3. Considering the techniques and instrumentation used to perform these experiments, identify 2 potential sources of error if you were to repeat this experiment in the "real world". Human error is not an acceptable answer. Discuss at least two possible sources of error that could contribute to your percent error. A student is performing Reaction 2 and does not completely dissolve the NaOH pellets before acquiring the final temperature. How will this affect his value for AH, and his percent error calculations?
3. Considering the techniques and instrumentation used to perform these experiments, identify 2 potential sources of error if you were to repeat this experiment in the "real world". Human error is not an acceptable answer. Discuss at least two possible sources of error that could contribute to your percent error. A student is performing Reaction 2 and does not completely dissolve the NaOH pellets before acquiring the final temperature. How will this affect his value for AH, and his percent error calculations?
Chapter2: Crystallization
Section: Chapter Questions
Problem 3Q
Related questions
Question
Transcribed Image Text:00 P
and Hei
3. Considering the techniques and instrumentation used to perform these experiments, identify 2 potential sources of error if you
were to repeat this experiment in the "real world". Human error is not an acceptable answer. Discuss at least two possible
sources of error that could contribute to your percent error.
4. A student is performing Reaction 2 and does not completely dissolve the NaOH pellets before acquiring the final temperature.
How will this affect his value for AH, and his percent error calculations?
Transcribed Image Text:Meme
X
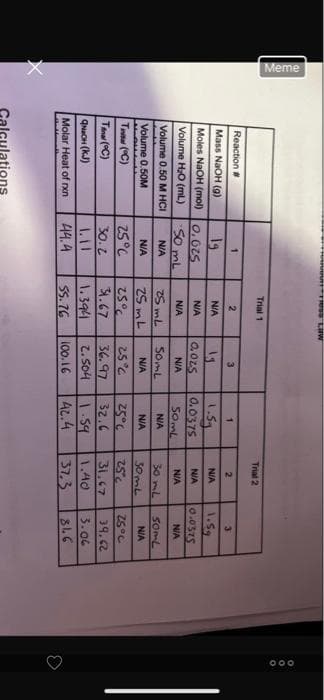
Reaction #
Mass NaOH (g)
Moles NaOH (mol)
Volume H₂O (ml)
Volume 0.50 M HCI
Volume 0.50M
Ta (°C)
Tina (°C)
QNaOH (KJ)
Molar Heat of rxn
ilations
19
0.625
50 mL
N/A
N/A
25°C
30.2
44.4
TF10ss w
Trial 1
2
N/A
N/A
N/A
25 mL
25mL
25%
31.67
1.394
55.76
3
13
0.025
N/A
50mL
N/A
25°C
36.97
2.504
100.16
1
1.5₂
0.0375
50mL
N/A
N/A
Trial 2
25°C
32.6
2
N/A
NIA
N/A
30 mL
30mL
25%
31.67
1.59 1.40
42.4
37.3
3
1.5g
0.0575
N/A
50mL
NIA
25°C
39.62
3.06
816
ooo
O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT