Q: 1. Please explain how an FTIR works 2. Why it's so important and useful for organic chemistry
A: Two questions based on IR spectroscopy, which are to be accomplished.
Q: A conjugated system involves O A.- two orbitals separated by one sp -hybridized carbon B. two p…
A:
Q: 1. Which of the bonds would have the highest bond dissociation energy? Why 2. How many sigma and pi…
A: Bond dissociation energy is a measure of the strength of the bond and is the amount of energy…
Q: 23. Which molecules is/are polar? A. H2O B. NH3 C. All are polar D. CH3F
A: The molecules that are polar has to be identified. The chemical compounds which are held together by…
Q: What orbitals are used to form each highlighted bond in the following molecule? In what type of…
A: The given compound has only sigma bonds and no pi bonds so all atoms in compounds have sp3…
Q: Statement 1: The boiling point of compound2 is lower because of the number of bondingregions present…
A: As the percentage of s-orbitals increases then the acidity of the compound also increases.
Q: Hybridization of marked carbon is * C=C. H3C `CH3 O sp2 O sp3d O sp3 O sp O None of these
A: Given Compound = H3C - C ≡ C - CH3 Hybridization of dark carbon = ?
Q: 7. In each set of structures, arrange_the labeled bond s in the order indicaleed Set 1 a Increasing…
A: We have to tell about the bond energy. We know that if a bond having greater s-character, then the…
Q: (a) How many π molecular orbitals are present in deca-1,3,5,7,9-pentaene (CH2 = CH – OH = OH – CH =…
A: (a)Since there are 5 pi bonds there will be 10 (5 x 2) molecular orbitals present in…
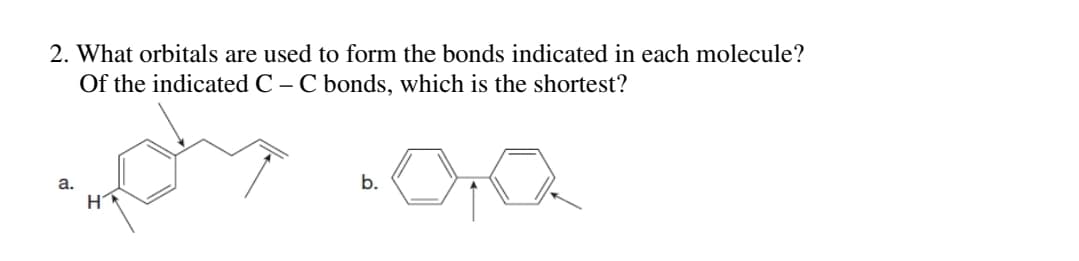
Q: What orbitals are used to form the labeled bonds in the following molecule? Of the labeled C—C…
A: 1--> C-H bond is formed due to overlap of sp2-s orbitals 2--> C-C bond is formed due to…
Q: A type of hyperconjugation occurs in the F-CH2-NH¯ anion even though there are no double bonds,…
A: The HOMO is the highest occupied molecular orbital. If the HOMO and LUMO orbitals can interact, then…
Q: [1] a. What orbitals are used to form the indicated bonds in a-sinensal? (3] -[2] :O: b. In what…
A: a) The orbitals involved in the formation are as follows: [1] σ orbital of sp2 hybridized C of one…
Q: An sp hybridizedc-CI bond is more polar than an sp? hybridized C- CI bond. (a) Explain why this…
A: Polarity is defined as the displacement or movement of electrons between two atoms. The electrons…
Q: The most stable MO of 1,3,5-hexatriene and the most stable MO of benzene Which compound is more…
A: Conjugated cycloalkenes has a property of stabilizing the molecule by the delocalization of pi…
Q: ービ Allene Cond) =Cニ THS コー CこCつ 1t
A:
Q: According to MO theory, which species is most stable? O CN+ O CN O CN
A:
Q: Norethynodrel is a synthetic hormone usedin Enovid, the first oral contraceptive.(a) Determine the…
A: In norethynodrel, the number of nonhydrogen atoms is seven. These are one carbonyl oxygen and six…
Q: Which orbital of O in H2O can form a π bond with an LCAO of the two H atoms?
A: Water molecule contains two sigma bond with two Hydrogen atoms by the overlapping of half filled…
Q: sp The hybridization of the nitrogen and the triple-bonded carbon are giving sp? geometry (C-C=N are…
A: sp sp Linear 180 sp2 Planar 120
Q: In the sketch of the structure of BF3 label all bonds. Drag the appropriate labels to their…
A: To label all the bonds.
Q: (i) how many π bonds does the molecule have? (ii) how many unhybridized p-orbitals total does the…
A: I have given a detailed solution to the question as follows:
Q: Which atomic orbital is most symmetrically suitable for forming a bond with the tw left lobes of the…
A: Most suitable symmetrical orbital to form bond with dxz orbital.
Q: Consider the three compounds shown below and then answer the question that follow: H H. H H H -C H…
A: If there is a C-C single bond present in the compound then the bond is Sp3 hybridized. If there is…
Q: H-S-H H2S Part 5 SO2
A: Steric number (SN) By the steric number we get the number of regions of electron around the central…
Q: For each of the indicated bonds, state which type of orbital from each atom is used to make the…
A: Carbon with 4 sigma bonds is sp3 hybridized Carbon with 3 sigma bonds is sp2 hybridized Carbon…
Q: A bond arises from "sideways" overlap of two parallel p orbitals. The electron density lies above…
A:
Q: Calculate the energy of isomer B if each 1,3-diaxial interaction costs 3.8 kJ/mol: Br CH -H O 3.8…
A: Presence of any bulky group at axial position will cost two 1,3-diaxial interaction. In the given…
Q: Which bond in the compound would require the minimum energy to undergo homolytic cleavage? a. Bond D…
A:
Q: Shown below is the structure of a potent natural anti-inflammatory agent, NPD1. Select the stater…
A: The “s–cis” is considered as a conformation in which both double bonds are present on the same side…
Q: 9. Januvia is the trade name for sitagliptin, a drug that increases the body's ability to lower…
A: The structure of the given drug molecule is: The 5-membered ring in the molecule is a triazole…
Q: Explain the Bond dissociation energies of the C–C ?
A: Bond dissociation energy is defined as energy required to break the bond present between the…
Q: Santalbic acid, a fatty acid isolated from the seeds of the sandalwood tree, is an unusual fatty…
A: Given,
Q: 2. Show the Atomic and molecular orbitals of acetic acid. Draw the energy diagram for the AO and MO.…
A: To show: atomic and molecular orbitals of acetic acid.
Q: How many conjugated n-electrons are in n-bonding MOs for the molecule shown? 4 6
A: In a molecule, there are two types of electrons, bonded and non-bonded. The bonded electrons are of…
Q: 6. Phomallenic acid C (shown) has antibacterial activity about 20-fold better than other drugs…
A: Let us answer the question in parts. Since there are 4 parts, we will answer only the first three…
Q: In the molecule shown below, determine which of the highlighted C-H bonds (from a to e) is expected…
A: The stability of the C-H sigma bond can be compared by the stability of the carbocation or the free…
Q: N: N–CH2CH3 | b CH,CH; a
A: During the orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals…
Q: most t ONE () of the following, then (a) draw twn possible resonance hybrids, and (b) circle the…
A: Resonance structures: structure which have least separation of charges is the best contributing…
Q: Identify the orbitals that are used to form each indicated bond. Rank the indicated bonds in the…
A: hydrocarbons divided into two types 1) Saturated 2)Unsaturated Unsaturated compounds are alkene…
Q: Which of the following molecules would be the least polar? A. H В. C. None of the above. All of…
A: When the electron pair is equally attracted by the two identical atoms, then the electron pair gets…
Q: 15. Label each lone pair in the molecule below as localized (L) or delocalized (DL). :NEÖH (a) What…
A: Localized lone pair: lone pair is perpendicular to the pi-electron system Delocalized lone pair:…
Q: a. b. H
A: The carbon atom joined to four substituents by single bonds is sp3 hybridized and that having one…
Q: What hybridization is taking place in the image shown below
A: According to the given diagram, In the excited state, one unpaired electron is present in the…
Q: lick to see additional instructions hown is a blank MO diagram depicting ONLY the valence shells.…
A:
Q: The bonding between the carbon and oxygen in a ketone can be described as C(sp²)-O(sp²), o-bond and…
A: The two pi-bonds in ketene are perpendicular to each other
Q: How many 1. the isobutyl cation? o bond orbitals are available for overlap with the vacant p orbital…
A: σ bonds are formed by linear overlap of atomic orbitals. Only σ bond orbitals are attached to an…
Q: On a piece of paper or electronic device, draw the most and the least stable Newman projections of…
A:
Q: Which C-H bond is the shortest? (Hint: sp3 carbon atoms have normal AOs, while sp carbon atoms have…
A: We have to predict the smallest C-H bond.
Q: Which molecular formula is correct for a saturated compound (containing no double or triple bond/s)?…
A: The given compounds are: (a) C4H8F2 (b) C4H8S (c) C5H9 (d) C6H6 (e) C4H4O
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- What orbitals are used to form the bonds indicated in each molecule? Of the indicated C – C bonds, which is the shortest?Represent the bonding in each molecule or ion by drawing the orbitals (hybridized and unhybridized) of each atom in the bond. Label the σ and π bonds and label each bond by the orbitals that it is made from. For instance, the C–H bond in CH4 would be made from the overlap of: C (sp3) – H (1 s). You may draw the hybridized sigma orbitals as sticks and the unhybridized p-orbitals as lobes for clairity and ease. a. HONO b. CH3 CCH c. C3 H4 d. C2 O4 2-What orbitals are used to form each highlighted bond? For multiple bonds, indicate the orbitals used in individual bonds.
- Norethynodrel is a synthetic hormone usedin Enovid, the first oral contraceptive.(a) Determine the hybridization of eachnonhydrogen atom. (b) How many total σbonds and π bonds does norethynodrelhave?Which of the given resonance structures (A, B,or C) contributes most to the resonance hybrid? Which contributes least?Which of the given resonance structures (A, B, or C) contributes most to the resonance hybrid? Which contributes least?
- A. Find the moleular formula of diazomethane. b. Draw iits lewis structure and resonance forms as neccessary c. Explain why diazomethane is not very stableeach reagant must have 3 or less carbons . one image is the directions and one is the question.Explain why the bond dissociation energy for bond (a) is lower than the bond dissociation energy for bond (b).
- What orbitals are used to form the labeled bonds in the following molecule? Of the labeled C—C bonds, which is the shortest?How many σ and π bonds are in the following molecules? And what these σ and π bonds mean?The unmistakable odor of a freshly cut cucumber is largely due to cucumber aldehyde. (a) How many sp2 hybridizedcarbon atoms does cucumber aldehyde contain? (b) What is the hybridization of the O atom? (c) What orbitals are used toform the carbon–oxygen double bond? (d) How many s bonds does cucumber aldehyde contain? (e) How many p bondsdoes it contain?

