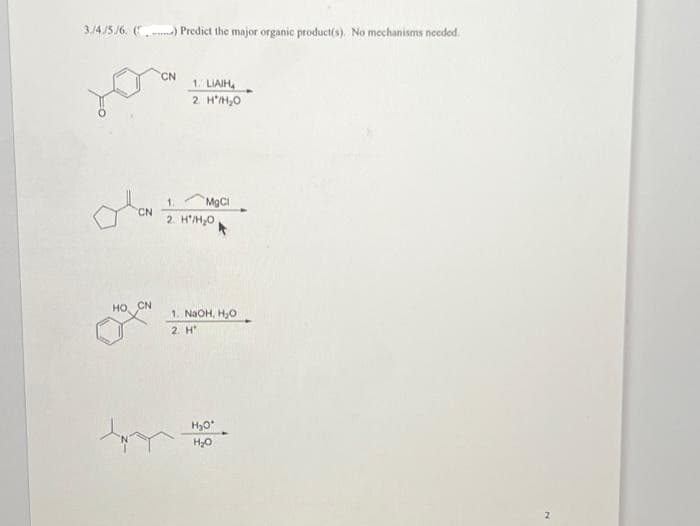
3./4/5./6. () Predict the major organic product(s). No mechanisms needed. CN HO, CN CN 1. LIAIH 2. H'H₂0 MgCl 1. 2. H'/H₂O 1. NaOH, H₂O 2. H H₂O' H₂O
Q: 1. Predict (draw) the ¹H NMR and 13C NMR spectra for each molecule. Your illustration should include…
A: 1) The 1H and 13C NMR spectroscopy will provide us information about with number of the signals in…
Q: Give the correct stereochemical indicator for the following alkene: None required ΟΕ ΟΖ Submit…
A: The given alkene is shown below.We have to determine the correct stereochemistry of the alkene.
Q: and after blowing air into it for Q1. The pH of unbuffered water before blowing exhaled air into…
A: A buffer is a solution which resists any change in pH by adding a small amount of acid or base. It…
Q: Find the wavelength in nm of electromagnetic radiation with a frequency pf 6.13 x 1014 s-1.
A: Frequency = 6.13 X 10 14 S-1We need to calculate the wavelength in nm of electromagnetic radiation.
Q: The pH of an acidic solution is 5.53. What is [OH-]?
A: The pH of the solution is = 5.53We know that:
Q: The molar absorptivity for the FD&C dye “Red No. 3” (Erythrosine) 8.25104 M‒1·cm‒1 at 526 nm.…
A:
Q: Problem 3: An empty sealed container has a volume of 0.652 L and a mass of 2.50 g. When filled with…
A: In the given question we have to calculate the molar volume of nitrogen gas at STP. We have to use…
Q: 5) Draw two reasonable resonance structures and the hybrid of the intermediate formed in the…
A: Given is organic reaction. This is allylic bromination reaction.
Q: In Einstein's photoelectric experiment, the kinetic energy of an electron displaced from a metal by…
A:
Q: The titration of 50.00 mL of 0.150-M HCl with 0.150-M NaOH (the titrant) is carried out in a…
A: The balanced equation for the reaction between HCl and NaOH is HCl + NaOH --------> NaCl + H2O…
Q: (a) he reagent(s) type yo Choose the reagents from the ones shown below (A-L). E للہ علہلہ پہلا میں…
A: Formation of beta keto ester
Q: 3. Propose a mechanism and product for the reaction below. -OH H₂SO 1800 4. Propose a synthesis for…
A: Dehydration of alcohol is acid catalyzed reaction which results the formation of alkene. Dehydration…
Q: The n+1 rule on the ¹H NMR spectroscopy predicts A) that a multiplet containing n lines will have n…
A: Also called proton spectroscopy.It is based on the atomic spin of Hydrogen-1 atom within the…
Q: Given the following values, calculate the mass of glycerol produced in the experiment. Please…
A: Molecular mass of glycerol trioleate = 885.432 g/mol. Molecular mass of glycerol = 92.09382 g/mol…
Q: Convert 4752 miles to meters
A: This question is based on conversion of units where given physical quantity needs to be converted…
Q: Measurements show that the energy of a mixture of gaseous reactants decreases by 209. kJ during a…
A: Decrease in Energy of gaseous reactant = ∆E = -209 kJ Work done = -104 kJ Change in enthalpy of gas…
Q: The gas phase reaction H2 + I2 → 2 HI is 2nd order. The rate constant at 400.0°C is 2.34×10–2 dm3…
A: The order of the reaction is second order and for the second order reaction rate law will be, Rate =…
Q: Hydrogen gas is produced when 295.0 mL of a 0.6399 M HCl solution reacts according to Zn(s) + 2…
A: Volume of HCl solution = 295.0 ml = 0.295 LitreMolarity of HCl solution = 0.6399 M and reaction…
Q: Predict the product for the following reaction. 000 || -||| ✓ Saved CI CN ∞∞ IV NaCN CN 111 CN ad CN…
A: The reactions in which an atom or a group of atoms are replaced by another atom or group of atoms…
Q: Based on the structures of lycopene and β-carotene, which are two chemicals found in another common…
A: According to Bartleby Q&A guidelines we are supposed to answer only first three parts. Since you…
Q: Br NO₂ NaO
A:
Q: The maximum amount of copper(II) hydroxide that will dissolve in a 0.292 M copper(II) acetate…
A: Given:Molarity of copper (II) acetate solution = 0.292 MSolubility refers to the maximum amount of a…
Q: pls anser all of them
A: Synthesis reaction: When two or more reactants react or get added to form product, then the reaction…
Q: Select the product/s for the following reaction: Br Br Br NBS DMSO (aq) Br Br Br ? Br OH Br IV OH
A: 1) The reaction of alkene with NBS in aq. solvent always leads to bromohydrins. 2) When the alkene…
Q: Figuur 3 + KMnO4 4 H₂O
A: Reaction of alkenes with 'cold' KMnO4 is stereospecific (cis addition) - Under 'cold' reaction…
Q: The answer is 0.45
A: The given redox reaction is the reverse oxidation reaction of iron to iron (II) by the metal X.So,…
Q: Multistep Problems: Avogadro's Number and Molar Mass 10. How many atoms of tin (Sn) are in 1.84 g of…
A:
Q: NaOH OHO < 1. BH-THF 2.H₂O₂, NaOH) HOCH₂CH₂CH₂ OH TSOH B C D < NaOH H₂O HOCH₂CH₂CH₂OH TSOH, H₂O…
A: Organic reaction: conversion of acetal to ketone.
Q: Determine the quantity of molecules in 1.40 moles of CO
A: The number of molecules or atoms present in the one mole of the substance is equal to the Avogadro…
Q: Draw ALL products of the following reaction, then upload all drawings to the question at the end. N
A: Given is organic reaction.In which reactants are amine and acid chloride.
Q: CI هنده НО MgBr (2 eq) Et₂0 2. dilute HCI / H₂O -MgBr (1 eq) Et₂0 1. PhMgBr, Et₂0 2. HCI, H₂O
A: Grignard reagent can act as a base to abstract acidic proton first . It can also act as a…
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into…
A: Given :Energy absorbed by the reaction, ∆E= +272KJ where positive sign indicates energy is…
Q: Determine the energy, in J, of a photon with a wavelength of 311 nm. (h = 6.626 x 10-34 J.s and c =…
A:
Q: How would one calculate the protein concentration for each sample if the original protein…
A: Given that, the stock protein solution concentration is Ci = 2 mg/mL.We have to calculate the…
Q: alcohol 000 OH 3.0 3500 2.6 CH (sp³) 3000 2.2 1 1.8 Molecule 6 2500 2000 wavenumber (cm-¹) 1.4 8…
A: Given is IR and 1H NMR spectra of same molecule.
Q: (3) You are asked to bring the pH of 0.500 L of 0.500 mol/L NH4Cl to 7.00. What volume, in drops, (1…
A: The objective of the question is to determine the volume of drops.It is given that,The pH of the…
Q: 3. Show the reaction and products formed when methyl magnesium iodide (CH3-Mg-I) is reacted with…
A: When methyl magnesium iodide is treated with butanal there occurs nucleophilic addition reaction of…
Q: please explain
A: In an E2 reaction beta proton is abstracted and at same time leaving group departs to produce…
Q: If the arrows proceeded in the direction indicated (remember actually though that NH3 is a better…
A: According to the Bronsted Lowry theory, an acid is a compound that when dissolved in water releases…
Q: Calculate the wavelength, in nanometers, of the spectral line produced when an electron in a…
A: The wavelength can be defined as the distance between any two consecutive identical points on the…
Q: Answer There are n a the following questions about the Lewis structure for sulfur trioxide, SO3.…
A:
Q: 5) A gaseous mixture containing 0.250 mol each of H2(g) and I2(g) is introduced into a 4.10 L flask…
A: Initially, the moles of and in the container = the volume of the container, V = The equilibrium…
Q: 31. Use the structure below to identify the configuration (E, Z, cis, or trans) of the labeled…
A: Given is organic compound.The name of given compound is Lovastatin.
Q: Question 5 Draw three major fragments that can result from the following molecule (toulene) on a…
A: The objective of the question is to draw the major fragments of toluene on a mass spectrum.The mass…
Q: H لام :0: Select to Add Arrows .H
A: This reaction is the dehydration reaction of the aldol reaction to form the -unsaturated aldehyde.
Q: Calculate the energy of one photon with a frequency of 5.09 x1014 s-1. What is the energy of a mole…
A:
Q: Predict the major product for this reaction. Ignore inorganic byproducts. 1. BH3-THF 2. H2O2, NaOH…
A: The given reaction is hydroboration oxidation reaction on alkyene
Q: 1. Nitric aud NO Explanation of Reactivity: itiegen NO₂
A: The objective of the question explain the reactivity of and .The most common oxides of nitrogen as…
Q: Where on the x-axis in the mass spectrum will the M+ be at if the molecule whose structure is…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: Which is the organic product for the following reaction? KMnO4 H₂O
A: The alkyl group attached to the benzene ring having at least one hydrogen atom at the key atom can…
bha
Step by step
Solved in 3 steps with 2 images

- Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)Find out 1. (Prove)mechanism,2. order of reaction and 3. effect of use of catalyst and 4 variation of physical properties also discuss 5. imporatnce and 6 uses for Williamson Synthesisprovide the mechanism for the following reaction. only typed solution
- could you show mechanism and products, and explain the function of the "workup" step? ThanksI have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:the 1-pentyne is reacting with CH3CH2CH2Li, then with the ketone, and lastly with NH4Cl and H2O. someone pls explain this with the mechanism. thank you!
- Don't provied handwriting solution. 0Can you please explain the reasonin behind why we split the molecule the way we do to obtain the desired product in addition to showing the mechanism? Thank you!Orgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???! ( plz explain and give mechanism , I will upvote )

