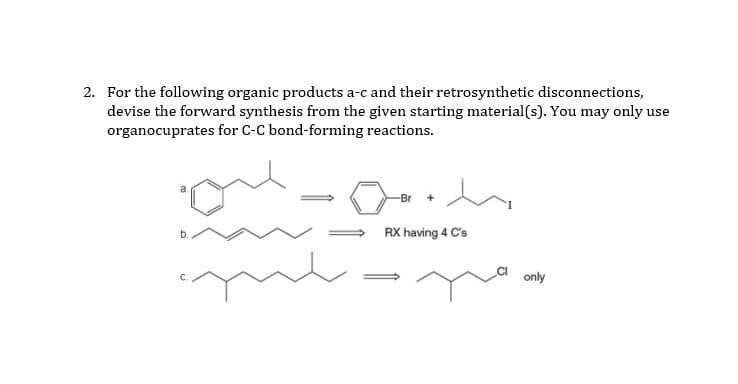
2. For the following organic products a-c and their retrosynthetic disconnections, devise the forward synthesis from the given starting material(s). You may only use organocuprates for C-C bond-forming reactions. RX having 4 C's only
Q: a) Briefly explain the reason by showing what kind of reaction mechanism the main product (major)…
A: Given: We have to predict major and minor products for the following given substrates.
Q: 1) Write a detailed, stepwise mechanism for the following reaction. In your mechanism, provide three…
A:
Q: Migratory aptitude is defined as the relative ability of a group to migrate in a rearrangement…
A:
Q: The reaction below produces two neutral organic products that are formed in a 1:1 ratio. Provide the…
A: Nucleophilic substitution reactions are those reactions in which nucleophile attacks the alkyl…
Q: 1. The dramatic reaction below was reported in the Evans synthesis of colchicine in 1981. MeO CO₂Me…
A: Note : Acid catalyzed electrophilic aromatic substitution followed by dehydration. ( see below).
Q: 1) (10 points) Draw the major organic product generated in the reaction below. Pay particular…
A:
Q: Part A Give the solvolysis (SN1) products expected when the following compound is heated in ethanol:…
A:
Q: With 2° or 3° alkyl groups bonded to the ether oxygen, the C—O bondis cleaved by an SN1 mechanism…
A: The replacement of any atom or group of atoms by a nucleophile is known as nucleophilic substitution…
Q: Designing Synthetic Schemes with Aromatic Rings: For the reaction below, begin with benzene and…
A:
Q: 2. Strychnine (6) is a notorious poison for both humans and rats. The first synthesis of this…
A:
Q: Consider the reactions below and the pool of choices. عيله میلی BAT HO Reagent 2 MAT Alkene 1 SAT…
A:
Q: (i) Provide the product for the reaction below. (ii) How would you know if the reaction was an SN1…
A:
Q: 2. Above is S-adenosylmethionine (SAM). Note that R'-S-R is a very good leaving group. You do not…
A:
Q: Circle the electrophilic and nucleophilic atoms in each substitution reaction below. Provide the…
A:
Q: For the reaction below: BH3 THF (solvent) a Draw the structure of the major organic product.
A: The above given reaction shows the hydroboration of alkene. Here, borane and hydrogen gets attached…
Q: (i) Provide the product for the reaction below. (ii) How would you know if the reaction was an SN1…
A:
Q: 1. CH2-CH2 B 2. H30* Br2. Mg AIBr3 Et,0
A: The reaction sequence is as follows:
Q: Draw the major organic product generated in the reaction below. Pay particular attention to regio-…
A:
Q: 1-butene Reagents Reagents n-butylbromide sec-butylbromide
A:
Q: 2) Write a detailed mechanism for the following reaction: H2S04 CH;CH;C=CH + H,O CH;CH¿CCH3
A:
Q: 1. Draw the MAJOR organic product obtained in the following reactions. Be sure to include…
A: We have to draw product for the following given reactions as follows in step 2:
Q: Account for complete retention of configuration in the solvolysis of (1).
A:
Q: SN2 Reaction lodide ion is an effective nucleophile in S2 displacements. In acetone solution, other…
A: Above reaction is 2° R-X.
Q: This substituent deactivates the benzene ring towards electrophilic substitution but directs the…
A:
Q: Propose mechanisms compatible with 1) and 2) of the following reactions.
A: Since we know that HBr adds with alkene by ionic mechanism in absence of peroxide so Markovnikov's…
Q: When drawing organic chemistry mechanisms with NaOCH3, do we automatically dissociate it into Na+…
A:
Q: 2. For the following organic products a-c and their retrosynthetic disconnections, devise the…
A: Retrosynthesis is given we have to give reagents for the forward synthesis. Organocopper reagents…
Q: 2) Write a detailed mechanism for the following reaction: H2SO4 CH;CH,C=CH + H2O CH;CH,CCH3
A: Hydration reaction of Alkyne takes place in acidic medium as follows:
Q: Provide a plausible arrow pushing mechanism for the reaction below. You may abbreviate the…
A: This is an example of Diels-Alder reaction followed by cyclization and aromatization.
Q: 2. Propose a complete and accurate arrow pushing mechanism for the reaction below. HNO3 H2SO4 O2N 3.…
A: Nitration: It is the chemical reaction in which there will be the addition of nitro group (NO2) to…
Q: 2) After each reaction arrow, draw the structure of the expected major organic product(s). (A) 1)…
A: Applying concept of wittig reaction
Q: Provide the mechanism for the Wittig reaction below.* H3C H3C- PPh3 CH3
A: In organic chemistry, an aldehyde or ketone when react with wittig reagent ( phosphorus ylide) gives…
Q: 2) Synthesizing the target molecule (below), it is suggested to start from the epoxide and benzene…
A: Reaction 1 is an acid-catalyzed reaction.
Q: Which one of the following is not a mechanistic step in the hydrolysis of benzamide? -NH + :NH3 -OH…
A:
Q: 1.1 Consider the following Friedel-Craft's reaction of the type shown in page 438, Fig 11.3.,…
A: Let us study the mechanism.
Q: 4. Provide a detailed, stepwise mechanism for the reaction below. (CH3)2CHCH2CH2CH21 + CN- →…
A: Primary alkyl halide reacts with cyanide ion to form alkyl cyanide. The reaction follows the SN2…
Q: 5. Retrosynthesis. Suggest how the molecules mentioned below can be synthesized (give the reagents…
A:
Q: 4) Provide the major organic product(s) in the reaction below. Br/light
A: This reaction proceeds through free radical mechanism.
Q: 2. Draw out the forward synthesis (fill in the boxes) only for the following transformation (it'll…
A: Here we have to make a desired forward synthesis for below conversion .
Q: - Provide the structure of the major organic product for the reaction sequence shown below. 1.…
A: It is a 3 step reaction as shown in the following step.
Q: 5) Provide detaled mechanisans for the following conversions that include all important resonance…
A: Reaction of cyclic ester with excess of Grignard reagent.
Q: 6. Provide a total synthesis for the target molecule starting from benzene using any reagents…
A: (a) When acetone is treated with phenyl magnesium Bromide (Grignard reagent) then 2-phenyl -2-…
Q: Provide a plausible arrow pushing mechanism for the reaction below. You may abbreviate the…
A:
Q: 66) Which of the following statements is false? Reactions following Markonikov’s Rule, form the…
A:
Q: Design a synthesis of 1,2-epoxypentane (drawn in problem 3 above) starting with 1-pentanol,…
A: Synthesis of 1,2-epoxypentane from pentanol.
Q: Based on the retrosynthesis above, provide a synthesis of the starting aldehyde from acetylene: H.…
A: The reaction will proceed in the forward direction if it reacts with a reagent and crosses the…
Q: CH3 CH2BR NBS hv
A:
Q: Br, Br HBr (xs)
A: SN1 type mechanism follow.
Q: a) b) Br de c) CI NaOCH3 d) F
A: We have to write the products for (A-I). Label products with the mechanism (Sn2, Sn1, E1, E2) that…
Please help answer the attached question....thank you!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- D10) The above synthesis was designed using the Organic Chemistry Roadmaps in the appendix of your textbook. In this synthesis, reagents from the table are used to carry out the indicated steps (shown in blue). In the box below, draw the structure of compound c.Nitrating methyl benzoate. 1.Describe the mechanism of EAS clearly and in detail 2. What is the effect of subtituent of the outcome of EAS;include 3 classes of subtituentsOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…A) Provide a reasonable multistep synthesis of the following molecule from the indicated starting material, using any reagents necessary. Please include both a retrosynthesis and a forward synthesis (that includes the product and reagents of each individual step in your synthesis). B) If you started with 1 mole of starting material and each step of your synthesis gave 100% yield, how many moles of the product drawn would you end with?1. Draw a reasonable arrow-pushing mechanism for the transformation shown along. 2. Identify nucleophiles and electrophiles 3. Name any type of reactions taking place like E2 or E1 4. Account for any regio- or stereoselectivity
- Choose one from each parenthesis. Based on the reactions below, the probable structure of alkene 1 is (BAT / MAT / SAT / FAT) while the process using Reagent1 is (reaction via acidic KMnO4 / reaction via cold, dilute, neutral KMnO4 / Ozonolysis) and that of Reagent2 is (reaction via acidic KMnO4 / reaction via cold, dilute, neutral KMnO4 / Ozonolysis).Provide a step-wise synthesis for the following: image 1 The last step of the synthesis needs to be a nucleophilic aromatic substitution reaction with a secondary amine.Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the help

