2. A sample of neon gas is contained in a bulb at 150 °C and 350 kPa. If the pressure drops to 103 kPa, find the new temperature, in degrees Celsius. NOTE: in the video, temperatures are given on the Kelvin scale. If they had been given in degrees Celsius instead, you would have to convert them to the Kelvin scale -- Gay-Lussac's Law does NOT work if the temperatures are in degrees Celsius. P₁ = 350 kPa P2 = 103 kPa T1= 150°C T2=?
2. A sample of neon gas is contained in a bulb at 150 °C and 350 kPa. If the pressure drops to 103 kPa, find the new temperature, in degrees Celsius. NOTE: in the video, temperatures are given on the Kelvin scale. If they had been given in degrees Celsius instead, you would have to convert them to the Kelvin scale -- Gay-Lussac's Law does NOT work if the temperatures are in degrees Celsius. P₁ = 350 kPa P2 = 103 kPa T1= 150°C T2=?
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter3: Measurement And Chemical Calculations
Section: Chapter Questions
Problem 99E
Related questions
Question
Solve using gay-lussac’s law.
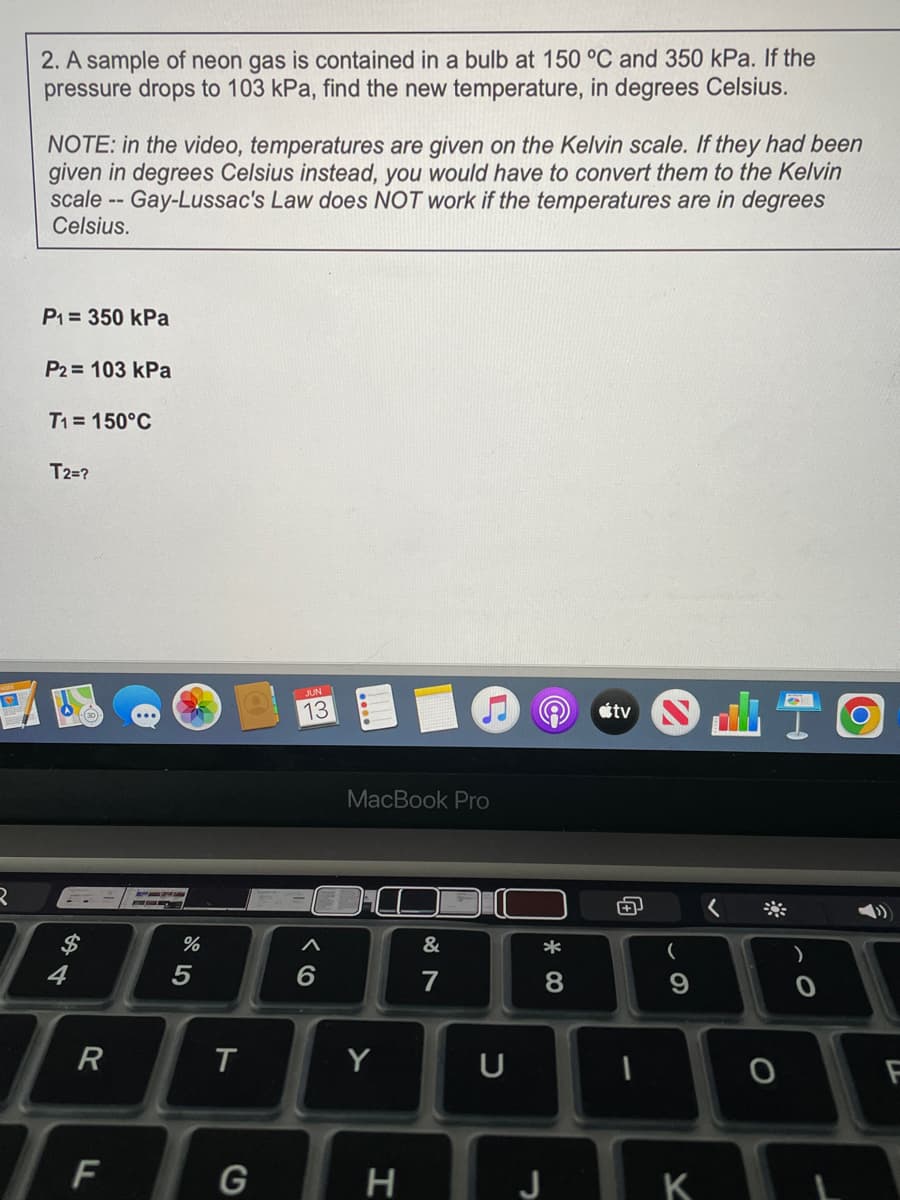
Transcribed Image Text:2. A sample of neon gas is contained in a bulb at 150 °C and 350 kPa. If the
pressure drops to 103 kPa, find the new temperature, in degrees Celsius.
NOTE: in the video, temperatures are given on the Kelvin scale. If they had been
given in degrees Celsius instead, you would have to convert them to the Kelvin
scale -- Gay-Lussac's Law does NOT work if the temperatures are in degrees
Celsius.
P₁ = 350 kPa
P2= 103 kPa
T1= 150°C
T2=?
tv
TO
MacBook Pro
FINE
R
J
<
%
&
*
(
AAAA666
4
5
7
8
9
R
T
Y
U
I
O
F
G
JUN
13
H
J
K
F
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning