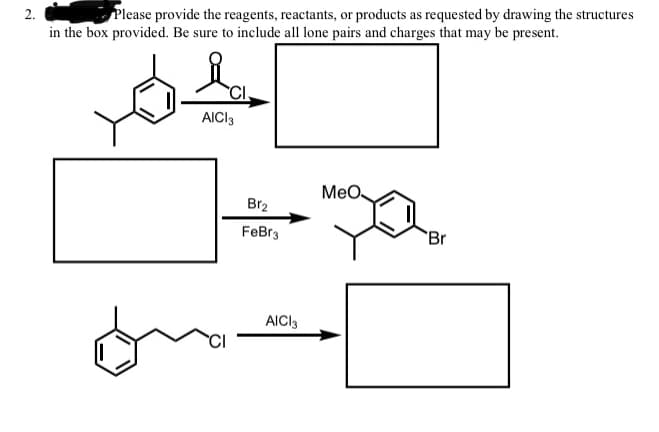
2. Please provide the reagents, reactants, or products as requested by drawing the structures in the box provided. Be sure to include all lone pairs and charges that may be present. AICI3 MeO. Br2 FeBr3 Br AICI3
Q: 2 of 2 Part B nstants 1 Periodic Table Draw the structure of 4-bromo-2-hexyne drocarbons. Alkenes n…
A: Organic chemistryHere we have to drawn the structure of an organic molecule, given is the IUPAC…
Q: . CH3 C CH2CH2-OH + CH;CH2C-OH >
A: The major functionality present in both the reactants which take part in the reaction are hydroxy…
Q: N' NBS N' NBS CN- N' NBS C N- Br2 FeBr3
A: We have to explain if each of the reactions can happen or not. If the reaction can happen, explain…
Q: 1. Provide the IUPAC names of the following structures NO2 Br NO2 C. b.
A:
Q: Which bond is non-polar in the molecule of trans-1,4-dichlorobutene-2?
A:
Q: Complete and balance the equation below: (Use curve arrows to predict the product) NaNH2 CH3C=CH
A:
Q: Which compound has the shortest carbon-carbon bond(s)? Clearly explain the reason, why? ) Нехаne II)…
A: A) In hydrocarbon the C-C bond length is depend on the hybridization of carbon which are bonding…
Q: Question attached
A: The organic conversions are used to convert one compound into other compounds. They also guide about…
Q: Question attached
A: Introduction: Molecular structure shows the arrangement of atoms in a space. The organic structures…
Q: how do i determine degree of unsaturation for an equation if it includes elements such as N, O, F.…
A: Degree of Unsaturation:Degree of Unsaturation is based on the ring and multiple bond in a compound.…
Q: Consider the reaction scheme below. Br2 FeBr3 CI AICI3 NaOH Y warm What are the structures of X, Y,…
A: When benzene is treated with Bromine in presence of Ferric bromide, then bromo benzene (X) is…
Q: Complete the reaction map by providing the answer from A-E. Write the IUPAC name of the products…
A: The correct answer is given below
Q: Which structures have the correct IUPAC names? * CH3 CH3 .Cl Br Br I. 2-bromotoluene II. para…
A: First structure is correct. When benzene is attached with -CH3 group , it is known as Toluene and…
Q: The order of increasing priority to select principal functional group (in IUPAC nomenclature) is -CN…
A: The group of atoms in the molecule which have their individual property than the other atom in the…
Q: е. CH3 (S) CH3C CHCH3 + HBr | | H3C OH
A:
Q: 2-chloro-3,3-dimethylhexane
A:
Q: The alkyl group (CH3)2CH- is a(n). group. (a) tert-butyl (b) iso-butyl (c) n-butyl (d) sec-butyl (e)…
A: Two compounds having similar molecular formula but distinct structural/bonding arrangements of atoms…
Q: label alpha carbon, beta carbon, leaving group, primary secondary, tertiary, typea of functional…
A: When 1 carbon is attached with carbocation is called 1° carbocation. When 2 carbon atom is attached…
Q: Please help me please
A:
Q: 3-hexyne + 1 mole of Br2 → (c) CH3CH2CECCH2CH3 + H2O/H2SO4 → (d) stable structure
A: Alkyne in the presece of Br2 give addition reaction and in the presence of H2O / H2SO4 give…
Q: T III) For each molecule shown, drow an example of each type of isomer. If one type does not exist,…
A: Structural Isomers are compounds having same molecular formula but different structure. Types of…
Q: Correctly name the alkyne shown here. A) 1-butyne bromide B) 1-bromo-1-butyne C) 1-bromo-1-propyne…
A: Given compound: We have to write the name of the compound.
Q: Which structure does not represent 2-methylpentane? (a) (b) (c) (d) 1.
A: (a) In this compound you numbering from left side and given number on long chain carbon then you see…
Q: 5. Outline practical laboratory stops that could be used to synthesize the alkane shown as target…
A: Step 1: HBr addition to 2,5-dimethyl hex-2-ene. Step 2: Grignard generation (2-bromo-2,5-dimethyl…
Q: Please draw the line structures for the C8H17+ carbocations that have the carbon skeleton below…
A: The carbocation bonded to three alkanes is most stable .because it has more Alpha hydrogen due to…
Q: Which structures have the correct IUPAC names? CH3 CH3 Br Br I. 2-bromotoluene II. para methyl, meta…
A: All are Aromatic compound. I. Methylbenzene is called toluene. One bromine is present as…
Q: Complete the Following reactions with the proper structures ( C2(PCY3)2Ru=CHC6HS CH,C, 25C HN-CH3…
A:
Q: Please do them in it's provided structuralform
A: The reaction is an example of nucleophilic substitute reaction of 2- bromo butane in presence of aq.…
Q: Br CI NaOCH3 heat `NO2
A: The given reaction is an example of an aromatic nucleophilic substitution reaction.
Q: 2) Which out of following hydrocarbons will not react with Br2? Write reactions with br2 for all…
A: IUPAC nomenclature is defined for compounds based on certain rules which are given by the IUPAC.…
Q: how do i name this? including r or s/ e and z if needed?
A:
Q: Q1. How would you sxatbesize the following substances starting from benzene? Draw all the reagents…
A:
Q: Hello, I want a simple explanation on this topic (Stability of cycloalkanes) Can you explain to me…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Follow the arrows and draw the product…
A:
Q: Introduction to Organic Chemistry I. Arrange the set of scrambled letters to form the required word.…
A: "Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: H Br NacN ethanol ? + NaN, ethanol H₂C-1 + H₂CH₂ CH₂ + NaCN CH₂-Br ? acetone ethanal ? ?
A: The all given reactions are example of the bimolecular nucleophilic substitution reaction (SN2) .…
Q: Circle the organic substances below which are aliphatic. `OH benzoic acid 5,6-dimethylnonan-1-ol…
A: Solution- (1). Non Aromatic is know as aliphatic. Therefore, in above given example option 2nd- (5,6…
Q: a T CH₂
A:
Q: ) Draw 2 sets of molecules. The first set should be constitutional isomers. The second set should…
A: Constitutional isomers are isomers that have different arrangements of substituents. Let's consider,…
Q: hat type of alkene is Compound X? (Use all reaction information to solve for Compound X's…
A: First find the double bond equaivalent (DOU) for X : molecular formula : C8H14 DOU = ((2C +2) -…
Q: Br2 ? How many moles of Br are required to completely halogenate the alkene? A. Three moles B. Four…
A: Here we have to write properties of addition of Bromine to the following alkene.
Q: a) Name the following molecules according to IUPAC: i) ii) iv) E S s
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Please complete the following reactions: Br2 CCl4 (a) CH=CH2 (b) CH=CH2 HBr (c) CH=CH2 Br2, H2O
A: Since you have posted question with multiple subparts as per guidelines we can solve only three . If…
Q: 2. What are the resonance structures for the following carbocation? What are the partial charges for…
A: Given carbocation is : Resonance structures of the given carbocation and resonance hybrid…
Q: Will there be a color change if KMnO4 is added to (a) Cyclohexane , (b) Cyclohexene, (c) Toluene ?…
A: Given data Cyclohexane + KMnO4 ——-> Cyclohexene + KMnO4 ——-> Toluene + KMnO4.…
Q: 1. Please, name the following compounds according to IUPAC rules. OCH3 OH b) c) HOOC .COOH СООН Br…
A: IUPAC nomenclature is used to determine the name of any compound in a world-wide acceptable manner.…
Q: Conversion of Compound into complete structure. A.CH3(CH2)8CH3 B. CH3(CH2)4OH C. CH3CCl3 D.…
A: The given compounds has to be converted into its complete structure.
Q: 2. Provide the missing curved electron arrows for the reaction below. нн H CH3 NaOH CH3 + H20 + NaBr…
A: The movement of electrons are shown by curved arrows. There are two types of arrows to show the…
Can someone help me please I just want to make sure my answer is right
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →Provide a name for the molecules belowhow do i determine degree of unsaturation for an equation if it includes elements such as N, O, F. when each of these are present in the formula do i subtract, add, or leave it the same from the amount of carbons present. example includes C6H7N or C4H7Br
- Consider the following hydrocarbons:I. butane II. methyl benzene III. octane IV. benzeneWhich of the following will produce a less soothy upon ignition?Answer all questions 1.What are the various ways by which alkenes may be synthesized?2.i. Give two examples each of Unsymmetrical alkenes and reagents.ii. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsI need to know the names of these two structurers for my notes but I am not sure how to name them.
- The compound whose structure is shown here is acetyl acetone. It exists in two forms:the enol form and the keto form The molecule reacts with OH–to form an anion, [CH3COCHCOCH3] (often abbreviatedacac–for acetylacetonate ion). One or the most interesting aspects of this anion is thatone or more of them can react with transition metal cations to give stable, highlycolored compounds (a) Are the keto and enol forms of acetylacetone resonance forms? Explain youranswer.(b) What is the hybridization or each atom (except H) in the enol form? What changesin hybridization occur when it is transformed into the keto form?(c) What are the electron-pair geometry and molecular geometry around each C atomin the keto and enol forms? What changes in geometry occur when the keto formchanges to the enol form?(d) Draw three possible resonance structures for the acac–ion.(e) Is cis-trans isomerism possible in either the enol or the keto form of acetylacetone?(f) Is the enol form of acetylacetone polar?…Consider the following aromatic compounds a para-substituted compound?, the most reactive towards EAS? an ortho-substituted compound? does not react with RX, FeX3?Please explain question 1 and 8 1. How many degrees of unsaturation are present in your unknown monosubstituted benzene starting material? 8. Based on all of the data provided, what is the identity of the “G” group? (Please NEATLY & CLEARLY draw its full Lewis structure in the space provided) Thank you!

