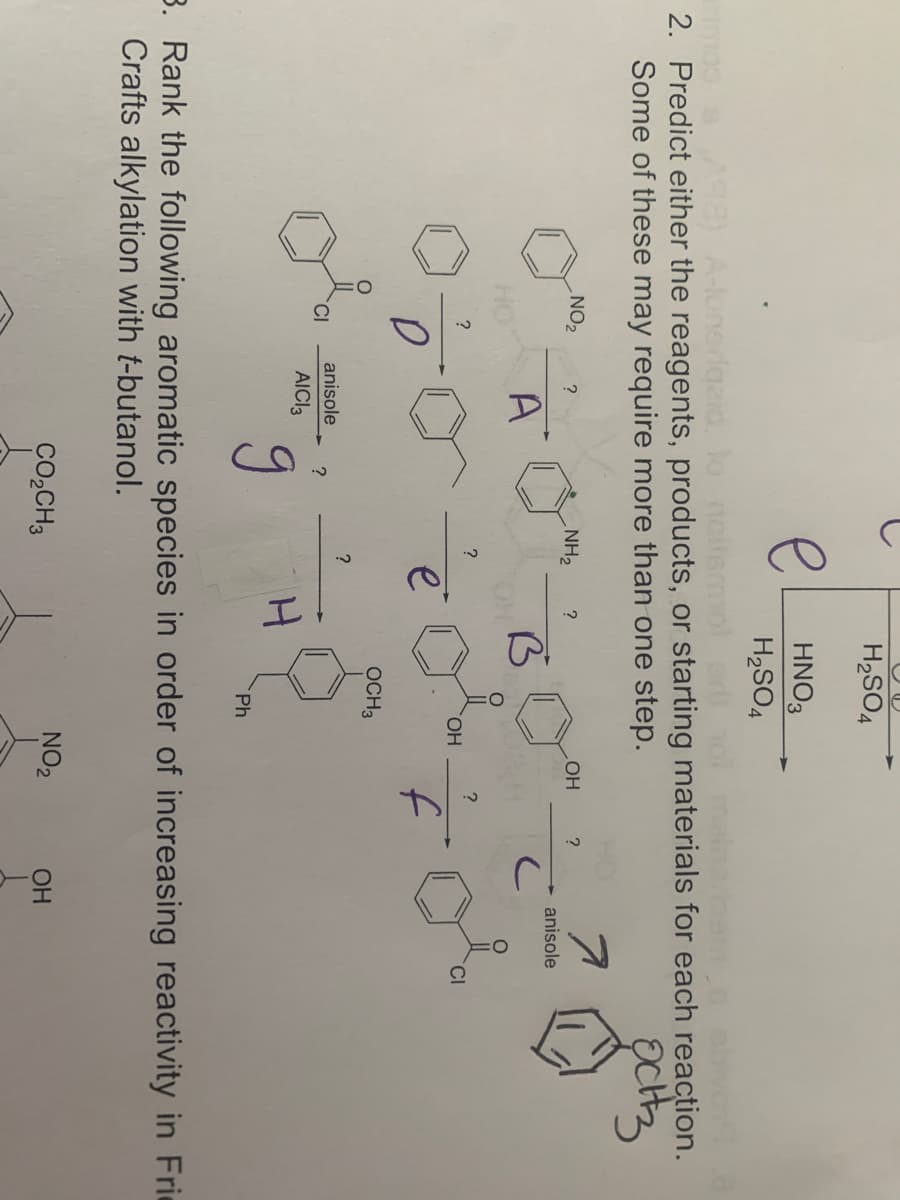
2. Predict either the reagents, products, or starting materi Some of these may require more than one step. NO2 ? NH2 HO ? A ? ? ? HO. e f OCH3 ? anisole CI ? AICI3 Ph
2. Predict either the reagents, products, or starting materi Some of these may require more than one step. NO2 ? NH2 HO ? A ? ? ? HO. e f OCH3 ? anisole CI ? AICI3 Ph
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 30MP: The carbocation electrophile in a Friede1-Crafts reaction can be generated by an alternate means...
Related questions
Question
100%
Predict the product or reagents. I’ve started it and want to make sure I’m on the right track!
Transcribed Image Text:H2SO4
e HNO3
H2SO4
mooaAS8) A-lonerdgeid. lo noilemolerolmainean oobvo
2. Predict either the reagents, products, or starting materials for each reaction.
Some of these may require more than one step.
NO2
NH2
?
OH
anisole
A
?
HO.
CI
f
OCH3
anisole
AICI3
Ph
3. Rank the following aromatic species in order of increasing reactivity in Frie
Crafts alkylation with t-butanol.
CO,CH3
NO2
OH
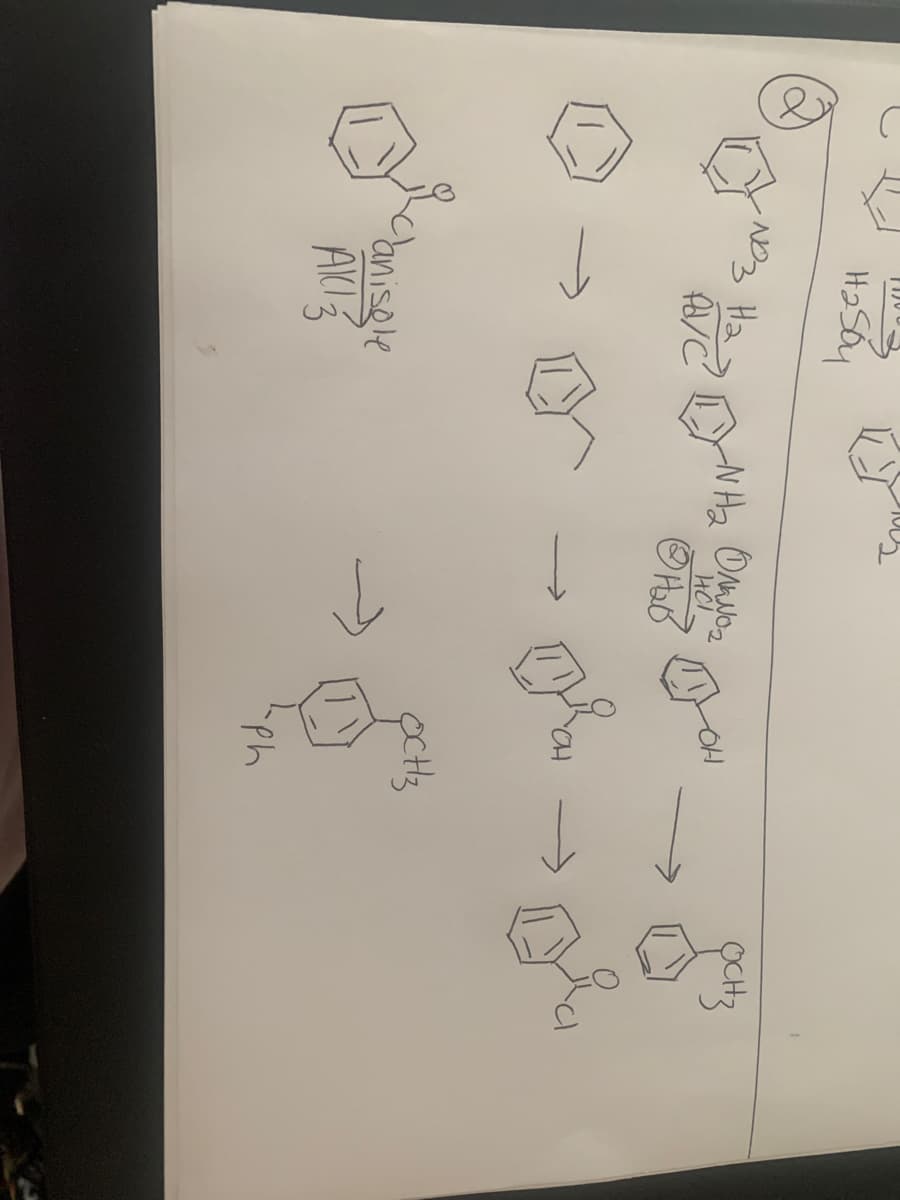
Transcribed Image Text:H2 Soy
Ha
N Ha Onyo
Canisele
Ph
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

