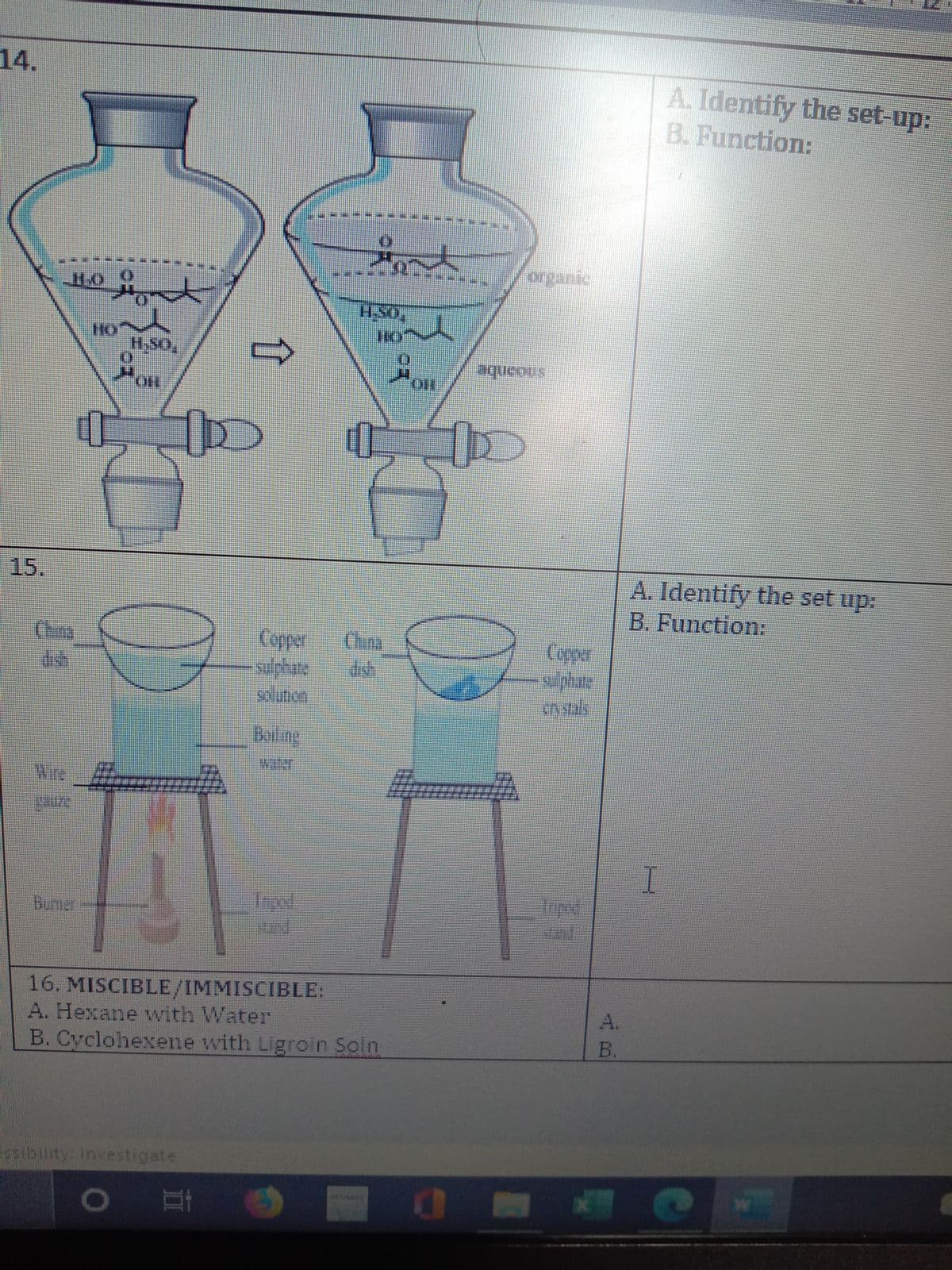
4. A. Identify the set-up: B. Function: organic H.O HO HSO OH snoonbe 15. A. Identify the set up: B. Function: China dish Copper China Copper sul phate cn stals -sulphate dish solution Bailing water Wire gauze Tnped Surd Inpod Nand Burner 16. MISCIBLE/IMMISCIBLE: A. Hexane with Water A. B. Cyclohexene with Ligroin Soln B. sibilit Investigate
Q: what wt. of limestone containing 9.57% Mg must be taken for analysis in order to precipitate of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 32) How many hours will it take to deposit 3.37 g of Al (MM = 27.0 g/mol) from an aqueous AICI,…
A:
Q: Suppose you forget which layer is which, and evaporate both the aqueous layer and the DCM layer.…
A: 7. a) The aqueous layer component that formerly contained NaCl can be recognised by flame test if…
Q: you've been tasked to compound 30 grams of a 0.5% Zinc oxide ointment. you have a stock preparation…
A: The formula used to calculate the amount need to be dilute is as follows: Amount need to be dilute =…
Q: Q/ Describe the preparation of 900 mL of 3.00 M HN03 from the commercial reagent that is 70.5% HNO 3…
A: Volume Of HNO3 = 900 mL Molarity Of HNO3 = 3.00 M Specific gravity of HNO3 = 1.42 g mL-1 %(w/w) by…
Q: Percentage purity of a sample of 0.1350 g of As2O3 assayed iodometrically using 23.5 mL of 0.1055N…
A: Here we have to determine percentage of purity ofAs2O3 in 0.1350 g of sample when it use 23.5 ml of…
Q: 24 Whose hydride hydrolyses in water to give a basic solution? LAL Bond are broken on mixing,…
A: In this given question we have option a)carbon B)silicon C)sulphur D)sodium 24) Answer is option…
Q: A melting "point analysis is a bit of a misnomer; how so? What do we measure when doing such an…
A: Melting point analysis helps in analyzing the purity of the compound.
Q: 12. The species depicted in the Frost diagram will tend to go under what kind of reaction? Ovidon…
A:
Q: How was the precipitate obtained after filtration? (A By scraping from the filter paper and…
A: Filtration- the process in which solid particles in a liquid are removed by the use of a filter…
Q: Cr2072- + Fe2+ Cr3+ + Fe3- quation for alkaline solution:
A: Dear student I have given answer to your question in the image format.
Q: A 20.0 mL sample was analyzed using the Mohr method. Calculate the percent (w/v) CI (FM = 35.45) if…
A:
Q: How many ml of 0.241 M Ba(OH)2 will react with 25.00 mL of 0.115 М НCI?
A: This is an example of acid-base reaction. Ba(OH)2 + HCl -------> BaCl2 + H2O
Q: How was the precipitate obtained after filtration? a. By drying the precipitate on the filter paper…
A: The precipitate is obtained by drying the precipitate on the filter paper using a desiccator.
Q: Calculate the Ionic strength of solution prepared by mixing of 0.2 M NaNO3 and 0.02M of K2Cr2O7: a)…
A: We can calculate the ionic strength of solution by using the formula of ionic strength : I = ionic…
Q: [6] Which of the following may have caused the formation of bands of the components? (base on the…
A: The correct answer about TLC is given below
Q: When the compounds analyzed by Robrob were passed on to the next test, another scientist, Kikoko,…
A: Given: Concentration of 20 % (w/w) solid active compound (M1) = ? Volume of 20 % (w/w) solid active…
Q: 1000 litres of hard H2O is softened by zeolite process. The zeolite was regenerated by passing 20…
A: Volume of hard H2O softened by Zeolite process = 1000 L Strength of NaCl solution = 1500 mg/L= 1.5…
Q: How many milliliters of 2.15 wt% dimethylglyoxime solution should be used to provide a 50.0% excess…
A:
Q: Why is iodine almost always used in a solution containing excess I2?
A: If a standard iodine solution is used as titrant for an oxidizable analyte, the technique is…
Q: HWPrepare one liter of 0.IN solution of H2SO4 concentrated if density is 1.84 and percentage is…
A: Normality of the solution is the number of gram Equivalents of Solute present in one liter of the…
Q: A sample of iron ore weighing 800 mg was treated with HNO3 , boiled to dryness and redissolved in…
A:
Q: Answer all parts of this question a) Discuss THREE therapeutic advantages of microparticulate…
A: The following questions are answered as follows
Q: Formula mass: K2CO3, 138.21; KHCO3, 100.12; K, 39.10; C, 12.01; O, 16.00; and H, 1.01 A 2.126 g…
A: The mass of sample of a solid mixture containing only K2CO3 and KHCO3 is 2.126 g. The volume of…
Q: (a) How many grams of H202 are contained in a solution that requires for titration 14.05 mL of KMNO4…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: 0.105g of the K3[Fe(C2O4)3] (MW= 437.2 g/mol) were dissolved in sulfuric acid and got rated with 9…
A: Balanced Reaction 5 C2O42- + 2 MnO4-+16 H+ →2Mn2+ +10CO2 + 8 H2O So, from this we can determine the.…
Q: The waste liquid solution that forms from the flux and gangue is usually a silicate. True or false?…
A: Given statement: The waste liquid solution that forms from the flux and gangue is usually silicate.…
Q: It is desired to prepare a 250 ppm 500 ml stock copper (II) solution to work at A.A.S. According to…
A:
Q: Potassium iodate, KIO3, was prepared by dissolving 2.023 g of KIO3 in a 500 mL volumetric flask. A…
A:
Q: Plumber's solder is composed of 67% lead and 33% tin by weight. Describe what happens to this…
A: Plumber's solder is composed of 67% lead and 33% tin by weight.
Q: * OWLV2 | Online teachinX C The Deep Blue Compound (+…
A: Concentration of the solution (solution 2) obtained on diluting 0.164 M HCl can be determined as…
Q: Q2- A 30.0 mL H2C204 acidified solution, was treated with 25.0 ml of 0.102 M KMNO, solution. The…
A: The moles of Fe2+ ions can be calculated by - Moles = Molarity × volume in Liter Moles = 0.053 ×(…
Q: 6. The following are disadvantages of using filter paper in gravimetry, which is not included? а.…
A: 6) c is the correct answer. IN the Gravimetry we uses the ashless filter paper to achieve the…
Q: A melting "point" analysis is a bit of a misnomer: how so? What do we measure when doing such an…
A: By the term melting point, we mean that it is a fixed point of temperature where a substance or…
Q: An acidified and reduced iron sample required 40.2 ml of 0.0206 M KMN04. Find mg Fe (at wt =…
A: Reaction occurred is 8H+ + 5 Fe2+ + MnO4- ----------------> 5 Fe3+ + Mn2+ + 4 H2O Volume Of…
Q: Caustic potash that has been exposed to air is found on analysis to contain 90.00% KOH, 2.38% K2CO3…
A:
Q: A bottle of liduckiime C ciBtaoNz Oz 236g|ma). used as da local artestheAichlontarns s0 mt 0t 1…
A: Given data set: Bottle of lidocaine contains 50 ml of a 4.1% solution. Density = 1 g/ml Molar mass…
Q: What is the best procedure for phosphorus extraction from biochar by organic acids?
A: Biochar is defined as charcoal generated by biomass pyrolysis, however is used as a soil improver…
Q: A 0.1017-g sample of KBrO3 (MW = 167 g/mole) was dissolved in dilute HCl and treated with an…
A: In iodometry iodide ion(I-) reacts with oxidising agent and forms iodine which reacts with sodium…
Q: What are the different methods to pack a column? Point out two ways in which thev differ from each…
A: A question based on analytical separations that is to be accomplished.
Q: or each item, explain in one (1) sentence why the step is done in the gravimetric analysis of…
A:
Q: Q2- A 30.0 ml H2C2O4 acidified solution, was treated with 25.0 ml of 0.102 M KMNO4 solution. The…
A: We have to find Molarity of H2C2O4
Q: How many milliliters of 1.5% alcoholic dimethylglyoxime should be used to provide a 36.0% excess…
A:
Q: Which statements are true about gravimetric analysis? The gauze cathode is inert O The final form of…
A: Interpretation: To find true statements regarding gravimetric analysis.
Q: ) What amperage is required to plate out 0.250 mol Cr from a Cr3+ solution in a period of 8.00 h?
A:
Q: Give and explain two precautions in brine solution experiment.
A: Brine solution also known as Sodium Chloride Solution. While doing any experiment involving to brine…
Step by step
Solved in 3 steps

- Any special precautions or purifications of the reagents required? Limiting and excess reagent? Why is excess necessary? 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude…Quickly plezz!! The levigating agent role in Suppositories manufacturing ??( give mechanusm of all plz
- On being heated, the fullerene solvate C60 xCHBr3loses solvent in a two-step process. The final weightloss is 41%. Account for these data and determine x.Thank you It's very well organized. This is part B, I'd appreaciate an explanationNeading help with part a and b The experimental value for part b is 44.65%
- Weight of crude/pure product: 0.819/0.047g Percent yield: Theoretical Yield: Melting Point crude/pure:195/190 Q: What would the theoretical yield froms the Diels-Alder Adduct with your product. Also, what is the actual yield? How do i calculate those? i used anthracene 200mg, maleic anhydride 110 mg and 6ml of xyleneSolution:- 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react with 15.72g cyclohexene to give 1,2-dibromocuclohexanw. Assume 12% excess is needed in order to react completely. a . How much 1,2 -dibromocyclohexane would theoretically be produced ? c. How many ML of the 2.20 M Br2 solution are required?Rec, 1, part1. Provide clear explanation of the method you used
- Hi! can anyone here help me to create a schematic diagram about the experiments: " 21. Preparation of Ethyne" by Pdst Chemistry, and "Activity 10: Functional Group Tests for Hydrocarbons" by FRETZ RMT since i dont know what a schematic diagram is, and what processes/contents should be in it. thank youDescribe every aspect of the procedure clearly and explicitly. Include temperatures, appearance of the reaction (include pictures!). How is the reaction monitored? Is the order of addition important? 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs…1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the above

