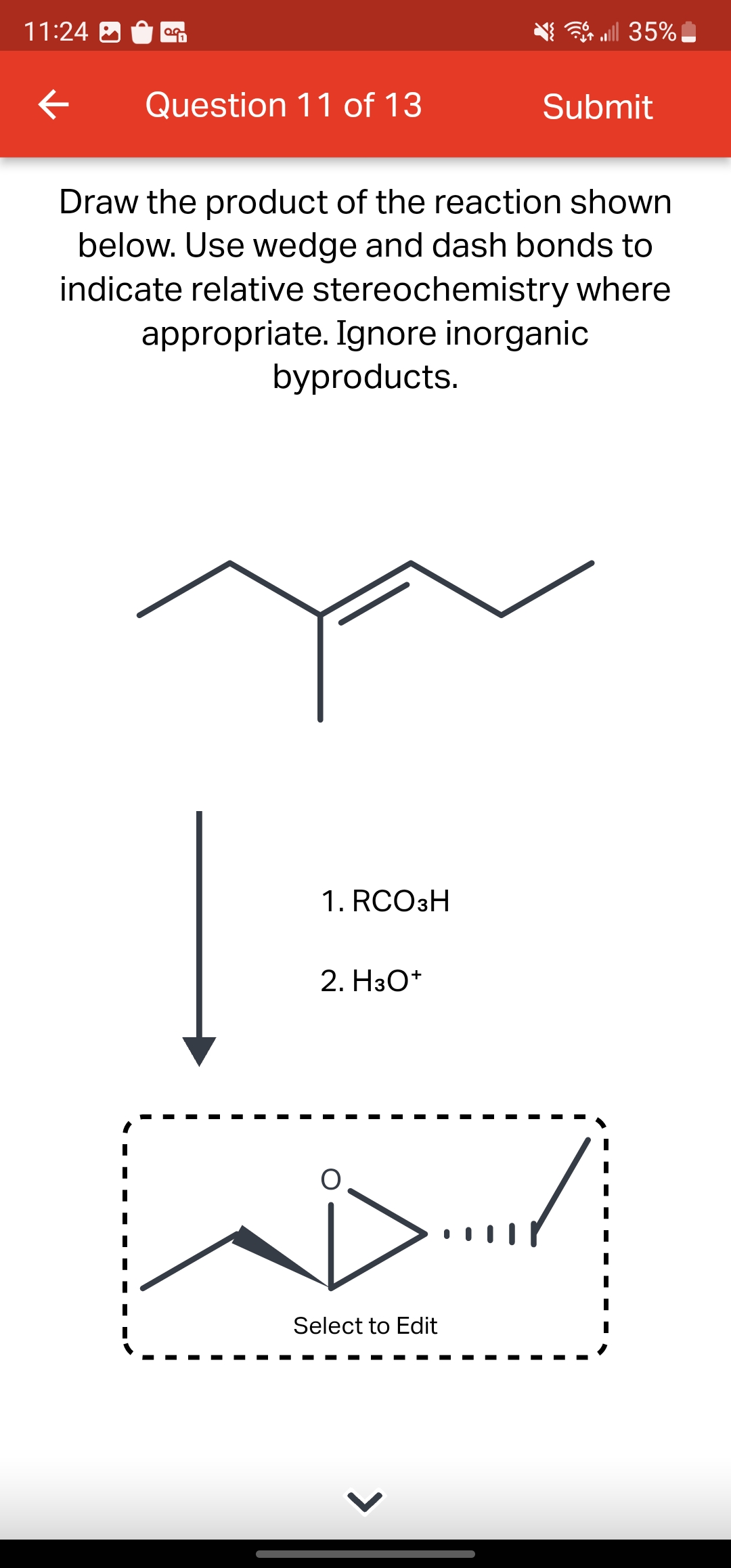
Draw the product of the reaction shown below. Use wedge and dash bonds to indicate relative stereochemistry where appropriate. Ignore inorganic byproducts. 1. RCO3H 2. H3O+ I I I I
Q: A chemist designs a galvanic cell from the following two half-reactions NO3 (aq) + 4H+ (aq) + 3e →…
A:
Q: Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic…
A: we have to fill the given table on the basis of polarity of the molecules
Q: What is the reason for higher Capillary Electrophoresis plate number compared to Liquid…
A: Capillary Electrophoresis is an analytical separation method. The sample is introduced into a…
Q: reaction. Use wedge and dash bonds to indicate stereochemistry where appropriate. Ignore inorganic…
A: -> When Br2 reacts with alkene first of all cyclobromonium ion formed then Br- can open the ring…
Q: 0 OH O || --CH-C H --0 NAD*
A: The Concept used here is from oxidation and reduction reactions Here we have... NAD+ which is an…
Q: Show how the following molecule could be "cut" into fragments for the Mass Spectrum. Cut the…
A: Mass spectrometry is one of the best techniques for analyzing the structure of a molecule and…
Q: Calculate the energy for the transition of an electron from the n = 6 level to the n = 1 level of a…
A:
Q: Below are rate constants for the respective outer-sphere redox reactions between the complex in the…
A: The concept used here is from Co Ordination chemistry. Here we have the reaction of [ Co(NH3 ) 5 I…
Q: A first order reaction initially contains 1.20 × 10²⁰ molecules. If the reaction has a half-life of…
A: The process of disintegration of unstable nuclei to stable nuclei and emission of alpha , beta and…
Q: Use a bond energy table to determine the amount of heat energy released per mole of propane when it…
A:
Q: CHEM 101 1- A gas known to have a formula of NOx was found to effuse at a rate that is 0.834 times…
A: Graham's law:
Q: A chemistry graduate student is studying the rate of this reaction: 2C1₂O5 (g) → 2C1₂ (g) +50₂ (g)…
A:
Q: "
A:
Q: A Lewis structure for NCS- is shown below, however, its formal charges are not minimized. Starting…
A:
Q: Hydrogen atom abstraction by a bromine radical in bromination of an alkane would be fastest with?
A: We have to answers the following questions. Introduction: During radical bromination, a carbon…
Q: balancing coefficients
A: How to balance
Q: fod + H₂O-> a. Draw the structure of the tetrahedral intermediate INITIALLY FORMED in the reaction…
A: Acid anhydride reacts with water to give corresponding carboxylic acids. O-atom of a molecule of…
Q: What is a correct cell notation for a voltaic cell based on the reaction below? Cu2+(aq) + Pb(s) +…
A:
Q: Use the Born-Haber cycle and the data shown to calculate the lattice energy of SrO. Express your…
A: This question is based on calculation of lattice energy by using Born -Haber cycle. In this question…
Q: Show all the steps and labels for full credit. 8 Propose a structure for the following: C6H13Br. NMR…
A:
Q: Consider the mechanism. A B + C Step 1: Step 2: C + D E Overall: A+ D→ B+E Determine the rate law…
A: The sum of the exponent term in the rate law for a chemical reaction is known as the order of the…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A:
Q: In the following acid-base titration experiment HNO3 (aq) + KOH (aq) ---------> KNO3 (aq) + H2O(l)…
A:
Q: Draw the structure of the following molecules in skeletal format i) cycloheptane and write its…
A: These are simple organic molecules ,some are linear and some are cyclic , stereoisomers are also…
Q: what would need to happen if you wanted to melt NaCl?
A:
Q: What is an advantage of Gas Chromatography and Mass Spectrometry (GS/MS). a. It is time…
A: Both a and b are disadvantages of Gas Chromatography and Mass Spectrometry (GS/MS) Option d is also…
Q: ▼ Part A The forensic technician at a crime scene has just prepared a luminol stock solution by…
A:
Q: 16.22 What type of interaction would you expect between the R groups of the following amino acids in…
A:
Q: The following reaction occurs spontaneously. 2 Fe(s) + 3 Cl2(aq) → 2 Fe3+(aq) + 6 Cl–(aq) Write the…
A:
Q: How much heat (in kJ) is needed to convert 10.0 grams of H₂O(l) at 99.97°C to H₂O(g) at 99.97°C?…
A:
Q: It is known that vitamin C decomposes upon the exposure to air or heating. Briefly describe how you…
A: Answer:-
Q: At a unspecified temperature we place 0.940 mol of pure SO3 is placed in a evacuated 1.00-L…
A:
Q: can you answer question 2
A: In this , we would determine the oxidizing strength from the given two observations. Then combine…
Q: Listen If an excited electron drops from a higher shell to a lower shell in an atom, the energy is…
A: When an electron excites from lower energy state to higher energy state, it absorbs energy of…
Q: What kind of intermolecular forces act between two carbon dioxide molecules? List all intermolecular…
A: we have to determine the intermolecular forces present in CO2
Q: In this titration experiment, what are your two titrants (titrating solutions)? iron solution with…
A: Titration:- it is an analytical technique which is used for the determination of the unknown…
Q: Q21. How many litres of water will have to be added to 1125 litres of 45% solution of acid so that…
A:
Q: (g) The element whose +2 cation has the same electronic configuration as krypton. (h) The lightest…
A:
Q: A B C D 1. BH3-THF 2. H₂O2, NaOH 1. Hg(OAc)2, H₂O 2. NaBH4, NaOH mCPBA mCPBA 2. H3O+
A:
Q: Using the data in the table, calculate the rate constant of this reaction. k = A + B C + D Trial [A]…
A: Rate equation is represented by.... Rate = K [A]x . [B]y
Q: 6. Determine the structure of the following compound. To get full credit you must: Calculate the…
A:
Q: Provide the IUPAC name for each of the following acid anhydrides. (a) (c) ململة للا سلملي (b)
A: Symmetrical acid anhydrides are named like carboxylic acids but the ending -acid is replaced with…
Q: Write the rate law for the two elementary reaction equations. 2 A(g) + B(g) C(g) + D(g) rate = k…
A: The rate law for an elementary reaction can be derived from the coefficients of the reactants in the…
Q: In a wire, 7.23 x 1020 electrons flow past any point during 3.57 s. What is the magnitude I of the…
A:
Q: We can draw three inequivalent Lewis structures for the selenite ion, SeO3²- . The concepts of…
A: We know, the tendency of an atom in a molecule to attract the shared pair of electrons towards…
Q: What phenomenon enables outer-sphere redox reactions to occur? Group of answer choices relaxation of…
A: Electron transfer (ET) occurs when an electron relocates from an atom or molecule to another such…
Q: B. Reactions in Solution Consider the reaction that occurs when 75 mL of 1.2M sodium sulfate is…
A: A question based on stoichiometry. A precipitation reaction between Na2SO4 and Ba(NO3)2 is…
Q: For which of the following complexes is it possible to react with [Ru(NH3)6]2+ via an inner-sphere…
A: Since we know that in inner sphere mechanism complex must have bridging ligand...
Q: The standard entropy for the formation of SF6(g) from the elements, S(s) + 3 F2(g) → SF6(g) is…
A: As we know, The standard entropy for a reaction can be calculated S(s) + 3 F2(g) → SF6(g)
Q: precute Identify whether the sign of AH is positive or negative for each of the following. Explain.…
A: Enthalpy change ( ΔH) is defined as the amount of heat evolved or absorbed in a reaction. A negative…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Describe Suprafacial and Antarafacial Rearrangement:The molecular formula of unknown compound B is C10H16.Quantitative brominationof a 0.473-g sample of compound Brequired 48.22mL of a 0.216MBr2/CCl4to produce a color change. How many rings and pi bonds does compound Bcontain?Include any explanations/calculations to justify your answers.
- Stereoisomers of α-bromocinnamic acid: draw the reaction mechiansim with arows for the dehydrobromination of 2,3-dibromo-3-phenylpropanoic acid with ethanol and KOH to produce (Z)-α-bromocinnamic acid and (E)-α-bromocinnamic acid. Also draw the transition state for The (Z)-isomer syn-periplanar transition state and anti-periplanar transition state of the (E)-isomer.Fill in the missing reactants, reagents and products in the following reactions. Indicate stereochemistry if necessary. Unless otherwise specified, assume the reagents are in excess. PLease explaion with MUCH detail.One possible way of determining the identity of an alkene, is to let itundergo an oxidative cleavage reaction in the presence of hot basicpotassium permanganate. You are given two containers said to containdifferent alkenes. Container A is marked as cis / trans‐2‐butene andcontainer B as 2‐methyl‐1‐butene. Explain by referring to the formation ofproducts, how you would verify the identity of the alkenes.
- Using the Woodward–Hoffmann rules, predict the stereochemistry of belowreaction. a [6 + 4] thermal cycloadditionDiisopinocampheylborane (Ipc2BH) is a chiral organoborane, readily employed for the production of many asymmetric products used in total synthesis. It is a crystalline material that can be prepared as a single enantiomer via the hydroboration of two equivalents of α-pinene with borane.Explain why only one enantiomer of Ipc2BH is formed.Under basic conditions, the following substrates can undergo a [4+2] cycloaddition with regio- anddiastereo- control. Rationalize mechanistically how the reaction occurs as well as the regio- and stereochemical outcome with each base.
- Write structures for products A, B, C, and D showinf stereochemistry. (Hint: B and D are stereoisomers)Hydrocarbon 10 and ester 12 react upon heating to give a cycloaddition product D. (i) Suggest a structure for product D and provide a curved arrow mechanism that accounts for its formation. (ii) Draw the molecular orbitals corresponding to the HOMO of 10 and the LUMO of 12 and show that these are of suitable symmetry for a concerted cycloaddition to take place. (iii) Use frontier molecular orbital theory to account for the observation that the formation of D is faster in the presence of AlCl3. please answer all thank youPredict-the-product. Draw the structure(s) of the major organic profuct(s) formed in the following reactions. Please indicate stereochemistry if appropriate.

