Q: Which functional category CANNOT be an ester's isostere? carbamate. amide urethane. pyrrole.
A: isostere groups have same type of physical and chemical properties. They have same number of…
Q: what will be in the HNMR of the following compound?
A: NMR spectroscopy (Nuclear Magnetic Resonance spectroscopy) is a analytical technique that uses the…
Q: 7. Predict the product(s) AND draw the mechanism for the following reaction. TO CH3CH2OH/H2SO4 وی کے…
A: In ring opening of epoxide in presence of acid first protonation of epoxide takes place followed by…
Q: In a laboratory experiment, students synthesized a new compound and found that when 11.52 grams of…
A:
Q: Given the information A+B C 2D D calculate AG at 298 K for the reaction A + B 2C ᎪG° = AH = -641.9…
A: Given : reaction and thermodynamic data
Q: Using IUPAC guidelines, name the following organic compound CH, I CH, CH-C CH Answer: Using IUPAC…
A: we have to determine the IUPAC name of the given compounds
Q: how many joules are required to raise the temperature of 124 grams of water from 21.3 C to 45.6 C
A: According to the question, The mass of the water is given by = 124 grams The initial temperature is…
Q: a. Given the balanced equation: C,H,(g) + 50₂(g) 3CO₂(g) + 4H₂O(g) and the data below, calculate the…
A: Note: Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: Example #6 Balance the following redox reaction which occurs in a basic solution: SO₂2- + MnO → SO²…
A: The unbalanced reaction is, SO32- + MnO4- → SO42- + MnO2 Using half-reaction method, the above…
Q: 6. Propose an efficient synthesis for each of the following transformations: a. b. 8- .OH -NO2 5-3…
A: nitration is a process in which there is the replacement of a hydrogen by nitro groups Kmno4 act as…
Q: The density of helium gas under certain conditions is found to be 0.164 kg/m³. What is the volume in…
A:
Q: Molecule H₂O PC15 Lewis Dot structure Molecule geometry with bond angles Polarity
A: We have to draw the Lewis structures for H2O PCl5
Q: Periodic Table A 107 g -g sample of steam at 100 °C is emitted from a volcano. It condenses, cools,…
A: Answer: In this question we have to find out the heat lost by steam to gets turned into ice. It is a…
Q: The mass percentage of hydrochloric acid within a solution is 10.00%. Given that the density of this…
A: The given data is The mass percentage of hydrochloric acid with in a solution (mm×%) = 10.00%. The…
Q: Give instructions for preparing 500 mL of a 0.15 M solution of NaCl
A: Please find your solution below : Molarity of a solution is a measure of concentration which is…
Q: When solid lead(II) phosphate is in equilibrium with its ions, the ratio of lead(II) ions to…
A: Given : lead(ii) phosphate is at equilibrium with its ions
Q: Final Aqueous Layer Burette Reading Initial Aqueous Layer Burette Reading Final Thiosulfate Burette…
A: For third titration, Initial Thiosulfate burette reading = 6.83 mL Final thiosulfate burette reading…
Q: The density of aluminum is 2,710 kg/m³. What is the volume, in cm³, of a 19.50 g aluminum block
A:
Q: In this lab, you will be provided with a stock solution of food dye. This is the solution from which…
A: The given data is as follows: The concentration of stock solution M1 = 7.000×10-5 mol/L The volume…
Q: The decomposition reaction, C(g) A(g) + 2B(g) has Kc = 41 at 298 K. What is the value of K, for this…
A:
Q: Circle each amino functional group. Amines are weak bases. CH H₂N-CH-COOH CH CH 4-toluidine -NH₂…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Identify the splitting patterns for each non-singlet hydrogen and draw the corresponding qualitative…
A:
Q: A student determined the concentration of total reducible iodine in the aqueous phase based on the…
A: Given: Total reducible iodine in the aqueous phase is 0.0177M. The concentration of I2 in the CH2Cl2…
Q: - Name the following compounds according to IUPAC rules: (1) (2) (3) (4) J
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: Which molecule has two ¹H NMR signals and will produce the following NMR spectrum? 3 II
A: Given : 1H NMR signal
Q: The mass of Fe2O3 isolated from analysis of 14 tablets was 0.282 g. What is the average mass of iron…
A: Molar Mass of Fe2O3 = 159.69 g/mil Hence, 159.69 g of Fe2O3 contains 55.845 g of Fe.
Q: 4. Predict the product(s) for the followi O O 1. excess LiAlH4 2. H₂O
A: LiAlH4 is a reducing agent. The carbonyl C=O gets reduced to C-OH. The lactone gets reduced to two…
Q: Davis performed an experiment similar to Toni's. Davis created a 0.468 m solution of LiCl, which had…
A: In this question we have to calculate the Vant hoff factor (i) for LiCl with the help of given…
Q: what is number of 3d electrons of the transition metal ion in [Ni(acac)2(OH2)2]
A: Transition metal complex [Ni(acac)2(OH)2]
Q: the energy needed to heat 12.6 g of silver from 112 °C to 275 °C
A:
Q: The molar heat of vaporization of water is 42 kJ/mol. How much energy is released by the…
A:
Q: The reaction A + 2B → C has the rate law rate = k[A][B]. By what factor does the rate of reaction…
A: Given, A + 2B ----> C rate = k[A][B] By what factor does the rate of the reaction increase when…
Q: The displacement volume of an automobile engine is 230 in3. What is this volume in liters
A: The displacement volume of an automobile engine is 230 in3 .We have to convert this volume in…
Q: 2NO2 → 2NO + O2 obeys the rate law: Rate = 1.40 x 10–2 M-1 s-1[NO2]2 at 500° K. If the initial…
A:
Q: Use the observation in the first column to answer the question in the second column. observation The…
A: In the given question, there are three subparts. These subparts are regarding the relation between…
Q: Consider the gaseous reaction CO(g) + Cl2(g) ⟷COCl2(g). What is the expression for Kp in terms of…
A:
Q: A certain element consists of two stable isotopes. The first has a mass of 14.0 amu and a percent…
A: The atomic weight of the element with isotope mass and its abundance is calculated using the formula…
Q: what is the solution?
A: Introduction A solution can be anything that can be made by mixing two or more substances. Soda…
Q: Evaluate the specific rate constant for the reaction at the temperature for which the data were…
A:
Q: D. Give the quantum numbers for the last electron of each element given in Part II following the…
A:
Q: In the year 2005, a picture supposedly painted by a famous artist some time after 1475 but before…
A: This is a question based on radiocarbon dating. Here we will use first order reaction rate to…
Q: Name each of the following NO₂ A OH
A: For naming following steps are required First of all identify the longest carbon chain containing…
Q: Atomic mass of a metal can be linked to its specific heat capacity in J/goC units by a simple…
A:
Q: Draw the 2D formula for the following condensed formulas
A:
Q: Which of the following is an alcohol? OH H3CC0 OH3C-NH₂ O NaOH H3C-C-CH3 CH 3 CH 2 OH
A:
Q: reaction vessel mol ¹12, mol 12, and 3.91 mol e at 493°C for the reaction of hydrogen and iodine to…
A: Given -> Volume=V = 13.9 L T = 493°C Mole of H2 = 0.873 mole Mole of I2 = 0.357 mole Mole of…
Q: A reaction mechanism has the following proposed elementary steps: Step 1: A ⇌ B + C Step 2: A + B →…
A: The given steps of the reaction are: Step 1: A⇌B+CStep 2: A+B→DStep 3: 2A+D→C+E In chemical…
Q: A OA OB OC Br OE Br NBS light All of them are possible products Br Products D Br Br.
A:
Q: C12 hv + da. Da ya + + HCI
A:
Q: References Use the References to access important values if needed for this question. Under certain…
A: We know , number of moles = Given Mass / Molecular Mass
Step by step
Solved in 2 steps with 1 images

- The molecular formula of unknown compound B is C10H16.Quantitative brominationof a 0.473-g sample of compound Brequired 48.22mL of a 0.216MBr2/CCl4to produce a color change. How many rings and pi bonds does compound Bcontain?Include any explanations/calculations to justify your answers.Name the following heterocyclic compounds using systematicnomenclature only:describe the mecanisme of cyclopentane monobromation in the correct order Need solution asap
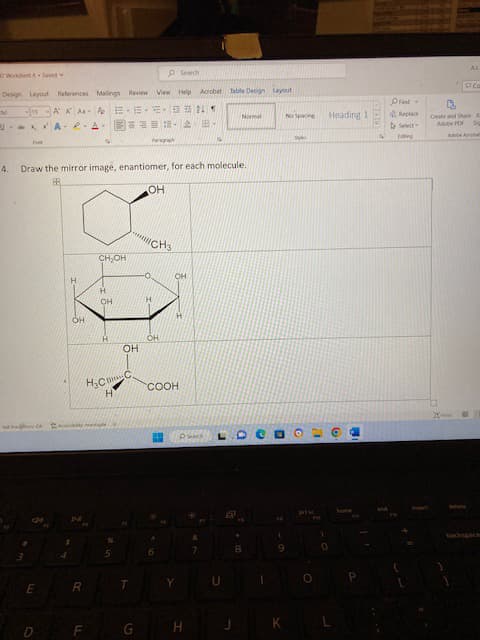
- Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.For the given ee values, calculate the percentage of each enantiomer present.a. 90% eeb. 99% eec. 60% eeFor each ee value (enantiomeric excess value), calculate the percentage of each enantiomer present
- 1) Are the molecules A and B... conformational isomers? Diastereomers? Enantiomers? Position isomers? Non-related? 2) What about the molecules B and C? 3) What about the molecules C and D?What is the correct order for the strenth of IMFA of the following substances? LiCl < H2O < C8H18 H2O < LiCl< C8H18 C8H18 < LiCl < H2O C8H18 < H2O < LiClOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- By considering the stereochemical requirements for E2-elimination, and using an appropriate illustration need help indicating whether the more stable conformer (conformer B) of trans-1 can undergo E2 elimination. Thank you :)The absolute configuration of BAGO is (dextrorotatory, levorotatory, R, S). The melting point of its enantiomer will be (higher, lower, equal) than/with 197.2. The relative configuration of its enantiomer will be (dextrorotatory, levorotatory, R, S). The density of BAGO is (higher, lower, equal, twice) the density of its enantiomer.For the given below ee values, calculate the percentage of each enantiomer present. 60% ee



