2.4e+05 2.2e+05 2.0E+05 1.8e+05 1.6e+05 1.4e+05- 1.2e+05 1.0e+05- 8.0E+04- 6.0E+04 4.0E+04- 2.0E+04- 0.0e+00 T 11 10 -8.933 1.00 -7.385 A 1.13 5.305 4.973 -4.831 4.594 3
2.4e+05 2.2e+05 2.0E+05 1.8e+05 1.6e+05 1.4e+05- 1.2e+05 1.0e+05- 8.0E+04- 6.0E+04 4.0E+04- 2.0E+04- 0.0e+00 T 11 10 -8.933 1.00 -7.385 A 1.13 5.305 4.973 -4.831 4.594 3
Chapter9: Energy For Today
Section: Chapter Questions
Problem 9E
Related questions
Question
2
Transcribed Image Text:2.4e+05
2.2e+05
2.0E+05
1.8e+05
1.6e+05-
1.4e+05-
1.2e+05-
1.0e+05-
8.0e+04-
6.0E+04
4.0E+04
2.0E+04-
0.0e+00
T
11
10
thanks!
-8.933
i
-7.385
ith
8
5.305
4.973
4.831
4.594
5
3
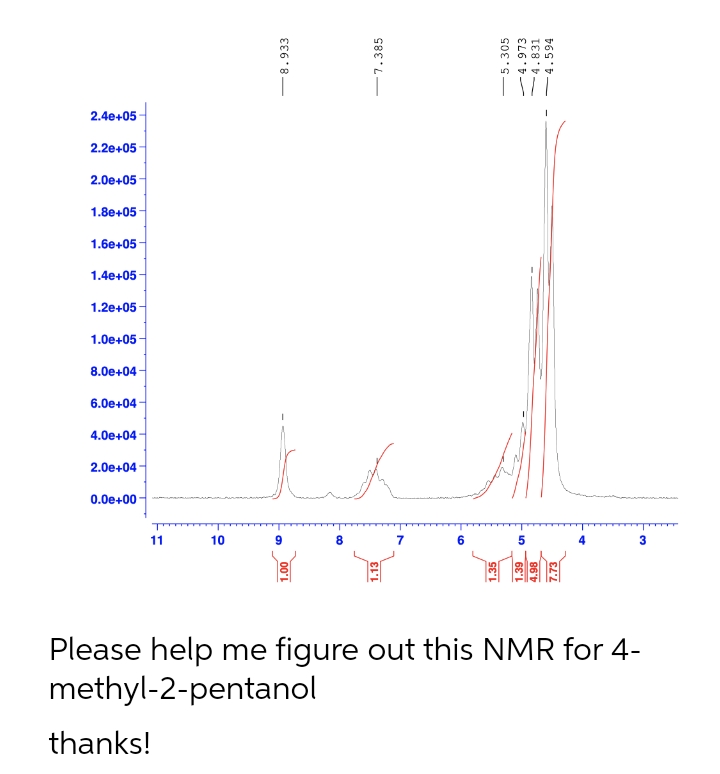
Please help me figure out this NMR for 4-
methyl-2-pentanol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you





