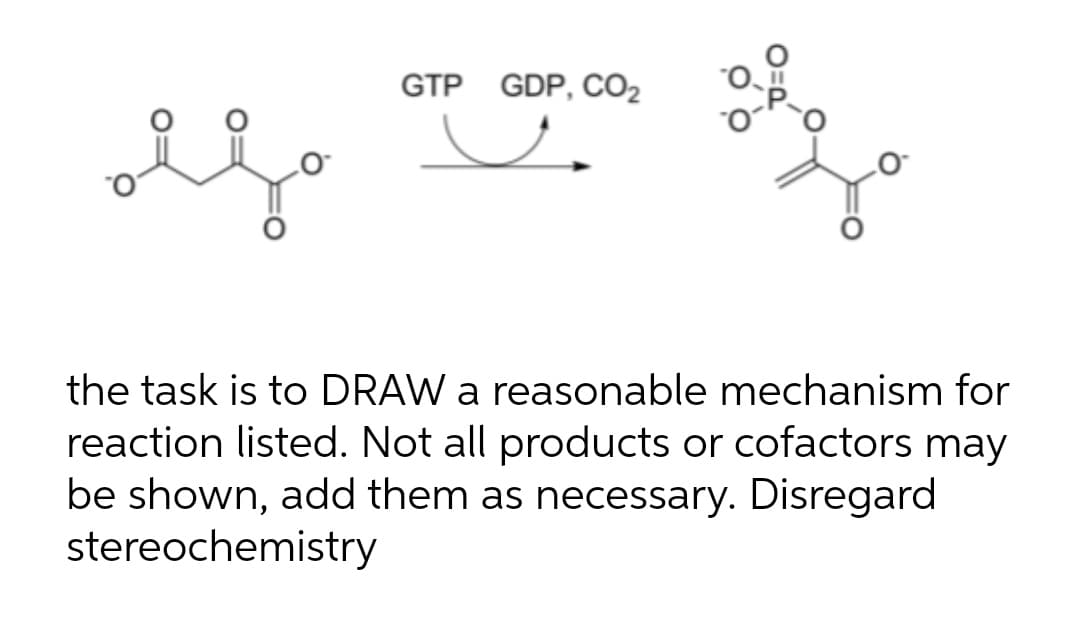
GTP GDP, CO2 the task is to DRAW a reasonable mechanism for reaction listed. Not all products or cofactors may be shown, add them as necessary. Disregard stereochemistry
Q: 5-Polymerization of methylmethacrylate monomer in alkali metals dispersed in liquid ammonia, THF…
A: Polymerizations of methyl methacrylate initiated by potassium and sodium amides in liquid ammonia…
Q: 14.Which explanation best fits the graph for pH's effect on an enzyme? Opi p O A. As the pH gets…
A: Each enzyme performs optimally at a certain pH level. The optimal pH for an enzyme is determined by…
Q: Biological Molecules– ModellingActivity AC1.1-lustrate or construct molecular models of simple…
A:
Q: Catalysis isa. restoring secondary structure.b. denaturation in reverse.c. protein enlargement.d.…
A: Catalysis is the speeding up of a chemical reaction by a chemical species.
Q: lomolecular laboratory, identify reaction that you ave certainly observed with the enzyme Lyase. ·…
A: The substrate binds to the enzyme's active site and is converted to the product. Enzymes alter…
Q: Select all that apply. The nylon synthesis reaction performed in this lab is actually occurring: in…
A: Nylon is a polymer synthesized through polymerization reaction.
Q: ?? = ??A + log [? -]/[??] An enzyme uses histidine as a general acid catalyst. If the active site…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: Describe in your own words, 5 assumptions that must be made in order to apply a Michaelis-Menten…
A:
Q: Protein,nfalded==> Proteinalded Under certain conditions, a specific protein has the following…
A: Proteins have four levels of the structural organization including Primary, secondary,…
Q: Połymer formation from a pool of available monomers with a range of chemical functionalities…
A: Introduction:- The quetion is about the biomolecules that are polymers that made up of small units…
Q: B-mercaptoethanol (BME) is used as a reducing agent in biochemistry labs. The structure below…
A: β mercaptoethanol is a reducing agent used to denature proteins by breaking the disulfide bonds…
Q: 1. Requirements of Active Sites in Enzymes The active site of an enzyme usually consists of a pocket…
A: Peptidases are enzymes that cleave peptide bonds and peptides. Carboxypeptidases are peptidases…
Q: 3. Trypsin selectively cleaves polypeptide chains on the carbonyl side of the amino acids arginine…
A: Enzymes are proteins that accelerate the rate of biochemical reactions. Proteins are polymers of…
Q: Search for a structure of one common drug that is available locally. Draw the structure of the drug…
A: Xenobiotics are chemicals which enter into the body. Xenobiotics are eliminated from the body…
Q: Calculate the number of ATPS produced from the complete oxidation of a TAG containing two caproic…
A:
Q: Activity 2: Enzyme Structure 1. Use the following terms to label the diagram below: Prodect…
A: Several metabolic pathways exist in the human body. Each pathway enables catalysis of the chemical…
Q: Proteins are thermodynamically unstable. The ΔG of the hydrolysis of proteins is quite negative, yet…
A: Proteins are macromolecules made up of individual amino acid monomers. Proteins are synthesized…
Q: Biomolecules' three-dimensional structure is more evolutionary conserved than their sequence. Why?
A: Biomolecules are macromolecules that are produced by living organisms and perform the major…
Q: In the structure of Alkaline Phosphatase enzyme what is the distance between the metal ion and the…
A: Alkaline phosphatase This refers to the homodimeric protein enzyme which is about 86 kilodaltons.…
Q: Cofactor Origin Structure Catalytic role Nicotinamide nucleotides Flavin nucleotides Adenosine…
A: Cofactor is the non-protein part of the enzyme. Cofactor is required for the biological activity of…
Q: What is edman degradation and why does no reaction occur when sungsanpin was subjected to edman…
A: Edman degradation is a method for sequencing of polypeptide chains.
Q: 22. The biochemical reaction shown below represents the complete digestion or breakdown of a polymer…
A: The monomers are the molecules that combine with each other to form the polymers. Due to this,…
Q: 1 Active Site Reaction Mechanism Inhibitor HN- 2•Zn NH2 HO. HO, NH2 N N- NH `NH NH NH, N' 'N N 'N…
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the…
Q: 31. The velocity of a particular enzyme-catalyzed reaction varied with pH as shown in the adjacent…
A: Enzyme activity is at its maximum value at the optimum pH. As the pH value is increased above or…
Q: 8. Phosphatases hydrolyze phosphate-ester bonds in all types of phosphate esters. Which of the…
A: Enzymes are biocatalyst. They increase the speed of reaction by lowering its activation energy.…
Q: ENZYME CLASSIFICATION BIOCHEMICAL PROPERTIES | COMMIS SION 2 3 4 5 6.
A: Almost every biochemical reaction is catalyzed by an enzyme. With the exception of a few catalytic…
Q: Suppose two enzymes in different reaction pathways have the same substrate but have different level…
A: An enzyme is a type of biological catalyst that aids in the acceleration of chemical reactions.…
Q: Describe the reaction oh Hatch-Slack (enzyme names are not required). While doing so make sure to…
A: C4 Pathway It was Hatch and Slack , who discovered the C4 pathway in 1966. Plants that are adapted…
Q: In human beings, what is the major control of de novo pyrimidine nucleotide synthesis? A. substrate…
A: De novo synthesis of pyrimidine nucleotides takes place in cytosol, first the pyrimidin is…
Q: the major components of complex biomolecules. For each of the two important biomolecules below…
A: GTP is an energy-rich molecule, just like ATP. Usually when it is hydrolyzed, the free energy of…
Q: H2C HO CH2 H. N. CH H2C. Enzyme X is an aspartyl protease. Here is the tetrahedral intermediate in…
A: Aspartyl proteases are proteases that contain two conserved aspartic acid residues at the active…
Q: IX. Insulin, a hormone vital in blood sugar regulation and having a polypeptide chain with disulfide…
A: Insulin is a hormone responsible for the regulation of sugar levels in the blood. Insulin hormone is…
Q: A: State the Lineweaver Burke equation. B: Calculate the kinetic parameters Vmax and KM from the…
A: Parameters such as Km and Vmax are used for comparing enzyme activities. If we know the initial rate…
Q: Biochemists have become increasingly interested in using organic reactions to modify proteins…
A: Russian chemist Vladimir Markovnikov studied the addition of HBr to numerous different alkenes in…
Q: 1. Chymotrypsin is a digestive enzyme that breaks down proteins. From the following attributes,…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want a…
Q: ppp 26. Why aren't strong bonds as useful for work in the cell? 27. Define chemical equilibrium with…
A: Since we are entitled to answer only one question at a time, I would be answering the ques. 26. If…
Q: Suppose two enzymes in different reaction pathways have the same substrate but have different level…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: Functional groups and linkages assignment Complete the reactions below. Circle and identify all…
A: The hydrocarbon compound is N-butylpropanamide which is reacting with water.
Q: III. Protein: QQICIMFELTQISS Predict the products of the following reactions with the protein given,…
A: Introduction Protein: QQICIMFELTQISS Aminoacids: Gln, Gln, Ile, Cys, Ile, Met, Phe, Glu, Leu, Thr,…
Q: 2. Characteristics of enzymes: a. One characteristic of enzymes is that they are reusable. This is…
A: Enzymes are biological catalysts that are proteins involved in speeding up the chemical reactions in…
Q: Under which of the following conditions could a biochemical reaction inside a cell have a positive…
A: Under constant temperature and pressure, the relationship between free energy change of a…
Q: Km value of an enzyme
A: The study of the rates of enzyme-catalyzed chemical reactions is known as enzyme kinetics. here they…
Q: Other substrate(s) Other product(s) Enzyme(s) Po,c. он NH HO он он CH,OPO, OPO,
A: Glycolysis is a metabolic pathway which converts the glucose C₆H₁₂O₆, into the pyruvate, CH₃COCOO⁻,…
Q: Energy Energy 2. For both of the graphs below, (A) draw a new blue line showing what the reaction…
A: exergonic biochemical reactions are high energy reactions where the reactants have higher energy…
Q: P3D.2 In biological cells, the energy released by the oxidation of foods is stored in adenosine…
A: The values provided in the questions are: T = 310 K H (enthalpy change) = -20 kJ.mol-1 G = - 31…
Q: 11. An enzyme requires a dinucleotide cofactor (shown below). Using the given letters, mark one…
A: Deoxy sugars are sugars that have hydrogen atoms instead of the hydroxyl group eg. deoxyribose is a…
Q: the substrates must be a high erall reaction is exergonic
A: Answer Substrate level phosphorylation in which One of the substrates must be a higher free energy…
Q: BONUS: JA transition state can decay either to products or substrates. What factor determines which…
A: The state of transition is the transitory molecular structure in which the molecule is no longer a…
Q: multiple choice, choose the correct answer Under which of the following conditions would an enzyme…
A: The enzyme is protein molecules that increase the rate of the reaction by decreasing the energy of…
4
Step by step
Solved in 2 steps with 1 images

- Cyanide ion (CN-) causes toxicity to humans, explain the reason of its toxicity to us. Suggest a method to remove its toxicity from the blood.The structure of a metalloenzyme active site is down below(black picture). Describe, from a chemical and structural perspective, how the reactive site is designed to facilitate its catalytic reaction. The example below(white pitcure) suggests the level of detail that is required. Make sure that you explain what the metal is doing, what the reaction is, and its biological significance.Suggest an appropriate reason for the observation : “On increasing temperature of the reacting system by 10 degrees, the rate of reaction almost doubles or even some times becomes five folds.”
- The structure of a metalloenzyme active site is down below(black picture). Describe, from a chemical and structural perspective, how the reactive site is designed to facilitate its catalytic reaction. The example below suggests the level of detail that is required. Make sure that you explain what the metal is doing, what the reaction is, and its biological significance.Predict the mechanism as SN1, SN2, E1 or E2 and draw the major organic product formed in each reaction. Consider any regioselectivity and stereoselectivity where applicable: I’d be grateful if you helped with all of these but if not can you please help me answer f,g,h & j ?True or False Immobilization improves the stability of the enzyme. EnaLne, has a half-life of 10 days in free solution, but under identical conditions of temperature, pH, and medium composition, the measured half-life of a packed column is 30 days. The enzyme is immobilized in a porous sphere 5 mm in diameter.
- When amphipathic biomolecules form micelles in polar solvents, this is an example of increasing entropy in the solvent True FalseThe three molecules in Figure Q2–2 contain theseven most common reactive groups in biology. Most mol-ecules in the cell are built from these functional groups.Indicate and name the functional groups in these mole-cules.An allosterically regulated enzyme in the R state is unable to bind substrate at the activesite. TrueFalse
- Label all 5 the indicted regions in this coenzyme (labels like site of redox activity, etc)An enzyme has a rate enhancement of 1.3x106. Calculate the value of ΔΔG‡ at 25.0 °C in kJ mol-1. (Hint: be sure to pay attention to units and signs.) (R = 8.3145 J mol-1 K-1)Understanding an Antidote for Cyanide Poisoning Sodium nitrite (NaNO2) is used by emergency medical personnel as an antidote for cyanide poisoning (for this purpose. it must be administered immediately). Based on the discussion of cyanide poisoning in Section 20.4. Suggest a mechanism for the lifesaving effect of sodium nitrite.



