250 ml volume of a mixture of 0.8g of NaHCO3 and 0.2g of NaOH in water. Take 50 ml of this mixture and titrate with o.1 N of HCI. Calculate the volume of acid? ( Atomic weight = Na=23, H=1,O =16, CI =35 , C=12, N= 14) * 30 ml. 5 ml. 25 ml. 10 ml. O 45 ml.
250 ml volume of a mixture of 0.8g of NaHCO3 and 0.2g of NaOH in water. Take 50 ml of this mixture and titrate with o.1 N of HCI. Calculate the volume of acid? ( Atomic weight = Na=23, H=1,O =16, CI =35 , C=12, N= 14) * 30 ml. 5 ml. 25 ml. 10 ml. O 45 ml.
Chapter20: Carboxylic Acids And Nitriles
Section20.3: Biological Acids And The Henderson–hasselbalch Equation
Problem 5P: Calculate the percentages of dissociated and undissociated forms present in the following solutions:...
Related questions
Question
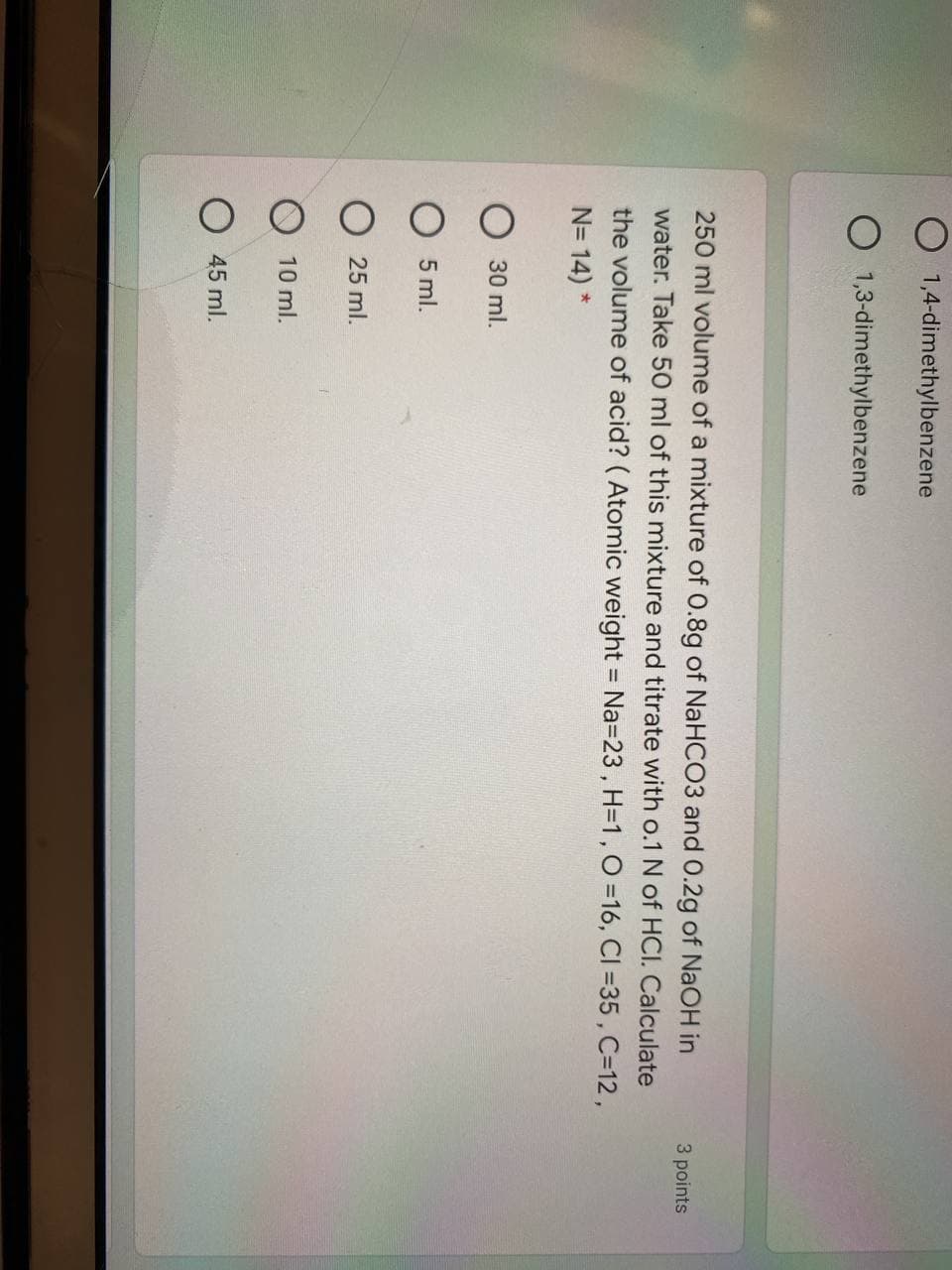
Transcribed Image Text:O 1,4-dimethylbenzene
O 1,3-dimethylbenzene
250 ml volume of a mixture of 0.8g of NaHCO3 and 0.2g of NaOH in
3 points
water. Take 50 ml of this mixture and titrate with o.1 N of HCI. Calculate
the volume of acid? ( Atomic weight = Na=23, H=1,O =16, CI =35 , C=12,
N= 14) *
30 ml.
5 ml.
O 25 ml.
10 ml.
O 45 ml.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



