2. Complete the chart below. You will need to look up Molecular Weights (MW), melting points (MP), boiling points (BP) and density. Keep in mind that liquids and solutions do not always have reported melting points (MP) or boiling points (BP). You do not need to find amounts, mmol or molar equivalence of 9-fluorenone. If the compound is a solid the boiling point (BP) and density (d) are often not reported. Molar equivalence is defined as the ratio of the moles of compound to the moles of the limiting reagent. MW MP Density Molar Compound Amount mmol BP (°C) (g/mol) C) (g/mL) Equivalence 9-Fluorenol 100 mg 6 ml Acetone 0.25 mL Glacial Acetic Acid 1.093 NaOCI 2 ml (5% wt/wt in H,0) 9-fluorenone 3. Calculate the theoretical yield for reaction based on the chart above and the reaction scheme at the top of this assignment.
2. Complete the chart below. You will need to look up Molecular Weights (MW), melting points (MP), boiling points (BP) and density. Keep in mind that liquids and solutions do not always have reported melting points (MP) or boiling points (BP). You do not need to find amounts, mmol or molar equivalence of 9-fluorenone. If the compound is a solid the boiling point (BP) and density (d) are often not reported. Molar equivalence is defined as the ratio of the moles of compound to the moles of the limiting reagent. MW MP Density Molar Compound Amount mmol BP (°C) (g/mol) C) (g/mL) Equivalence 9-Fluorenol 100 mg 6 ml Acetone 0.25 mL Glacial Acetic Acid 1.093 NaOCI 2 ml (5% wt/wt in H,0) 9-fluorenone 3. Calculate the theoretical yield for reaction based on the chart above and the reaction scheme at the top of this assignment.
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter7: Extraction
Section: Chapter Questions
Problem 7Q
Related questions
Question
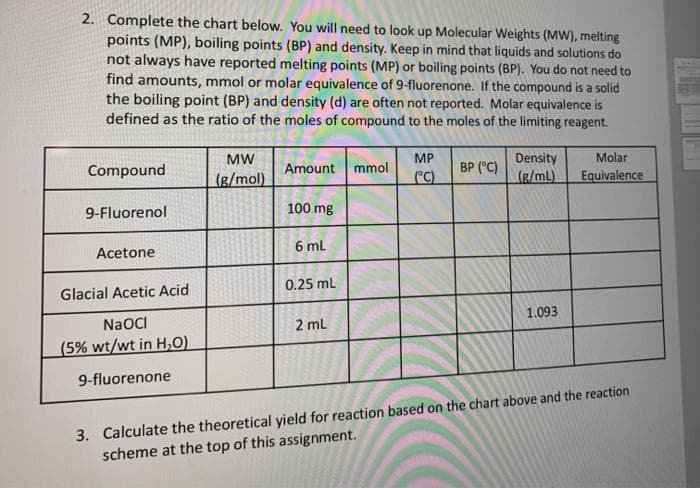
Transcribed Image Text:2. Complete the chart below. You will need to look up Molecular Weights (MW), melting
points (MP), boiling points (BP) and density. Keep in mind that liquids and solutions do
not always have reported melting points (MP) or boiling points (BP). You do not need to
find amounts, mmol or molar equivalence of 9-fluorenone. If the compound is a solid
the boiling point (BP) and density (d) are often not reported. Molar equivalence is
defined as the ratio of the moles of compound to the moles of the limiting reagent.
MW
MP
Density
Molar
Compound
Amount
mmol
BP (°C)
(g/mol)
°C)
(g/mL)
Equivalence
9-Fluorenol
100 mg
6 ml
Acetone
0.25 ml
Glacial Acetic Acid
1.093
NaOCI
2 ml
(5% wt/wt in H,0)
9-fluorenone
3. Calculate the theoretical yield for reaction based on the chart above and the reaction
scheme at the top of this assignment.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole