-33 SSM ILW Figure 20-29 shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Volume V. = 8.00V. Process bc is an adiabatic expansion, with p, = 10.0 atm and V, = 1.00 x 10-3 m. For the cycle, find (a) the energy added to the gas as heat, (b) the en- ergy leaving the gas as heat, (c) the net work done by the gas, and (d) the efficiency of the cycle. Adiabatic V. Volume Pressure
-33 SSM ILW Figure 20-29 shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Volume V. = 8.00V. Process bc is an adiabatic expansion, with p, = 10.0 atm and V, = 1.00 x 10-3 m. For the cycle, find (a) the energy added to the gas as heat, (b) the en- ergy leaving the gas as heat, (c) the net work done by the gas, and (d) the efficiency of the cycle. Adiabatic V. Volume Pressure
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 90AP: A cylinder containing three moles of a monatomic ideal gas is heated at a constant pressure of 2...
Related questions
Question
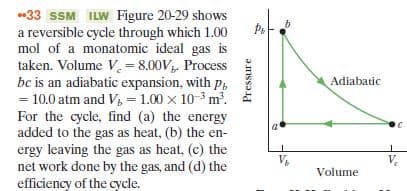
Transcribed Image Text:-33 SSM ILW Figure 20-29 shows
a reversible cycle through which 1.00
mol of a monatomic ideal gas is
taken. Volume V. = 8.00V. Process
bc is an adiabatic expansion, with p,
= 10.0 atm and V, = 1.00 x 10-3 m.
For the cycle, find (a) the energy
added to the gas as heat, (b) the en-
ergy leaving the gas as heat, (c) the
net work done by the gas, and (d) the
efficiency of the cycle.
Adiabatic
V.
Volume
Pressure
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 5 images

Recommended textbooks for you


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning