Q: Acetaminophen, a popular drug taken as pain reliever and fever reducer, is produced together with…
A: Given Reaction C6H7NO + C4H6O3 → C8H9NO2 + C2H4O2 Mass of 4 amino phenol =…
Q: GRAVIMETRIC ANALYSIS OF A TWO COMPONENT MIXTURE A mixture of NaHCO3 and Na2CO3 reacts with…
A: A numerical problem based on mole concept, which is to be accomplished.
Q: Add Stoichmetric factor: ______CH3CH2OH + _______O2 yields ________CO2 + _______H20
A: Balancing reaction
Q: measurement of volume capacity of volumetric flask: please help me fill in the blanks Temperature of…
A:
Q: 1. Calculate the GFW of chromium (III) carbonate, Cr2(CO3)3, to four significant figures. From…
A:
Q: A Moving to the next question prevents changes to this answer. Question 1 The combustion of ammonia…
A: 4NH3(g) + 7O2(g) .....> 4NO2(g) + 6H2O(g) Molar mass of ammonia = Molar mass of (N + 3(H)) =…
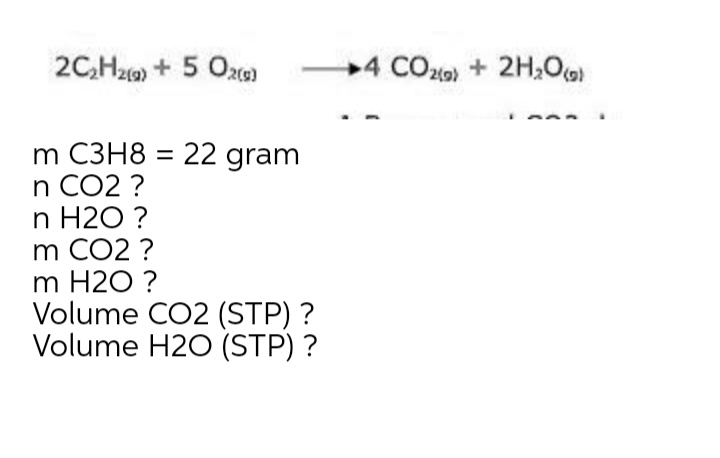
Q: C3H8(g) + 5O₂(g) →3CO₂(g) + 4H₂O(g) Rate = Fe(OH)3(s) +3HBr(aq) → FeBr3(aq) + 3H₂O(1) Rate =…
A:
Q: gma Sand Sigma Shale Sigma Hydrocarbon Shale volume, Vsh Porosity 6 cu 30 cu 20 cu 10% 25 pu…
A:
Q: The maximum dose over 24 hours for OTC (over the counter use) of acetaminophen (medication in…
A: Given that each Norco tablet contains 325 mg acetaminophen. We have to find the grams of…
Q: About 200 grams of glucose was fermented with 20 grams of yeast inside a fermenting vessel for 2…
A: Given 200 g of glucose give ethanol Actual yield = 84.5mL We have to find actual yield
Q: Find the mass of hydrogen gas produced by the reaction of 4.0g Al with excess sulfuric acid by using…
A: Here we are required to find the mass of hydrogen gas produced when 4gm of Al reacted
Q: Mass of unknown mixture (g) 0.5000 g Mass of NaCI formed (g) 0.4100 g Mass of Na,CO; in mixture (g)…
A: A mixture of NaHCO3 and Na2CO3 reacts with hydrochloric acid solution to produce three common…
Q: The combustion of ammonia in the presence of excess oxygen yields NO2 and H2O:…
A: Ammonia reacts with oxygen to form nitrogen dioxide and water. The equation for the balanced…
Q: A group of students was tasked with synthesizing dibenzalacetone (DBA) in the lab. They used acetone…
A: A question based on general chemistry that is to be accomplished.
Q: Which are characteristics of a primary standard? has an anhydrous form as a well as hydrated form…
A: The characteristics of a primary standard have to be given,
Q: * Identity of Dehydrated salt (for afterwards) Mass of empty dish 70.874g Mass of dish + hydrated…
A: The number of moles of dehydrated salt is 0.002384 mol. nMnCl2=mMnCl2MMMnCl2=0.3 g125.844…
Q: Molly Cule weighed 8.61g of her hydrate (which is CaSO4*2H2O). After she did her hydrates lab, she…
A: a) Since the difference between the weight of hydrate and anhydrate should be equal to the weight of…
Q: HgI2+ 2KI + 2H2O —> K2[HgI4].2H2O 1 g of each reactant is weighed and the reactian is inserted and…
A: The reaction taking place is given as, => HgI2 + 2 KI + 2 H2O -------> K2[HgI4].2H2O Given :…
Q: 4. In the background reading, it was pointed out that heroin can be produced from morphine in a…
A: The Density of Acetic anhydride is 1.08 g/cm3, then the mass of Acetic anhydride is - Mass of Acetic…
Q: If a liter of diesel emits 26.4 g of carbon dioxide in one kilometer, how much carbon dioxide is…
A:
Q: How many grams of CO2 are produced from burning one gallon of biodiesel (3,311 g of biodiesel)?…
A:
Q: 1. The Starship Enterprise on Star Trek uses a compound, B;H9, and oxygen gas as fuel. The two react…
A: Balanced chemical equation 2 B5H9 + 12O2 = 5 B2O3 + 9H20
Q: For the reaction C4H10 + O2 --> CO2 + H2O a) what weight of C4H10 in grams is needed for 3.01 x…
A:
Q: The density of bromine is 3.1 g/ml. What would the amount be in mols, of a 25ml sample of bromine?…
A: Density can be calculated using the formula: Density = Mass / Volume…
Q: mixture of NaHCO3 and Na2CO3 reacts with hydrochloric acid solution to produce three
A: We have to find mass of sodium carbonate and sodium bicarbonate in 0.5 g mixture
Q: Carbon dioxide gas, CO2(g), is generated in the combustion of a sample of ethane (C2H6). This CO2 is…
A: Given statement is : Carbon dioxide gas, CO2 (g), is generated in the combustion of a sample of…
Q: How can you prepare 500 ml of 0.1M HCI, if the percentage is 35%, and density = 1.18g/ml,…
A: Mass percentage or percentage by weight is one of the concentration terms that indicates the mass of…
Q: 1-10. The table shows fuel efficiency for several automobiles CO2 emission (g CO2/km) Fuel…
A: (a)The number of miles in 1 km is 0.621371miles.
Q: convert 1. 0.7 pg into ng 2. 13 dm to mm 3. 2.5x10-15 mole into pmole 4. 3.5 x105μm into cm…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: 4AI203 + 3CH4→ 8AI + 3CO2 + 6H20 AH° rxn kJ exo ? endo thermic AH° = -1676 -75 -394 -242 kJ/mol…
A:
Q: ASS Gwin x O Homepage - 20218 x Classwork for 2021 x E Danesha Patterson x A Commonlit | The S + a…
A:
Q: Active Ingredient Raw Materials Actual Yield Formulation Dosage Form Packaging Seignette salt 1.20…
A: Active Ingredient : Siegnette saltRaw Materials: Mass of cream of tartar (KC4H5O6) = 1.5 kg = 1500 g…
Q: Obtain values from the pictures found in the procedure to complete the data table below. Remember to…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Mass of unknown mixture (g) 0.5000 g Mass of NaCI formed (g) 0.4100 g Mass of Na;CO; in mixture (g)…
A:
Q: ml. meles CUSO. meles NH. ml. meles cone conc 0.1 M CuSO. 2.5M Cu(NH) Ce(NH) NH. formed 0.0001…
A: First we need to write a balanced chemical reaction for formation of Cu(NH3)42+ . The using ICE…
Q: For the following table fill in the missing values and calculate the theoretical yield.…
A: According to the given data and the chemical equation, B is the limiting reagent in the reaction. It…
Q: H20 H,SO4 OH 1. O3 2. H2O2 CI Br Na, NH3(1) low temp.
A: The name of the starting material is (1S, 2R, 3S)-2-bromo-3-chloro-1,2,3-trivinylcyclopropanol.
Q: Ingliest mg po H. 3. H-C-O-C-H .. a. H. H H H-C-C-ÖH b. H H…
A: Boiling point of alcohol is always greater than ethers, aldehydes or alkanes having similar…
Q: How many grams of carbon is present in 100 g Ca(HCO3)2? Write your final answer to the nearest whole…
A: Mass of 1 mole Ca(HCO3)2 is 162 g In 1 mole calcium bicarbonate, there are 2 moles of C atom,, mass…
Q: 150 moles of A reacts with 200 moles of B via the reaction 4A+B - 3C+4D The conversion of the…
A: Given Number of mole of A = 150 mole Number of mole of B = 200 mole Limiting reactant = ? Number of…
Q: What is the molecular mass (in AMU) and molar mass (in g mol-1) for chloroform (CHCI3) ?
A: The compound given is CHCl3.
Q: In a converter, 9.33 kg of SO3 is fed and allowed to come into contact with a certain amount of…
A:
Q: The percentage of an additive in gasoline was measured six times with the following results: 0.13;…
A: The expression for the confidence interval is shown below: Confidence interval =μ±t×σN where; μ =…
Q: 2. AClsle) → ACI3(e) + Clz{e) AH = ? Given: Aa(s) + 6CI2(e) → 4ACI3{g) AH = -2439 kJ 4ACIS(e) →…
A:
Q: ΔH° f (kJ/mol) SO 2Cl 2( g) + 2 H 2O( l) → 2 HCl( g) + H 2SO4( l) ΔH° rxn = ? SO 2Cl 2( g) -364 H…
A: Interpretation- To determine the enthalpy of the reaction ( ΔH° rxn ) where given reaction is -…
Q: Uploaded is an example from Chapter 8 in my chemistry book. If you could break down the how and why…
A: The mole ratio in any reaction is can be find using the balancing coefficients because in any…
Q: Please help me, and double and triple check your answers previous tutors got questions wrong
A: Given Data, Molar mass of SO2 = 64.064 g/mol Molar mass of CS2 = 76.143 g/mol
Q: Mass of unknown mixture (g) 0.5000 g Mass of NaClI formed (g) 0.4100 g Mass of Na;CO, in mixture (g)…
A: Given : Mass of mixture = 0.5000 g And mass of NaCl produced = 0.4100 g The balanced reactions…
Q: Wite missing combustion reaction coefficients in the following equation: CgH12 + (02 + N2) = 5.7CO2…
A:
Q: 2 NOCI(g) → 2 NO(g) +C12(g) AH° =+75.56 kJ 2 NO(g) + 02(g) → 2 NO2(g) AH° =-113.05 kJ 2 NO2(g) →…
A:
Step by step
Solved in 3 steps

- I am allowed to work with someone... Using the Volumetric Pipet Data from Part C Calculate the: 12. Percent error between the known density (from Data A) and the average experimental density (from Calc 10)Sum of coefficients C7H8 + O2 --> CO2 + H2O after balancingH2SO3 = 0.2556 M, Ka1 = 1.6 x 10-2 , Ka2 = 6.4 x 10-8 NaOH = 0.3106 M please show all work all information needed is here
- Given: F2(g) + 2 e- ⟶ 2 F-(aq) Eo = +2.87 v Hg2+(aq) + 2 e- ⟶ Hg(l) Eo = +0.87 v Calculate ΔGo for: Hg(l) + F2(g) ⟶ Hg2+(aq) + 2 F-(aq) ΔGo = -nFEo F = 96500Explain why it is problematic to include a constraint such as the following in an LP model for a blending problem: Total octane in gasoline 1 blend $ divided by Barrels of gasoline 1 blended daily which is greater thanor equal to 10if a volumetric pipet delivers 24.844g of deionized water at 31.00 degrees C, what is the calibrated volume of the pipet?
- Carbonic anhydrase of erythrocytes (Mr 30,000) has one of the highest turnover numbers known. It catalyzes the reversible hydration of CO2:This is an important process in the transport of CO2 from the tissues to the lungs. If 10.0 μg of pure carbonic anhydrase catalyzes the hydration of 0.30 g of CO2 in 1 min at 37 °C at Vmax , what is the turnover number (kcat) of carbonic anhydrase (in units of min −1 )?1. Estimate the density of a 25-API gravity dead oil at 100 F.10.5mL (mw 106) of benzaldehyde and 2.9g(3.63mL, mw 40) of acetone react with 5g of NaOH and 25mL of Ethanol to form 6.7g of Dibenzalacetone. What is the theoretical, actual and percent yield?

