4. (a) The sodium ion did not take part in this chemical reaction. What do we call such an ion? (b) Draw a simple diagram which shows how the sodium ion mixes with water in solution. What do we call this physical process?
4. (a) The sodium ion did not take part in this chemical reaction. What do we call such an ion? (b) Draw a simple diagram which shows how the sodium ion mixes with water in solution. What do we call this physical process?
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter8: Solutions
Section: Chapter Questions
Problem 8.5EP: For each of the following pairs of solutions, select the solution for which solute solubility is...
Related questions
Question
(a) The sodium ion did not take part in this
(b) Draw a simple diagram which shows how the sodium ion mixes with water in solution. What do we call this physical process?
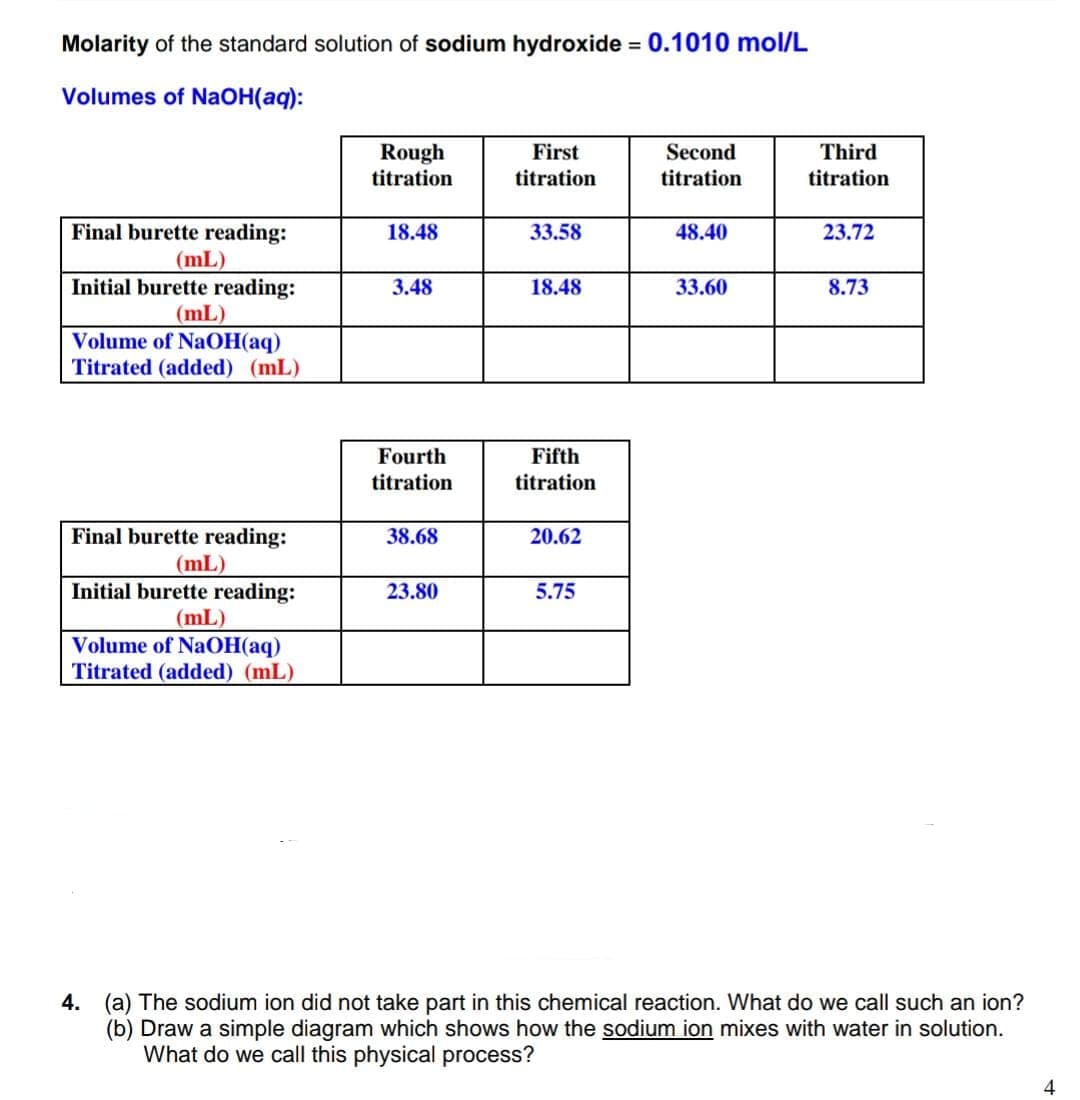
Transcribed Image Text:Molarity of the standard solution of sodium hydroxide = 0.1010 mol/L
%3D
Volumes of NAOH(aq):
Third
Rough
titration
First
Second
titration
titration
titration
Final burette reading:
18.48
33.58
48.40
23.72
(mL)
Initial burette reading:
(mL)
Volume of NaOH(aq)
Titrated (added) (mL)
3.48
18.48
33.60
8.73
Fourth
Fifth
titration
titration
Final burette reading:
(mL)
Initial burette reading:
(mL)
Volume of NaOH(aq)
Titrated (added) (mL)
38.68
20.62
23.80
5.75
4. (a) The sodium ion did not take part in this chemical reaction. What do we call such an ion?
(b) Draw a simple diagram which shows how the sodium ion mixes with water in solution.
What do we call this physical process?
4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning