Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter26: Synthesis And Degradation Of Nucleotides
Section: Chapter Questions
Problem 16P
Related questions
Question
Please complete the table
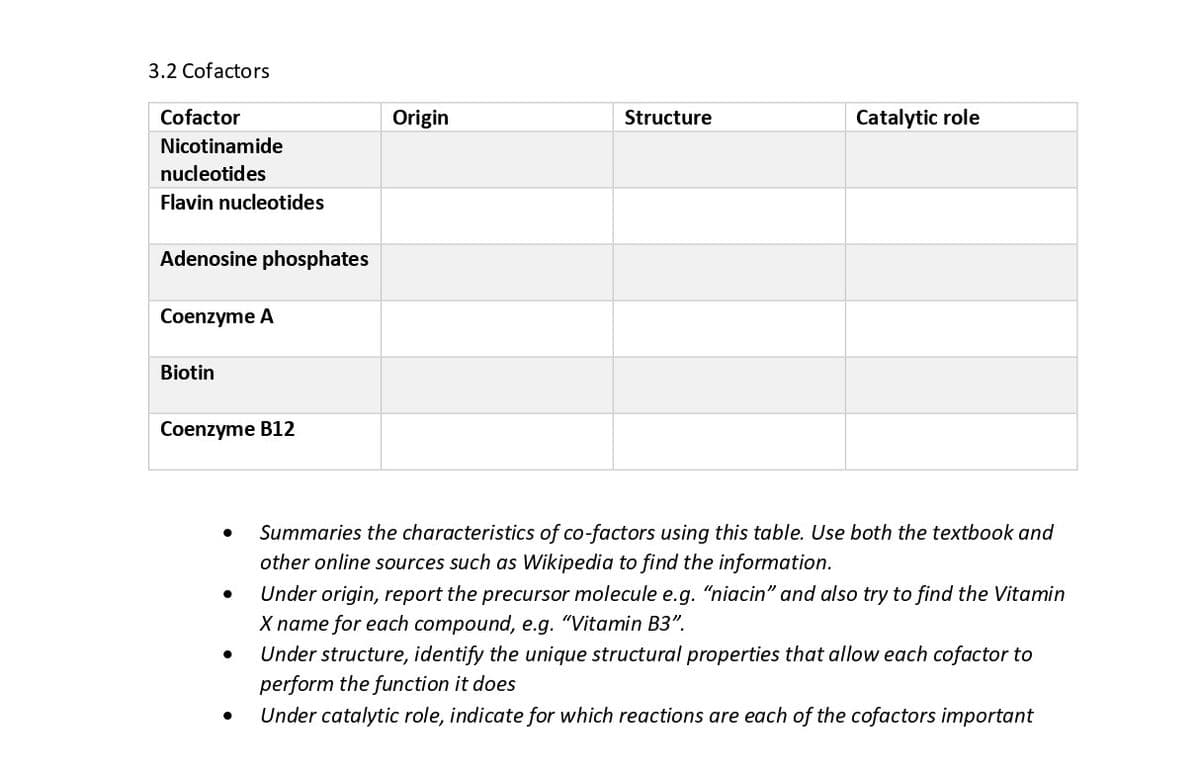
Transcribed Image Text:3.2 Cofactors
Cofactor
Origin
Structure
Catalytic role
Nicotinamide
nucleotides
Flavin nucleotides
Adenosine phosphates
Coenzyme A
Biotin
Coenzyme B12
Summaries the characteristics of co-factors using this table. Use both the textbook and
other online sources such as Wikipedia to find the information.
Under origin, report the precursor molecule e.g. "niacin" and also try to find the Vitamin
X name for each compound, e.g. "Vitamin B3".
Under structure, identify the unique structural properties that allow each cofactor to
perform the function it does
Under catalytic role, indicate for which reactions are each of the cofactors important
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning