н P OH `OH ÓH но OH write mechanisms for reaction shown by drawing each step. Not all cofactors may be indicated, use them as necessary. Disregard stereochemistry.
н P OH `OH ÓH но OH write mechanisms for reaction shown by drawing each step. Not all cofactors may be indicated, use them as necessary. Disregard stereochemistry.
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter2: The Chemical Level Of Organization
Section: Chapter Questions
Problem 39CTQ: AB+CDAD+BE Is this a legitimate example of an exchange reaction? Why or why not?
Related questions
Question
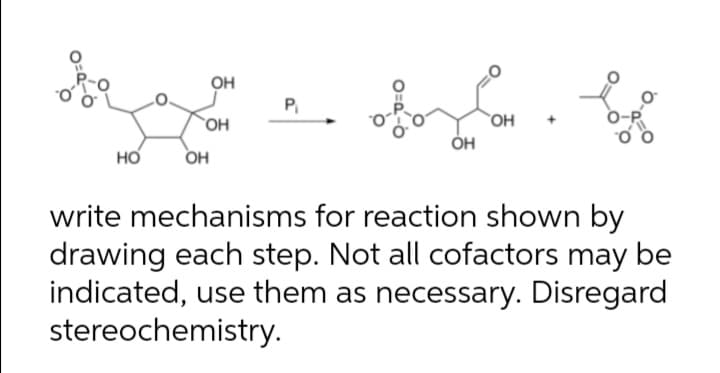
Transcribed Image Text:н
P
OH
`OH
ÓH
но
OH
write mechanisms for reaction shown by
drawing each step. Not all cofactors may be
indicated, use them as necessary. Disregard
stereochemistry.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College