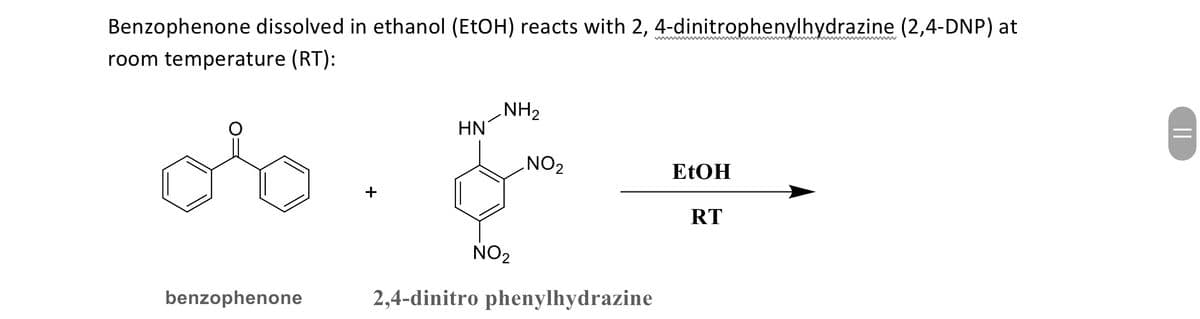
|| wwwwww wwwwwwww Benzophenone dissolved in ethanol (EtOH) reacts with 2, 4-dinitrophenylhydrazine (2,4-DNP) at room temperature (RT): ob NH2 HN NO2 EtOH RT benzophenone & NO2 2,4-dinitro phenylhydrazine
Q: ANSWER IN kJ!!!!!! Careful with sig figs
A: Step 1:Step 2:
Q: Draw structural formulas for organic products A and B in the window below. CI Li CH3CHCH3 pentane…
A: Step 1:Alkyl chloride reacts with Li to form alkyl lithium. Isopropyl chloride reacts with Li (in…
Q: 4. The solution of the HMOs for the allyl cation gives the following in order of increasing…
A: 4.The size of the dot represents the magnitude of the orbital coefficient and the color represents…
Q: What volume ( in Liters) will 2.0 g of hydrogen gas occupy at STP conditions?
A: Step 1: Calculation of moles of hydrogen gas Given,mass of hydrogen gas = 2.0 g molar mass of…
Q: Please help me with the question below. A detailed explanation to aid in understanding is welcome.
A: Explanation is along with answer. *****GIVE HELPFUL RATING*****
Q: Identify the reaction for this equilibrium expression. K = [H+]²[SO²-] [H2SO4] ○ H2SO4 (aq) = H+…
A: The objective of the question is to identify the correct chemical reaction that corresponds to the…
Q: H H рух H Q:O Select to Add Arrows H H HH H H Select to Add Arrows H3O+ heat H H ་ 0:0 H HH H3O+ 'H…
A:
Q: Draw the peptide Glycine-Alanine-Serine-Cysteine-Isoleucine-Glutamic acid Tryptophan-Valine. Circle…
A: Approach to finding a solution to the problem:To begin, you will need to draw the peptide sequence.…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. • • 1. in…
A: First one is the product. But due to the presence of acid, the ester undergoes hydrolysis to give…
Q: 1 A reaction between X and Y was carried out at various temperatures and the rate constant…
A: The objective of the question is to draw an Arrhenius plot using the given data. The Arrhenius…
Q: H3C-CH-CH=CH₂ C=C-H 2eq. KMnO4 NaOH, H₂O
A:
Q: Calculate the molarity of the acetic acid solution when 20.0 mL of that solution has been titrated…
A: Step 1: Step 2: Step 3: Step 4:
Q: how to sythesis this 1-chloro-2-(4-methylphenoxy)benzene from 1-methy-4-phenoxybenzene .
A: Step 1:Nitration: Start by nitrating 1-methyl-4-phenoxybenzene. This involves introducing a nitro…
Q: J9 cmsingi eltha noitulos eucoupe M 00 ebixonbyr Oxidation-Reduction Stoichiometry PL sin (1)ego to…
A: The objective of the question is to calculate the mass of copper produced, the excess reactant…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Step 1: Reaction of 2-iodopent-1-ene on heating with methyl amide ion gives E2 elimination…
Q: please help me with the answer B
A: Reaction involved is Pb(NO3)2(aq) + 2KCl(aq) -> PbCl2(s) + 2KNO3(aq)Molarity of lead nitrate…
Q: Show how you would synthesize each compound from benzene, toluene, or phenol using the following…
A: Step 1: 1. Sulfonation (Reaction 3): Toluene can be sulfonated to form toluenesulfonic acid by…
Q: Draw the major product of this reaction. Use a dash or wedge bond to indicate the stereochemistry of…
A: Step 1: Step 2: Step 3: Step 4:
Q: Identify unknown diatomic gas that filled a gas-tight cylinder to 33.5 mL, at pressure of 2.41 atm…
A:
Q: 3. Propose syntheses of the following two targets beginning with the indicated starting material. Но…
A: Step 1: Step 2: Step 3: Step 4:
Q: draw out the mechanism of the following reaction. be sure to draw all intermediates and use arrows…
A: This reaction is the conversion of GTP (Guanosine triphosphate) +IMP + L-Aspartate into GDP…
Q: A concentration cell is constructed of silver electrodes at 25∘C, and the half cells contain…
A: A concentration cell is a type of electrochemical cell that generates electrical energy from the…
Q: For the following equilibrium system,CO; (g) + Ca(OH)2(s) = CaCO (s) + H2O(1) + HeatAH = -113 kJ a.…
A: ### Reaction:CO2(g) + Ca(OH)2(s) → CaCO3(s) + H2O(l) + HeatΔH = -113 kJThis reaction is exothermic…
Q: Draw a peptide with four different amino acids. Name the peptide and describe the type of amino acid
A: Certainly! "Gly-Ser-Val-Ala" represents a peptide chain composed of four amino acids: Glycine (Gly),…
Q: I understand how to solve the questions, but in the overall question how do we know that the unknown…
A:
Q: The following information is given for bismuth at 1atm: boiling point = 1627 °C melting point = 271…
A: The objective of the question is to calculate the enthalpy change (ΔH) for the process of freezing a…
Q: 4. Evaluate the strain hardening increment to the yield strength of Li [BCC, G (shear modulus) = 32…
A:
Q: The figure to the right illustrates the long-run average cost curve for a company that makes motors.…
A: When analyzing the long-run average cost curve of a company, we're essentially studying how average…
Q: Include all lone pairs and charges as appropriate. Use wedges and dashes to indicate any chiral…
A:
Q: An electron in the n = 6 level emits a photon with a wavelength of 410.2 nm. To what energy level…
A:
Q: 14
A:
Q: A certain metal M forms a soluble sulfate salt M,SO4. Suppose the left half cell of a galvanic cell…
A: If you need any clarification, please comment.
Q: Given the background IR spectra, rank the three methods (Nujol mull, CCl4 solution, and KBr pellet)…
A: KBR is trans parent throughout whole region so this is the best. CCl4 only shows peaks at 800 cm-1 ,…
Q: Construct the wave function, psin,l,m(r, theta, phi), for the 3s, 2p and 3p and 3d orbitals of the…
A: The objective of the question is to construct the wave function for the 3s, 2p, 3p and 3d orbitals…
Q: MeO 22.55 One potential synthesis of the anti-inflammatory and analgesic drug nabumetone is…
A: <p>Regioselectivity refers to the preference of one direction of chemical bond making or…
Q: A sample of neon gas occupies a volume of 6.57 L at 67.0°C and 315 torr. If the volume of the gas…
A: The objective of this question is to find the resulting gas pressure when the volume and temperature…
Q: Chemistry
A: Step 1: Step 2: Step 3:
Q: Could I get a detailed explanation for c); d) i, ii, iii please
A: Step 1:(a) The charge of the M ion in M(OH)2 is +2, and in MCO3, it is +2 as well. Step 2:(b) (i)…
Q: (please correct answer and don't use hend raiting) How many moles are in 39.2 of Magnesium…
A:
Q: Calculate the freezing point and boiling point of each aqueous solution, assuming complete…
A:
Q: reaction conditions and follow the curved arrows and draw the intermediate and product in this…
A:
Q: Determine ∆S° for 2 O₃(g) → 3 O₂(g). Substance Sf° (J/mol・K) O₃ (g) 239 O₂ (g) 205
A:
Q: Show how you would synthesize the following compounds from benzene or toluene. Use any other organic…
A:
Q: Draw the starting structure that would lead to the major product shown under the provided…
A:
Q: The hand written solution is not allowed dear please
A: The given alkyl halide is a primary alkyl halide. Azide ion will readily reacts with the given alkyl…
Q: The pressure in Denver, Colorado (5280-ft elevation), averages about 25.5 in. Hg.
A: Step 1: The pressure in Denver, Colorado (5280-ft elevation), is about, P = 25.5 in. HgWe know that…
Q: Name a list of drugs that consist of aromatic compounds?
A: The objective of the question is to identify a list of drugs that consist of aromatic compounds.…
Q: Phosgene decomposes to carbon monoxide and chlorine gas. COCl2 (g) CO (g) + Cl2 (g) If the reaction…
A: The question is asking about the effect of adding more phosgene gas (COCl2) to a reaction that has…
Q: 1. Pushing arrows. Use curved arrows to show the movement of electrons in the reactions bellow. A)…
A:
Q: . Consider the reaction between sodium chlorate and sugar, shown below. 8NaClO3 (s) + C12H22O11 (s)…
A: thanks.
Step by step
Solved in 2 steps with 1 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Benzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.4 Show how the compound thiodiazine~C21H26N2S2~decomposes anaerobically and how these end products in turn decompose aerobically to stabilized sulfur and nitrogen compounds.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?
- Plz provide ALL the (carbon-containing) products of the following reactions:Tributyltin hydride (Bu3SnH) is used synthetically to reduce alkyl halides, replacing a halogen atom with hydrogen. Free-radical initiators promote this reaction, and free-radical inhibitors are known to slow or stop it. Your job is todevelop a mechanism,Alcohols can undergo a lot of different reaction mechanims. If the alcohol group (OH) is attaached to an aromatic core, how will the chemistry change as compared to a typical alkyl alcohol? A) The OH group will become more polarised and more nucleophilic. B) The OH group will become more susceptible to oxidation C) The OH group will become more polarised and therefore basic D) The OH group will become more polarised and therefore acidic.
- Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained: Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.4.4 Show how the compound thiodiazine - C21H26N2S2 - decomposes anaerobically and how these end products in turn decompose aerobically to stabilized sulfurand nitrogen compounds.A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions

