20.Use the following data to determine the reaction rate law for the system. Be sure to solve for the rate law constant as well including its units for full marks NO(g) + H2(g) →→→→→→HNO2(g) Experiment 1 2 3 4 5 6 NO (mol/L) 0.001 0.002 0.003 0.004 0.004 0.004 H2 (mol/L) 0.004 0.004 0.004 0.001 0.002 0.003 Initial Rate of Reaction (mol/(L.s)) 0.004 0.0016 0.036 0.016 0.032 0.048
20.Use the following data to determine the reaction rate law for the system. Be sure to solve for the rate law constant as well including its units for full marks NO(g) + H2(g) →→→→→→HNO2(g) Experiment 1 2 3 4 5 6 NO (mol/L) 0.001 0.002 0.003 0.004 0.004 0.004 H2 (mol/L) 0.004 0.004 0.004 0.001 0.002 0.003 Initial Rate of Reaction (mol/(L.s)) 0.004 0.0016 0.036 0.016 0.032 0.048
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section11.9: Enzymes: Biological Catalysts
Problem 11.14CE
Related questions
Question
Pls help ASAP on all ASKED QUESTIONS PLS PLS
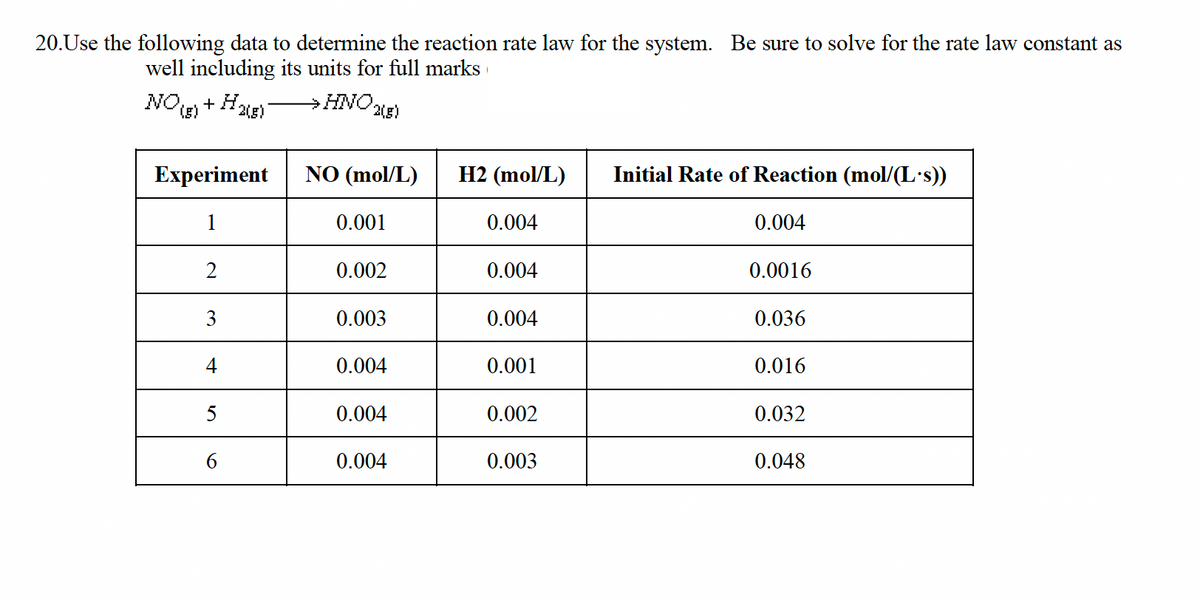
Transcribed Image Text:20. Use the following data to determine the reaction rate law for the system. Be sure to solve for the rate law constant as
well including its units for full marks
NO + H2(g)
→HNO,
Experiment
1
2
3
4
5
6
(2(g)
NO (mol/L)
0.001
0.002
0.003
0.004
0.004
0.004
H2 (mol/L)
0.004
0.004
0.004
0.001
0.002
0.003
Initial Rate of Reaction (mol/(L.s))
0.004
0.0016
0.036
0.016
0.032
0.048
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning