Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter13: Substitution
Section: Chapter Questions
Problem 58CTQ
Related questions
Question
Can i get help with these problems

Transcribed Image Text:The Nucleophile
Recall that the rate of an Sy2 process is dependent on the concentration of the nucleophile. For the
same reason, SN2 processes are also dependent on the strength of the nucleophile. A strong nucleo-
phile will speed up the rate of an SN2 reaction, while a weak nuclcophile will slow down the rate of
an Sy2 reaction. In contrast, an SN1 process is not affected by the concentration or strength of the
nucleophile because the nucleophile does not participate in the rate-determining step. In summary,
the nucleophile has the following cffect on the competition between Sy2 and Syl:
• A strong nucleophile favors Sy2.
• A weak nucleophile disfavors Sy2 (and thereby allows Sy1 to compete successfully).
We must therefore learn to identify nucleophiles as strong or weak. The strength of a nucleophile is
determined by many factors that were first discussed in Section 6.7. Figure 7.26 shows some strong
and weak nucleophiles that we will encounter.
Common nucleophiles
Strong
Weak
HS
но
H2S
RO
H20
RSH
NEC ROH
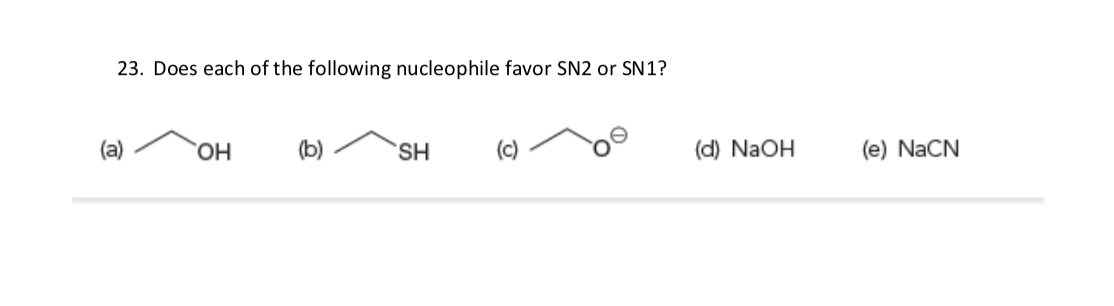
Transcribed Image Text:23. Does each of the following nucleophile favor SN2 or SN1?
(a)
HO,
(b)
S.
(c)
(d) NaOH
(e) NaCN
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning