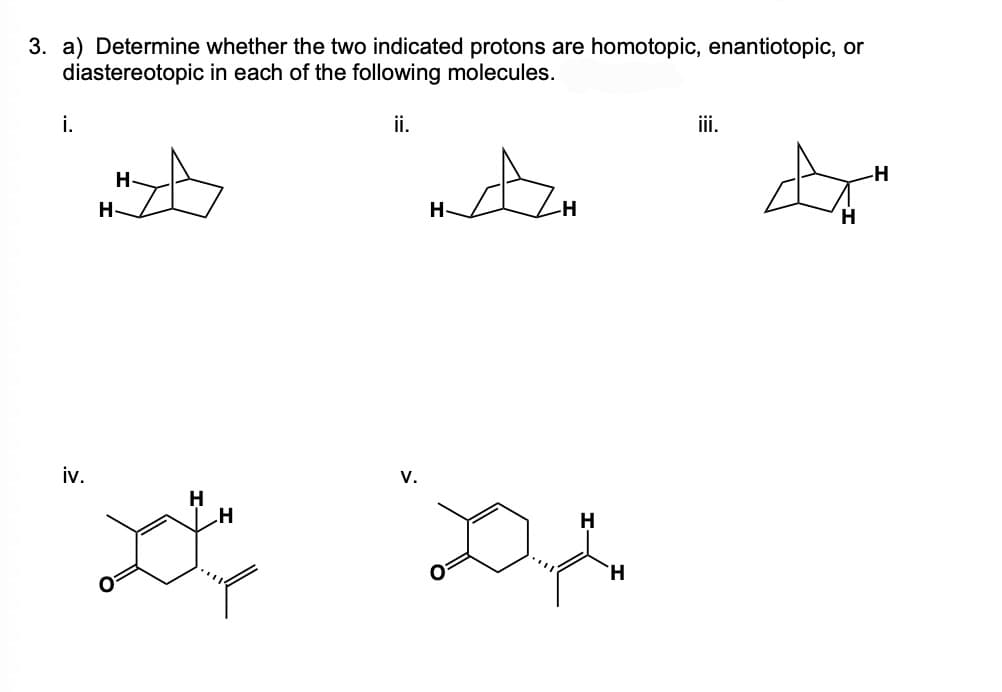
3. a) Determine whether the two indicated protons are homotopic, enantiotopic, or diastereotopic in each of the following molecules. i. iii. H H ii. A iv. V. H H to" H
Q: reaction vessel compound concentration expected change in concentration CH2CH2NH₂ 0.77 M ○ ↑…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: i need the answer quickly
A: Oxidation number (oxidation state) indicates the number of electrons that an atom has gained or…
Q: Please help me , i need fast solution. Please don't provide handwritten solution ...
A: The objective of this question is to calculate the pH of a solution of NH3 after the addition of…
Q: 3. Predict the product of the following multistep reaction. dis Br 1a) Mgo, ether b) iv c) H3O+ 2)…
A: Based on the given reagents and conditions, the final product is likely the corresponding ketone…
Q: How many molecules are in 79.6 moles of CO₂?
A: The objective of this question is to calculate the number of molecules in a given amount of moles of…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: LDA is a sterically hindered strong base and it is used in the formation of the kinetic enolate ion.
Q: What is the major neutral organic product for the following sequence? 1. KÊN 2. H+ 3. H3O+, heat ?…
A: -> Carbonyl compounds have electrophilic center and KCN has nucleophilic part which can give…
Q: 4. An enzyme (acetoacetate decarboxylase) catalyzes the decarboxylation of acetoacetate. The…
A: Decarboxylation is the chemical reaction by which CO2 is released through the removal of carboxylic…
Q: The generic metal A forms an insoluble salt AB(s) and a complex AC, (aq). The equilibrium…
A:
Q: In a correctly recorded measurement of 27.5 °C all of the digits are certain, meaning the true…
A: Generally, all the measured values have some degree of uncertainty. In a physical measurement, the…
Q: Which of the following salts will result in basic solution in water? A. NaCl B. Na2S C. BaCl2 D.…
A: A basic solution in water refers to a solution with a pH greater than 7, indicating the presence of…
Q: Parts of an atom worksheet Complete the following table for each atom. Then draw the Bohr-Rutherford…
A: The objective of the question is to complete the given table regarding the parts of an atom.An atom…
Q: Identify the correct dipole for the C-N bond in the Lewis structure of hydrogen cyanide shown using…
A: The objective of the question is to identify the correct dipole for .
Q: Give detailed Solution..show work..don't give Handwritten answer..don't use Ai for answering…
A: The objective of the question is to calculate the half-life of a zero-order reaction. The half-life…
Q: Predict the major organic product for the following multistep sequence. Be sure your answer accounts…
A: The Gilman reagent contains two alkyl groups, copper and lithium. The Gilman reagent selectively…
Q: Which statement about the charge-minimized Lewis structure of azide (N3) is true? (Charges not shown…
A:
Q: Identify the number of stereoisomers expected for the following: Save for Later OH OH
A: The required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: When a 17.1 mL sample of a 0.353 M aqueous hypochlorous acid solution is titrated with a 0.401 M…
A: Given ,(a) Determine pH at mid point in titration.(b) Determine pH at equivalence point.(c)…
Q: Question 3: Reaction conditions are given below. Draw the product of each step. Mechanism arrows are…
A: The objective of the question is to predict the products formed in the following reaction given.
Q: 4. A 500 ml solution of KOH was found to have a pH of 12.55. Calculate the concentration of [OH] in…
A: The pH value of a solution can be used as a measure of the concentration of hydronium ion (H3O+)…
Q: Use solubility to calculate Ksp- The solubility of Co(OH), is measured and found to be 3.76*10^-4…
A: The objective of the question is to calculate the solubility of .The given values:The value of is…
Q: 3. Calculate the pH of a 1.94x103 M solution of CH COOH at 25°C. K, 4.55x100
A: Concentration of acetic acid(CH3COOH) = 1.9410-3 MKa of CH3COOH = 4.5510-10
Q: Arrange the following substances according to their increasing acidity a) c) Fenol,…
A: The objective of the question is to arrange the given substances in the order of their increasing…
Q: Identify the missing reagent(s), reactant(s) or final product(s). Some transformations may require…
A: Given are organic synthesis reactions. All reactions involve different types of reactions.
Q: What is the major product for the following reaction? TBr2. CCl4 O Br Br St Br Brenantiomer +…
A: The addition of bromine to an alkene in a nonpolar solvent produces vicinal dibromo alkane.
Q: Which is the major organic product formed by this reaction? Studying the summary chart in Lesson…
A: -> NaBH4/CH3OH is reducing agent which can reduce carbonyl compounds(aldehydes and ketones) to…
Q: Arrange the following compounds in order of increasing lattice energy (weakest to strongest), so…
A: The enthalpy change during the formation of one mole of ionic crystal from cations and anions is…
Q: CI CI IZ
A: Given is organic synthesis reaction. The given starting compound is pyridine derivative.
Q: What CO2 partial pressure (torr) is required to yield a velocity of 0.045 M-s1 for the reaction?…
A: The velocity is given as v = 0.045 M/sIt is required to find the pressure in torr.
Q: Consider the following reaction that takes place in a closed 2.0L container: N2(g) + 202(g) 2 NO2(g)…
A: The objective of the question is to calculate the equilibrium concentrations of nitrogen and oxygen.
Q: (2) The reaction-rate data given below were obtained using a batch reaction system for the reaction…
A: TimeCA (g/m3)0300.525.4121.6216.9413.188.3163.320.31From the given reaction rate data and for the…
Q: Describe the components required for the synthesis of this dihydropyridine, MeO. EtO₂C. ZI CO₂Me
A: The objective of the question is to identify the components required for the synthesis of a specific…
Q: A monoprotic acid, HA, with a Ka of 3.65 × 10-5 has a distribution constant (KD) of 2.51 (favoring…
A: The distribution ratio (D) of a monoprotic acid can be calculated using the Ka value, distribution…
Q: Please don't provide handwritten solution ....
A: The objective of the question is to calculate the equilibrium constant in terms of concentration…
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Use a dash or wedge…
A: It involves an elimination reaction - E2 (bimolecular elimination) reaction. This reaction proceeds…
Q: If the rate of disappearance of H2 is 3.3 M/s, what is the rate of disappearance of Fe3O4? Answer to…
A: To determine the rate of disappearance of Fe3O4, we need to consider the balanced chemical equation…
Q: Give the products of periodic acid oxidation of each of the following. How many moles of reagent…
A: The objective of the question is to determine the products of periodic acid oxidation of the given…
Q: Part A OH Spell out the full name of the compound. hept-2-en-4-ol Submit Previous Answers Request…
A: (2E)-hept-2-en-4-olExplanation:
Q: What is the synthesis for a compound below?
A: Given is organic synthesis reaction. In given reaction, the product has two extra carbon than the…
Q: 1. Choose the best conditions to make the following aldol product
A: Given is Aldol condensation reaction. In aldol condensation reaction, aldehyde or ketone reacts in…
Q: Suppose a 250. mL flask is filled with 0.80 mol of CH 4, 1.6 mol of H2S and 1.0 mol of H2. This…
A: Answer:Molar concentration of gas in a gaseous mixture is always equal to the ratio of its number of…
Q: Suppose a 500. mL flask is filled with 0.80 mol of O2 and 0.30 mol of NO. This reaction becomes…
A: The objective of the question is to find the initial, change, and equilibrium molarity of each…
Q: + Propose a synthesis for the following compounds a) b) additional building blocks must contain no…
A: The given reactions are a) Benzene is converted to 5-phenylpent-4-yn-2-oneb) 4-hydroxybutanal is…
Q: Starting from the Newman projection below, rotate the back carbon to provide the structure in the…
A: We have to choose the most stable Newman projection.
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CI H₂O pyridine 0
A: The objective of the question is to find the major product of the given reaction substituted acid…
Q: What is the major product? HNO3 Sn ? HC1 H₂SO NO2 NH2 NO₂ 8628 b C NO₂ d
A: ->Aromatic electrophilic substitution reaction in which an electrophile replaces a hydrogen atom…
Q: Draw and write down the expanded formula (see previous question for expanded formula) for the…
A: In an expanded molecular formula, all the atoms and bonds within the molecule are shown.
Q: Provide a reasonable reaction mechanism for these reactions 1) 1) excess NaBH4 2) HCI, H₂O OH HO 2)…
A: An arrow always depicts from a region of high electron density to low electron density; the tail…
Q: Consider a system that absorbs 225 kJ of heat and performs 96 kJ of work on the surroundings at…
A:
Q: What is the major organic product obtained from the following reaction? OH H₂O CH3CH2CH2CH2C=CH…
A: The given reaction is an example of oxymercuration-demercuration, which is a type of electrophilic…
help me with part 2, 4, 5 please
Step by step
Solved in 1 steps with 1 images

- why is this S not R(I means the one that I circle in red color) 123 or 134???if go 123 it would be R,if it goes 134,it would be s?so confusing?Can anyone make it clear?Please help and explain in detailsWhich amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]- only ii i and iii only i ii and iv only iiPlease explain how to solve the following NMR:
- How do you assign R or S to a molecule when the lowest-priority group is not oriented toward the back, on a dashed wedge?Can you explain spectroscopy? Let's start with NMR Spectroscopy. What is the difference between a proton being enantiotopic and homotopic? What does it mean for a proton to be shielded or deshielded? How do you determine J (the coupling constant)? What does 'coupling is mutual' mean? How is that translated on the NMS Spectroscopy spectrum? Does that have to do with the polarity of the molecule? Is there an easier way to explain that? When thinking about C-13 NRM Spectroscopy, what it meant by the statement, "the carbons cannot integrate"? When looking at an NMR Spectroscopy spectrum, how do you know which peak belongs to which proton? (The intensity of the peak and the ppm of the peak) P.S. These questions come from after watching a lecture. There is no graded assignment attached to these questions. I simply do not understand the material.C13 HNMR Spectroscopy, please solve this problem and explain it..
- Determine which protons refers to each peak shown on the graph below. (there are 7 of them) ( plz do completely in detain plzz .given handwritten solution in A4 size paper , plz give ans in detail )Why is it that D hydrogens have a higher chemical shift than the E hydrogens? Aren't the D hydrogens less shielded than the E hydrogens which have carbons surrounding on both sides? And less shielding means higher chemical shift required?NMR HW help . which one is correct structure that match the NMR graph and explain why
- 1 Can one distinguish between the Markovnikov and anti-Markovnikov complement products I and II byinfrared spectroscopy? How about nuclear magnetic resonance? Justify your answersI need help finding the functional groups and the wavelegth for the coumpound and to draw the is spec based on the wavelegth and label them on the compund and ir spec I linked and example of one that i already did and it needs to look like itGive a to d answer plz ?

