1. Write the formula for the conjugate base for each of the following acids: а) НСО- b) HРО c) HBRO
A: The absolute concentration of acid is required to keep the buffer's pH stable when new ions are…
Q: 5. (a) How many covalent bonds does each of the following type of atom typically form? (i) H (iii) C…
A: INTRODUCTION Covalent bond Whenever there is an equal share of electrons between the atoms, covalent…
Q: HO, 1 2 3 HO- 4 OH 5 H OH 6 `OH The structure has six hydroxyl functional groups The absolute…
A: Monosaccharides are the simplest form of carbohydrates. Monosaccharides are linked to form…
Q: 6. What functional groups are present in the following molecul HạN. OH NH. A) amine and carboxylic…
A: There are several functional groups that can be found in a molecule. Some examples of functional…
Q: How is it possible for soap to have a pH greater than 10? What substance used in soap making that…
A: The majority of the soaps have a pH within the range of 9-10. The majority of the shampoos have a pH…
Q: The following amine acids are given. SH CH3 *HạN-CH- ČH2 *H,N-CH- or Cysteine Alanine Draw…
A: Introduction Fatty acids These are long-chain hydrocarbons with carboxylic group end. Three fatty…
Q: Summary Chart Macromolecule Monomer Polymers Bonds Functional Groups Examples CH2OH CH2OH он -…
A: Macromolecules are the molecules of high molecular masses. These are composed numerous small…
Q: 9. An ester is treated with aqueous acid. What is this reaction called? A. Esterification B. Acid…
A: Esters are neutral, class of organic compounds that react with water and produces alcohols and…

Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- What type of material is mycolic acid? Question options: ethylene glycol nitrogenous polysaccharide waxy compoundA prescription asked for a 500 mL 12% dextrose solution. How would you prepare the prescription if the available dextrose solutions in the pharmacy are 10% and 20%?Chemical Biology, halpp
- Consider the given data (Solubility Tests, Solubility Group, Chemical Tests, and Functional Group) in the experiment. Based on the results of the solubility tests and chemical tests, what is the most probable structure of C4H6O2? Draw your answer. You may draw the compound using line-bond formula OR Lewis structure.BIOCHEMISTRY Why does salt compound give off light (or colored flame) when burned?another name for ion
- Result and Discussion: Pauly Reaction: Samples: tyrosine, alanine, histidine Reagents: Cold Saturated Sulfanilic Acid, Cold 1 % Sodium Nitrite (NaNO2) and 10% Sodium Bicarbonate (Na2CO3) - Mix 0.5 ml of cold saturated sulfanilic acid solution (HANDLE WITH CARE) with 0.25 ml of cold 1.0% NaNO2. Cool in ice with constant shaking for 3 minutes. Add 0.5 ml of the sample and make alkaline with 10% Na2CO3. Record your observation.A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, and phosphatidylcholine was applied to a hydrophobic interaction chromatography column. The column was then washed with increasingly nonpolar solvents. IWhat order would the lipids be eluted from the column?(the pic cut off but this is shown below) (answer full question)Select all the pH values at which the charged form of the R group would predominate.* 2.2* 12.4* 6.2* 10.5



