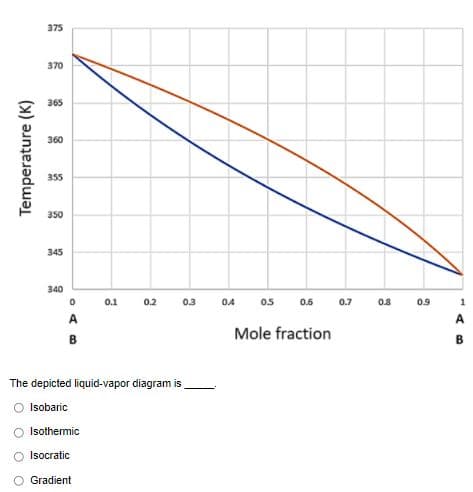
375 370 365 360 355 350 345 340 0.1 0A 0.6 0.7 0.8 0.9 0.2 0.3 05 A A Mole fraction B B The depicted liquid-vapor diagram is O Isobaric O Isothermic Isocratic Gradient Temperature (K)
Q: Which class of organic compound possess a carbonyl group? A) Alcohols B Ketone c) Ether D Epoxide
A: A functional group is an atom or group that determines the chemical behaviour of compound.
Q: What is the IUPAC name for the organic compound shown below? a 1,6-dimethylcyclohexene b ...
A: The naming of organic compounds is done by using rules given by international union of pure and appl...
Q: 7. How do the solubility of esters in water compare to that of comparable alkanes and alcohols. Expl...
A: The organic compounds that can form intermolecular hydrogen with water are water soluble provided h...
Q: Part A Determine the activation barrier for the reaction. Express your answer in units of kilojoules...
A: Given data : 1. Rate constant K1 = 1.26 × 10-4 s-1 2. K2 = 0.226 s-1 3. Temperature T1 = 27oC ...
Q: Rank the following compounds by relative reactivity from most-to-least reactive (i.e., more reactive...
A: Reactivity of carbonyl compound is inversely proportional to steric hinderence
Q: What reagents are used to make the following molecule?
A: Esterification : Carboxylic acid reacts with alcohol in acidic medium gives ester , where acid a...
Q: Some chemicals, like hydrogen peroxide, are prone to decomposition by photocatalysis. How can you pr...
A:
Q: Which of the following is an incorrect IUPAC name for a cycloalkane? a 3,3-dimethyl-cyclopentane...
A: We have to predict the incorrect iupac name.
Q: Question 4 A solution containing 0.584 mole/L of Cuso4 and had a 0.510 absorbance reading in a 1.0cm...
A:
Q: The correct arrangement of figure I is: light source → photomultiplier tube → sample → monoc...
A:
Q: An electron-withdrawing inductive effect generally A Increases acidity of carboxylic acids B Decreas...
A:
Q: Question 8 Which is NOT an appropriate detector for a spectrophotometer? photodiode array photomulti...
A:
Q: Determine the number of sigma bonds, hybridization state and bond order of temozolomide
A:
Q: 2 eq CH3CH20H H30* KMnOд, cold -CH2-CH2-C- H- dilute KMNO4, hot -CH2-CH2- H- H*
A: Major product are obtained by applying appropriate reaction mechanism as show in below steps.
Q: II *公日。 Ce The correct arrangement of figure I is: light source → photomultiplier tube sample → mono...
A:
Q: Balance the following equation with the smallest whole number coefficients. What is the coefficient ...
A: Reaction is :- LiBF4 + H2O ------> H3BO3 + LiF + HF
Q: Which of the following could result in a loss in yield in a mixed solvent recrystallization? i. Use ...
A:
Q: An environmental engineer analyzes a sample of air contaminated with sulfur dioxide. To a 500.-mL sa...
A: Answer: By the use of stoichiometry, we will calculate the number of moles of SO2 and then by the us...
Q: Which of the following will give a negative deviation to Beers Law? I. polychromatic radiation II. p...
A: Explanation for the problem is: Polychromatic radiation causes the Negative deviation of the light. ...
Q: The standard addition method is used to analyze a sample of a river water for mercury. Solution A is...
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: Each row of the table below describes an aque Complete the table. That is, fill in any missing digit...
A: Given,
Q: A five-fold dilution was performed on a standard solution. The diluted solution will have a higher _...
A: We have to predict the correct option about diluted solution.
Q: Which part in the figure can be a photovoltaic cell, phototube or photomultiplier tube? A ...
A: photovoltaic cell, phototube or photomultiplier tube are used as detector in spectrophotometer.
Q: A в D Which part in the figure can be a grating or prism? O A ов
A: There are four parts in the given diagram. Where A is photovoltaic cell which generates electricity...
Q: All of the following are classified as an instrumental method of analysis EXCEPT: O A. electroanalyt...
A: Answer: Different methods are used In chemical analysis and they can be classified into two categori...
Q: Provide the proper IUPAC or common name for the following compound.
A:
Q: 1. Write structural formulas for the following compounds: а. 2-chlorobutane b. 2,3-dimethylbutane 1,...
A: Answer: Structural formula is a way of representing a molecule in a way where all the atoms have bee...
Q: In which of the following ways, absorption is related to transmittance? A. Absorption is the n...
A: We have to find relationship between absorbance and transmittance. We know that :- A = log10(I0/I)
Q: Which of the following will cause an instrumental deviation in Beers Law? I. stray light II. uncle...
A: Answer Which of the following will cause an instrumental deviation in Be...
Q: * 00 Radon (Rn) is the heaviest and the only radioactive member of Group 8A(18), the noble gases. It...
A: Given data No of atoms of radon=10^15 atoms Average atoms=1.373×10^4 Given mass=3.96g Molar mass=...
Q: Acid Rain Formation Acid Rain S02 NOX
A: The picture is showing acid rain Concept.
Q: Question 36 What is could be the calculated lambda max of this compound? O 285 O 323 O 315 O 270
A: Woodward–Fieser rules: The Woodward–Fieser rules are used for the calculation of the wavelength of t...
Q: Some measurements of the initial rate of a certain reaction are given in the table below. N2 H2 init...
A:
Q: What is the IUPAC name of the following compound? CI Br 2-bromo-3-chlorocyclohexene 1-bromo-6-chloro...
A: for IUPAC nomenclature numbering will start from alkene position, hence the carbon which is contain ...
Q: Explain the term polarizability. What type of molecules tends to have high polarizabilities? What is...
A: The property by which the electron cloud of a molecule/ion is distorted by the interaction of other ...
Q: Is my answer correct? Is it okay to put the hydrogen in the back for the fourth carbon despite it be...
A:
Q: Which of the following statements are true of atomic emission spectroscopy (AES)? I. Atoms are prom...
A: AES is a type of spectroscopic technique which uses flame, spark or plasma to calculate the quantity...
Q: For the following reaction, 0.115 moles of aluminum oxide are mixed with 0.332 moles of sulfuric aci...
A:
Q: How many joules is a 2 cycle/sec photon of radiation of a beam of infrared radiation with a waveleng...
A: An infrared electromagnetic radiation has both frequency and wavelength and we are required to find ...
Q: Consider the reaction MnOza) + 4Ha + 2Bra> Mn?"ca + Bra0) + 2H20m At what pH is the voltage zero if ...
A:
Q: Direct oxidation TS zeolite catalyst H202 H20
A: Atom economy means maximizing the incorporation of material from the starting materials or reagents ...
Q: The standard addition method is used to analyze a sample of a river water for mercury. Solution A is...
A: Given, Percent transmittance of solution A = 56 % Percent transmittance of solution B = 33 % Percent...
Q: For the given compound ... a) Identify the 2 structures given as Blank 1 from the given 2 options (s...
A: Answer: Structural isomers are the compounds, those have same molecular formula but different struct...
Q: Which of the following will cause an instrumental deviation in Beers Law? 1 stray light I. unclean o...
A: Beer's law states that the absorption of the solution is directly proportional to the path lenght an...
Q: please help me analyze the mass spectrometry
A: Please find your solution below : Mass spectrometry is a technique which is used to measure mass to ...
Q: 7.
A: Cabonyl carbon is more electrophilic centre than alkene . For addition of nucleophile to alkene , we...
Q: Which of the following is FALSE about the absorbance of a compound? O A. Organic solvents have effec...
A: Ans. (A). In organic chemistry, solvents can have an effect on solubility, stability and reaction ra...
Q: Which of the following statements describe phosphorescence? A. the emission of a photon during...
A:
Q: Refer to the given pair of structure, the structures are
A: In alkenes, the same priority groups are in same side Z-isomer or if the same priority groups are in...
Q: Some measurements of the initial rate of a certain reaction are given in the table below. N2|| |H2| ...
A:
Step by step
Solved in 2 steps with 1 images

- The pressure of the saturated vapor of benzene (vapor in equilibrium with liquid) is 101.32 kPa at 80°C and 4.81 kPa at 5.5°C, respectively. The latter pressure and temperature correspond to the triple point of benzene. Calculate the approximate molar heat of vaporization ∆Hmv of benzene (Clausius - Clapeyron equation).The normal boiling point of liquid pentane is 309 K. Assuming that its molar heat of vaporization is constant at 26.8 kJ/mol, the boiling point of C5H12 when the external pressure is 1.36 atm is K. Please provide only typed answer solution no handwritten solution needed allowedWhat is the change in the boiling point of water at 1000 C per Pa change under atmospheric pressure conditions? The molar enthalpy of vaporization is 40.69 kJ mol-1, the molar volume of liquid water is 0.019 x 10-3 m3 mol-1, and the molar volume of steam is 30.199 x 10-3m3 mol-1, all at 100.000 C and 1.01325 bar. (Hint: change in temperature per change in pressure, dT/dP).
- if ethanol has a vapor pressure of 0.08 bar and a molecular weight of 46 g/mol and water has a vapor pressure of 0.03 bar and a molecular weight of 18 g/mol. c) if the total pressure measured at these conditions is 0.02 bar, what can we conclude from the interaction between ethanol-water, respect to ethanol-ethanol and water-water? Justify your answer briefly.At the pressure of 1.68 atm, the enthalpy of fusion is 4.96 kJ/mol at freezing point 140.83 K and enthalpy of vaporization is 32.82 kJ/mol at boiling point 398.75 K. Calculate the slope dP/dt (atm/K) of the solid-vapor boundary for gas A at 169.07 K. Assume gas A behaves ideally.The normal boiling point of liquid bromoethane is 311k. Assuming that its molar heat of vaporization is constant at 28.7 kj/mol, the boiling point of C2H5Br when the external pressure is 0.698atm is
- Carbon tetrachloride melts at 250 K. The vapor pressure of the liquid is 10539 Pa at 290 K and 74518 Pa at 340 K. The vapor pressure of the solid is 270 Pa at 232 K and 1092 Pa at 250 K. Calculate the normal boiling point and ∆Svaporization at the boiling point and calculate the triple point pressure and temperature.A rigid tank contains water vapour at 250oC and an unknown pressure. When the tank is cooled to 195oC, the vapour starts condensing as shown in the figure. Estimate the initial pressure in the tank.What is w when 1.83 kg of H20(1), initially at 25.0 °C, is converted into water vapour at 185 °C against a constant external pressure of 1.00 atm? Assume that the vapour behaves ideally and that the density of liquid water is 1.00 g/ml (Remember to include a+or-sign as appropriate.) Enter your answer in acceptable SI units, accurate to 3. significant figures. Use an acceptable Sl symbol for units.
- (3A) Calculate the Gibbs free energy of mixing for 50.0 g water and 50.0 g ethanol at constant 298 K and1.00 bar. What is it about your answer that tells vou that this mixing is spontaneous?AmixG = NRT (x1 In x1 + x2 In x2)The liquid-vapour equilibrium diagram of the acetone-chloroform mixture is shown below. A mixture containing 40% (molar percentage) of chloroform was heated to a temperature of 60°C, under a pressure of 1 atm. a) For a temperature of 60°C, what is the molar fraction of chloroform in the liquid phase and what is the molar fraction in the vapor phase, respectively?b) What is the molar fraction of chloroform and acetone at the azeotropic point?It is well known that the temperature at which water freezes to ice or boils at a pressure of 1 atm, are 0°C and 100°C. In the next two tasks, we will now investigate what the temperatures would be at the top of Mount Everest, where the pressure is only 0.337 bar. The standard molar enthalpy of fusion of water at 0°C is ΔfusHmo = 6.008 kJ mol-1 and the standard molar enthalpy of vaporization of water at 100°C is ΔvapHmo = 40.656 kJ mol-1. The density of liquid water at 0°C is ρ(l) = 999.9 kg m-3, while the density of ice at 0°C is only ρ(s) = 916.2 kg m-3. The densities and enthalpies are assumed to be independent of temperature. 1) Calculate the temperature at which ice melts on top of Mount Everest. 2) Calculate the temperature at which water boils on top of Mount Everest.



