25. A student hypothesizes that an increase in pressure will increase the amount of ammonia produced in the reversible reaction shown. N2(g) + 3H2(9) 2NH3(g) nitrogen hydrogen ammonia Which best explains how an increase in pressure increases the amount of ammonia present? O An increase in pressure decreases the rate of reaction between gases. An increase in pressure would cause the forward reaction to be endothermic. An increase in pressure tends to increase the temperature of the gases. An increase in pressure favors the product with fewer particles.
25. A student hypothesizes that an increase in pressure will increase the amount of ammonia produced in the reversible reaction shown. N2(g) + 3H2(9) 2NH3(g) nitrogen hydrogen ammonia Which best explains how an increase in pressure increases the amount of ammonia present? O An increase in pressure decreases the rate of reaction between gases. An increase in pressure would cause the forward reaction to be endothermic. An increase in pressure tends to increase the temperature of the gases. An increase in pressure favors the product with fewer particles.
Chapter13: Chemical Equilibrium
Section: Chapter Questions
Problem 10RQ: The only stress (change) that also changes the value of K is a change in temperature. For an...
Related questions
Question
Transcribed Image Text:All char
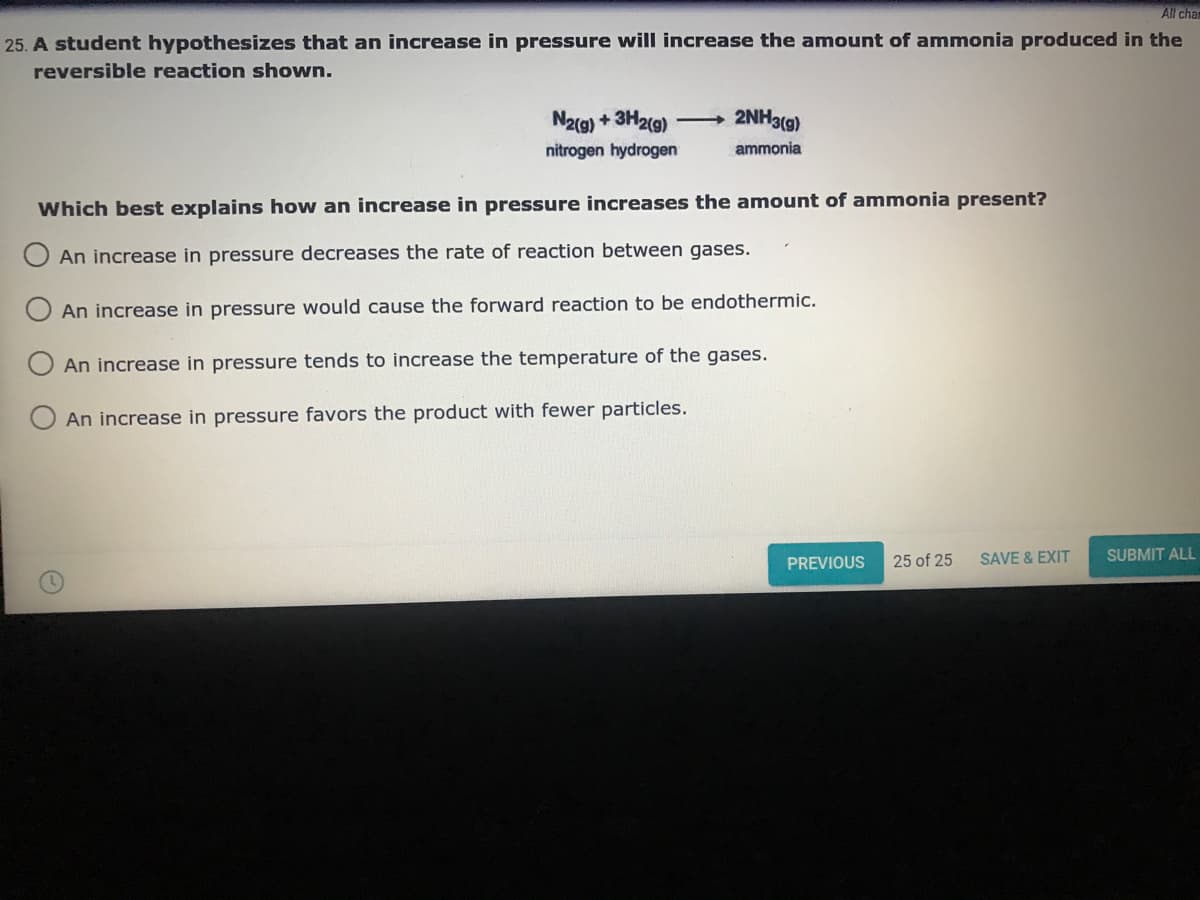
25. A student hypothesizes that an increase in pressure will increase the amount of ammonia produced in the
reversible reaction shown.
N2(g) + 3H2(g)
2NH3(g)
nitrogen hydrogen
ammonia
Which best explains how an increase in pressure increases the amount of ammonia present?
An increase in pressure decreases the rate of reaction between gases.
An increase in pressure would cause the forward reaction to be endothermic.
An increase in pressure tends to increase the temperature of the gases.
An increase in pressure favors the product with fewer particles.
PREVIOUS
25 of 25
SAVE & EXIT
SUBMIT ALL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning