2. A sketch of the phase diagram (not to scale) of water is given below. ETB ... Liquid F Solid Gas D Temperature) Which statement concerning the path (broken line) is false? a. Another way to vaporize all of the b. Once all the ice has melted (at the liquid water at point F is to increase the intersection of the broken line and line AB) the temperature of the water increases as heat is added until point F pressure. is reached. c. At point E the water is all solid (ice). d. If more heat is added upon reaching point F, the temperature will remain constant as the liquid water vaporizes. Pre ssu re
2. A sketch of the phase diagram (not to scale) of water is given below. ETB ... Liquid F Solid Gas D Temperature) Which statement concerning the path (broken line) is false? a. Another way to vaporize all of the b. Once all the ice has melted (at the liquid water at point F is to increase the intersection of the broken line and line AB) the temperature of the water increases as heat is added until point F pressure. is reached. c. At point E the water is all solid (ice). d. If more heat is added upon reaching point F, the temperature will remain constant as the liquid water vaporizes. Pre ssu re
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter9: Liquids And Solids
Section: Chapter Questions
Problem 64QAP
Related questions
Question
Answer the attached photo.
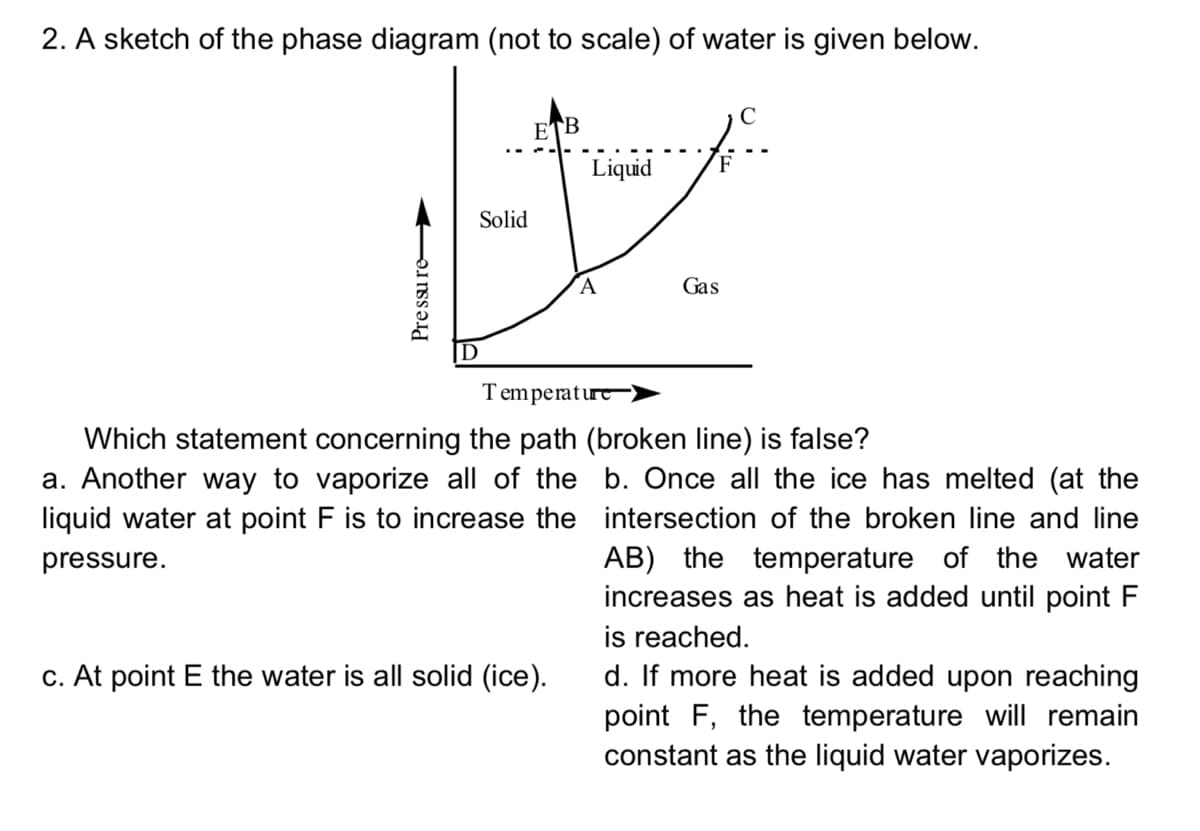
Transcribed Image Text:2. A sketch of the phase diagram (not to scale) of water is given below.
ETB
Liquid
Solid
Gas
Temperaturt
Which statement concerning the path (broken line) is false?
a. Another way to vaporize all of the b. Once all the ice has melted (at the
liquid water at point F is to increase the intersection of the broken line and line
AB) the temperature of the water
increases as heat is added until point
pressure.
is reached.
c. At point E the water is all solid (ice).
d. If more heat is added upon reaching
point F, the temperature will remain
constant as the liquid water vaporizes.
Pre ssu re
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning