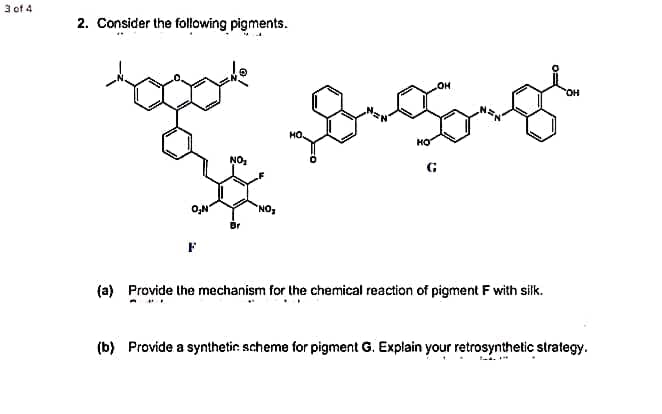
2. Consider the following pigments. OH HO NO G O,N NO (a) Provide the mechanism for the chemical reaction of pigment F with silk. (b) Provide a synthetic. scheme for pigment G. Explain your retrosynthetic strategy.
Q: Proteins, nucleic acids, lipids, and carbohydrates are based on which element? охудen carbon hydroge...
A: Proteins are the linear chains of amino acids attached together via peptide bonds, Carbohydrates are...
Q: "VDAATFKQANDNG" is the sequence of an a helix. Which of the following interactions is probable? (. d...
A: VDAATFKQANDNG In alpha-helix : H-bonding occurs b/w ith(CO group) and i+4th(NH group) amino acids.
Q: A group of researchers are interested in a question about the osmolarity of the cells of a particula...
A: It is assumed that a positive percent difference indicates that cell volume has increased. Water is ...
Q: What are the different types of solutions according to their concentration? What is its effect of ea...
A: Solutions: A solution is a homogenous mixture that contains a similar kind of substance (solute) di...
Q: Which of the following are true about aspirin
A: The question is all about the drug aspirin i.e acetylsalicylic acid that is used to reduce pain and ...
Q: 9) Propose a synthetic sequence for the dipeptide A-D-G (single-letter codes) starting from the free...
A: Hi! Since you have posted a multipart question , we will solve the first 3 subparts for you accordin...
Q: A solution has a hydrogen ion concentration of 0.01 mol/L. What is its pH? What is its hydroxide ion...
A: Given Values: [H+] = 0.01 Mol/L = 1×10-2 M pH is a measure of negative logarithm of hydrogen ion con...
Q: 6. Kwashiorkor is the discase causcd by a deficiency of proteins in the diet that is adequate in cal...
A: Protein deficiency (in Kwashoirkor) and hence the lack of protein reduces the production of lipoprot...
Q: Cytosol Mitochondrion A. PATHWAY 1. major pathway of ethanol metabolism NAD* NADH + H* NAD* NADH + H...
A: In the human body, three steps are involved in alcohol metabolism. Ethanol is oxidized to acetaldeh...
Q: Describe in general terms the steps involved in creating transgenic plants
A: Transgenic plants have modified genome using genetic engineering techniques. This can be either addi...
Q: The aerobic oxidation of 2 FADH2 and 10 NADH in perspiratory chain reaction will produce. . *
A: NADH and FADH2 are the reducing equivalents produced in the glycolytic and the TCA cycle pathway. Th...
Q: How are blood glucose concentration affected by heavy alcohol consumption? Please explain with extre...
A: Alcohol plays major role on health. Alcohol can effect every organ of the body. In an average person...
Q: kinetically perfect enzymes a) Using the Gibbs energy formula, explain why engergonic reactions can ...
A: Most enzymes follow Michaelis -Menten (MM)kinetics and hence obeys the MM equilibrium assumption tha...
Q: Draw the Haworth projection of β-D-Altopyranose given the structure of D-Altrose.
A: Altrose is an aldohexose. Altrose is a C-3 epimer of mannose. The Fischer projection shows the open ...
Q: The disaccharide that gives two same monosaccharide units on hydrolysis is .. ...? .....
A:
Q: Explain how the carbonate-bicarbonate buffer system works in balancing acid-base in the blood.
A: Buffers are solutions that have weak acid and its conjugate base. They nullify small changes in the...
Q: Explain the importance of buffers and what are the main buffers in the body?
A: Almost all the biological processes are pH-dependent. A small change in the pH creates a drastic cha...
Q: 1. A short stretch of amino acid residues that connects consecutive strands of an antiparallel sheet...
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since yo...
Q: Describe the types of pathogens. Give an example of an obligate pathogen, a facultative saprophyte, ...
A: Microorganisms, often known as microbes, are microscopic organisms that can be found all over the pl...
Q: you have the following peptide Arg-Ile-Pro-Leu-Asp-Lys-Glu The net charge on this peptide at pH 7.0...
A: Amino acids are biomolecules that are comprised of two functional groups, these are an amino group (...
Q: do archaebacteria have autotroph or heterotroph nutrients
A: Nutrients are the components of food like carbohydrates, fats, vitamins, proteins, etc. These nutrie...
Q: What do pathogenesis related proteins and phytoalexins have in common? How do they differ?
A: Pathogenesis related (PR) protein – These proteins are produced by plant in the event of pathogen at...
Q: Well 1 contains 20 uL of serum. Wells 2-6 contain 90 uL of water. To do a serial dilution of 10% c...
A: 10% serial dilution is mixing 1 part of stock solution with 9 parts of diluent so that concentration...
Q: You are working in a bioengineering lab, and you are asked to create a peptide with a helix with len...
A: Proteins are the macromolecules composed of amino acids bound together by peptide bond between amino...
Q: Purification of a protein of unknown structure has been achieved. The natural protein has a molecula...
A: Proteins are polymers of amino acids and form primary structure. It can form secondary, tertiary and...
Q: Carbohydrate polymer that contains inter- and intra-sheet H-bonds is O Starch O Glycogen Blood Group...
A: The correct answer is option (a) Starch Explanation: Starch is composed of linear or branched glucos...
Q: What are the advantages of the presence of organelles in eukaryotic cells? explain
A: The eukaryotic cell has a nuclear membrane surrounding the nucleus inside which chromosomes are pres...
Q: One of the most common features of anion binding sites is the presence of an a-helical structure. Us...
A: Helical peptides have periodic and ordered structures in which every backbone dipole vector result f...
Q: Which of the following has the highest iodine content? Which of the following has the lowest meltin...
A: 1. Linoleic acid has the highest iodine content. 2. Unsaturated fatty acids have lower melting poin...
Q: proteins are deamidated in an aqueous solution to produce ammonium and aspartate, respectively, from...
A: Isoelectric focusing (IEF), also known as electrofocusing, is a technique for separating different m...
Q: b. What is happening in panel B? You are looking at the southern blot image. What is the radioactive...
A: This is an experimental data where experimenter is trying to look on function of PHT4;2 gene allele....
Q: 1. Would you say that indicator tests are qualitative or quantitative? Explain your answer.
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: In the figures of ferricrocin and enterobactin, circle the parts of the molecule that are b) respons...
A: Ferricroxin ,is a siderophore involved in intra and transcellular iron transporter in Aspergillus fu...
Q: 1) Describe what happens to a cell in a hypertonic solution. 2) Describe what happens to a cell in a...
A: 1) When the cell is put in hypertonic solution, the concentration of solutes in the solution is grea...
Q: Given the following question on the image identify the following:1. Total number of acetyl coA molec...
A: Maltose is a carbohydrate and it gets oxidized through glycolysis in cytoplasm and through the TCA c...
Q: is an example for aldotriose.
A: The question is about the functional group that out of given example which is the example of aldotri...
Q: Consider two enzymes catalyzing two reactions (A --> B --> C ) in a metabolic cascade with their pro...
A: Ans) The order of concentration of A, B, C are in order [C] > [B] > [A] Explanation: The abov...
Q: An antibody (Ab) can bìnd to its antigen binding site (B) with a high affinity (large negative AG°)....
A: The antibodies bind to its antigen via non n covalent interactions like hydrogen bonding, hydrophobi...
Q: A peptide has the following amino acid composition: 2 Met, 2 Phe, 2 Glu, 1 Arg, 1 Lys, 1 Val, 1 Leu,...
A: Small molecules called amino acids to form the building blocks of proteins. In chemistry, an amino a...
Q: Using Molisch's reagent, an unknown carbohydrate solution was found to be positive (+). Within three...
A: Molisch's test is done for detection of carbohydrate. Barfoed's test is used for detection of monosa...
Q: Distinguish among fats, phospholipids, and steroids, and describe the composition, characteristics, ...
A: Lipids are organic molecules that are insoluble in water and soluble in organic solvents. The three ...
Q: Write a short description on ALL of the following: a) The application of turbidostat against chemost...
A: In various assays and biological experiments, microoorganisms are allowed to grow in liquid cultures...
Q: You've isolated a novel protein, but you believe what you actually have is a mixture of the unmodifi...
A: Amino acids are biomolecules that are comprised of two functional groups, these are an amino group (...
Q: Propose one amino acid substitution that would disrupt the leucine zipper structure and explain why
A: Leucine zipper structure is formed by leucine residues and these leucine residues are held together ...
Q: Describe how the following properties affect the function of a protien: A.) R group orientation B.) ...
A: Amino acids are biomolecules that are comprised of two functional groups, these are an amino group (...
Q: Speculate on what the receptor sites for each of these molecules might be in terms of shape and pola...
A: Receptors are membrane proteins consisting of proteins, and glycans. Its extracellular domain contai...
Q: What are the main advantages of the presence of organelles in eukaryotic cells?
A: Cells are the basic units of life. Cells are broadly classified as prokaryotic and eukaryotic cells....
Q: 1. What is the chemical basis for a positive test in Molisch test? 2. What is the chemical basis for...
A: MOLISCH TEST:- chemical test used to check for the presence of carbohydrates in a given solutions. I...
Q: Why is biochemistry called the chemistry of life?
A: The question is all about the subject biochemistry that it is called as chemistry of life as our hum...
Q: For the equilibrium below, pKa1 = 7.9 and pKa2 = 10.1. At what pH is [HA-] equal to [A2-]? H2A ...
A: The pH of a solution is a measurement of its acidity or basicity. The pH scale ranges from 0 to 14. ...
Step by step
Solved in 2 steps

- 15. How much powder and water to make a 0.1 concentration of resazurin and please explain the reaction from resazurin to resorufin?1. Depict the structure features in the active site of cysteine protease 2. provide a step-wise reaction mechanism to show the hydrolysis of the peptide bond catalyzed by a cysteine protease1. How does the resin facilitate purification in glutathione S-tranferase (GST) affinity? 2. Does protein stick to the resin in glutathione S-tranferase (GST) affinity? If so how do you get it off? 3. Explain how protein of interest would be purified in glutathione S-tranferase (GST) affinity?
- 1. Know the structures of DNP-glycine and of the chromophore associated with cytochrome C. Low Salt Volume of blue band = 5.2 ml (Void Volume) Volume of yellow band = 2.6ml Volume of red band = 11ml High Salt Volume of blue band = 4.2 ml -> Void Volume Volume of yellow band = 5.2 Volume of red band = 1.2 2. What is the exclusion limit and the fractionation range for CM-Sephadex resin used? 3. What are the ion exchange reactions which occur when the coloured mixture is first applied to the column under low salt conditions? What exchange reactions occur upon the change of buffer from low salt to high salt concentrations? 4. What were the values for Vo and Vi for this column? Where the values for Vo and Vi the same under both conditions? 5. Explain the order in which the various components of the mixture elute under each condition in terms of their physical properties and the nature of the CMSephadex G-50 column. Would the same order be observed with a G-50 Sephadex column (i.e. no…1. provide three reasons why most of the research on carotenoids concentrates on b-carotene. 2. draw the retinol ester that has stearic acid as the carboxylic acid portion of its structure.5 25) Although the UV280 extinction coefficient of BSA is well known, why was the BCA assay employed to measure protein concentration of the folate labeled BSA? Please provide an explanation. (C) Would the use of reducing agents have an affect on the tertiary structure of BSA—please explain your answer (you will need to investigate the structure of BSA beyond this article)?
- 1b. Indicate an important difference between polyethylene produced using Ziegler-Natta polymerization compared to normal addition by radical mechanism. 1c. Why secondary forces in polymers have a very significant effect on the overall properties of polymers?6 The following structure is part of the lipopolysaccharide found on the outside of vibrio cholerae. I know that the glycosidic linkage connecting residues B and C and the glycosidic linkage connecting residues C and D are both alpha, but what are the numbers that follow the alpha? Ex: alpha- ? -> ?6. If catalase is tested with the biuret reagent, what would be the ideal result? Why?
- 1)Since bromelain is an endopeptidase, what types of bonds does it work on? 2)Since bromelain is an endopeptidase, what types of bonds does it work on?describe a detailed experimental procedure for the chemical synthesis of proteins with the α-ketoacid hydroxylamine (KAHA ligation), using (S)-5-oxaproline (Opr) as a key building block.1. Glycosphingolipids function via cis regulation and trans recognition. Explain these terms and provide examples of each.

